Abstract
Background
Although the sentinel lymph node (SLN) hypothesis has been applied to many tissues and organs, liver has remained unstudied. Currently, it is unclear whether hepatic SLNs even exist. If so, they could alter the management of intrahepatic cholangiocarcinoma and other hepatic malignancies by minimizing the extent of surgery while still providing precise nodal staging. This study investigated whether invisible yet tissue-penetrating near-infrared (NIR) fluorescent light can provide simultaneous identification of both the SLN and all other regional lymph nodes (RLNs) in the liver.
Methods
In 25 Yorkshire pigs, this study determined whether SLNs exist in liver and compared the effectiveness of two clinically available NIR fluorophores [methylene blue and indocyanine green (ICG)], and two novel NIR fluorophores previously described by our group (ESNF14 and ZW800-3C) for SLN and RLN mapping.
Results
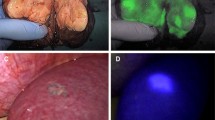
In this study, ESNF14 showed the highest signal-to-background ratio and the longest retention time in SLNs without leakage to second-tier lymph nodes. The findings showed that ICG had apparent leakage to second-tier nodes, and ZW800-3C had poor migration after intraparenchymal injection. However, when injected intravenously, ZW800-3C was able to highlight all RLNs in liver during a 4- to 6-h period. Simultaneous dual-channel imaging of SLN (ESNF14) and RLN (ZW800-3C) permitted unambiguous identification and image-guided resection of SLNs and RLNs in liver.
Conclusion
The NIR imaging technology enables real-time intraoperative identification of SLNs and RLNs in the liver of swine. If these results are confirmed in patients, new strategies for the surgical management of intrahepatic malignancies should be possible.



Similar content being viewed by others
References
Morton DL, Wen DR, Wong JH, et al. Technical details of intraoperative lymphatic mapping for early stage melanoma. Arch Surg. 1992;127:392–9.
Cox CE, Pendas S, Cox JM, et al. Guidelines for sentinel node biopsy and lymphatic mapping of patients with breast cancer. Ann Surg. 1998;227:645–51; discussion 651–643.
Ikai I, Arii S, Okazaki M, et al. Report of the 17th Nationwide Follow-up Survey of Primary Liver Cancer in Japan. Hepatol Res. 2007;37:676–91.
de Jong MC, Nathan H, Sotiropoulos GC, et al. Intrahepatic cholangiocarcinoma: an international multi-institutional analysis of prognostic factors and lymph node assessment. J Clin Oncol. 2011;29:3140–5.
Igami T, Nishio H, Ebata T, et al. Surgical treatment of hilar cholangiocarcinoma in the “new era”: the Nagoya University experience. J Hepatobil Pancreatic Sci. 2010;17:449–54.
Nagino M, Ebata T, Yokoyama Y, et al. Evolution of surgical treatment for perihilar cholangiocarcinoma: a single-center 34-year review of 574 consecutive resections. Ann Surg. 2013;258:129–40.
Mavros MN, Mayo SC, Hyder O, Pawlik TM. A systematic review: treatment and prognosis of patients with fibrolamellar hepatocellular carcinoma. J Am Coll Surg 2012;215:820–30.
Fonseca GM, Varella AD, Coelho FF, Abe ES, Dumarco RB, Herman P. Downstaging and resection after neoadjuvant therapy for fibrolamellar hepatocellular carcinoma. World J Gastrointest Surg. 2014;6:107–11.
Ito K, Ito H, Allen PJ, et al. Adequate lymph node assessment for extrahepatic bile duct adenocarcinoma. Ann Surg. 2010;251:675–81.
Birnbaum DJ, Vigano L, Russolillo N, Langella S, Ferrero A, Capussotti L. Lymph node metastases in patients undergoing surgery for a gallbladder cancer: extension of the lymph node dissection and prognostic value of the lymph node ratio. Ann Surg Oncol. 2014;22(3):811–8.
Ashitate Y, Hyun H, Kim S, et al. Simultaneous mapping of pan and sentinel lymph nodes for real-time image-guided surgery. Theranostics. 2014;4:693–700.
Choi HS, Nasr K, Alyabyev S, et al. Synthesis and in vivo fate of zwitterionic near-infrared fluorophores. Angewandte Chem Int Ed Eng. 2011;50:6258–63.
Choi HS, Ashitate Y, Lee JH, et al. Rapid translocation of nanoparticles from the lung airspaces to the body. Nat Biotechnol. 2010;28:1300–3.
Ashitate Y, Tanaka E, Stockdale A, Choi HS, Frangioni JV. Near-infrared fluorescence imaging of thoracic duct anatomy and function in open surgery and video-assisted thoracic surgery. J Thorac Cardiovasc Surg. 2011;142:31–8; e31–2.
Gioux S, Choi HS, Frangioni JV. Image-guided surgery using invisible near-infrared light: fundamentals of clinical translation. Mol Imaging. 2010;9:237–55.
Troyan SL, Kianzad V, Gibbs-Strauss SL, et al. The FLARE intraoperative near-infrared fluorescence imaging system: a first-in-human clinical trial in breast cancer sentinel lymph node mapping. Ann Surg Oncol. 2009;16:2943–52.
Edge SB, Byrd DR, Compton CC, Fritz AG, Greene FL, Trotti A. AJCC Cancer Staging Manual. 7th ed. New York: Springer; 2009.
Sobin LH, Gospodarowicz M, Wittekind C. TNM classification of malignant tumours. 7th ed. Oxford: Wiley-Blackwell; 2009.
Nakayama A, Bianco AC, Zhang CY, Lowell BB, Frangioni JV. Quantitation of brown adipose tissue perfusion in transgenic mice using near-infrared fluorescence imaging. Mol Imaging. 2003;2:37–49.
Gibbs-Strauss SL, Nasr KA, Fish KM, et al. Nerve-highlighting fluorescent contrast agents for image-guided surgery. Mol Imaging. 2011;10:91–101.
Winer JH, Choi HS, Gibbs-Strauss SL, Ashitate Y, Colson YL, Frangioni JV. Intraoperative localization of insulinoma and normal pancreas using invisible near-infrared fluorescent light. Ann Surg Oncol. 2010;17:1094–100.
Parungo CP, Ohnishi S, Kim SW, et al. Intraoperative identification of esophageal sentinel lymph nodes with near-infrared fluorescence imaging. J Thorac Cardiovasc Surg. 2005;129:844–50.
Schaafsma BE, van der Vorst JR, Gaarenstroom KN, et al. Randomized comparison of near-infrared fluorescence lymphatic tracers for sentinel lymph node mapping of cervical cancer. Gynecol Oncol. 2012;127:126–30.
Vera DR, Wallace AM, Hoh CK, Mattrey RF. A synthetic macromolecule for sentinel node detection: (99m)Tc-DTPA-mannosyl-dextran. J Nucl Med. 2001;42:951–9.
Baker JL, Pu M, Tokin CA, et al. Comparison of [Tc]Tilmanocept and filtered [Tc]sulfur colloid for identification of SLNs in breast cancer patients. Ann Surg Oncol. 2014; 22(1):40–5.
Acknowledgment
We thank Rita G. Laurence for assistance with animal surgery, David J. Burrington Jr for editing, Eugenia Trabucchi for administrative assistance, and Frank Kettenring and Florin Neacsu for assistance with development and maintenance of the FLARE imaging system and software. This study was supported by the following grants from the National Institutes of Health: NCI BRP Grant #R01-CA-115296 (J.V.F.), NIBIB Grant #R01-EB-010022 (J.V.F. and H.S.C.), and NIBIB Grant #R01-EB-011523 (H.S.C. and J.V.F.). The content of this report is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Disclosures
John V. Frangioni, MD, PhD, currently is CEO of Curadel, Curadel ResVet Imaging, and Curadel Surgical Innovations, which have licensed FLARE imaging systems and contrast agents from the Beth Israel Deaconess Medical Center.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wada, H., Hyun, H., Vargas, C. et al. Sentinel Lymph Node Mapping of Liver. Ann Surg Oncol 22 (Suppl 3), 1147–1155 (2015). https://doi.org/10.1245/s10434-015-4601-5
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-015-4601-5