Abstract
Background
Up to 25% of patients with metastatic colorectal cancer (CRC) present with peritoneal carcinomatosis (PC) as the only site of metastases. Complete cytoreductive surgery (CCRS) followed by hyperthermic intraperitoneal chemotherapy (HIPEC) aims for locoregional disease control and long-term survival. Oxaliplatin is effective for treating advanced CRC. This study assesses the safety and efficacy of CCRS with HIPEC with oxaliplatin for patients with PC of CRC.
Methods
A Belgian prospective multicenter registry was performed to monitor perioperative morbidity and assess mortality, disease-free survival (DFS), and overall survival (OS).
Results
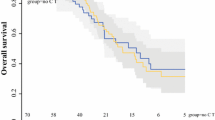
Forty-eight consecutive patients underwent CCRS (R0/1) with HIPEC (male/female ratio 17/31, median age 60 years, range 24–76 years). Median PC index was 11 (range 1–22). Median operation time was 460 (range 125–840) min, with a median blood loss of 475 (range 2–6,000) ml. Thirty-day mortality was 0%. Complication rate (any grade) was 52.1%. Anastomotic leakage occurred in 10.4% of patients, bleeding in 6.3%, and bowel perforation in 2.1%. Median hospital stay was 20 (range 5–65) days. At median follow-up of 22.7 (range 3.2–55.7) months, OS was 97.9% [95% confidence interval (CI) 86.1–99.7] at 1 year and 88.7% (95% CI 73.6–95.4) at 2 years. DFS at 1 year was 65.8% (95% CI 52.3–76.2) and 45.5% (95% CI 34.3–55.9) at 2 years. Median time until recurrence was 19.8 months (95% CI 12–upper limit not defined). Only after dichotomizing PC index was a significant difference in OS found between low and high PC index.
Conclusions
CCRS followed by HIPEC with oxaliplatin for PC from CRC can be implemented with acceptable morbidity. Long-term DFS and OS can be achieved in selected patients.


Similar content being viewed by others
References
Verwaal VJ, Zoetmulder FAN, et al. Long-term survival of peritoneal carcinomatosis of colorectal origin. Ann Surg Oncol. 2005;12:65–71.
Cararo P, Segala M, Cesana D, et al. Obstructing colonic cancer: failure and survival patterns over ten-year follow-up after one-stage curative surgery. Dis Colon Rectum. 2001;44:243–50.
Jayne D, Fook S, Loi C, et al. Peritoneal carcinomatosis from colorectal cancer. Br J Surg. 2002;89:1545–50.
Russell A, Pelton J, Reheis C, et al. Adenocarcinoma of the colon: an autopsy study with implications for new therapeutic strategies. Cancer. 1985;56:1446–51.
Koppe MJ, Bleichrodt RP, et al. Peritoneal carcinomatosis of colorectal origin: incidence and treatment strategies. Ann Surg. 2006;243:212–22.
Sadeghi B, Arvieux C, Glehen O, et al. Peritoneal carcinomatosis from non-gynecological malignancies: results from the EVOCAPE 1 multicentric prospective study. Cancer. 2000;88:358–63.
Shen P, Levine E, Hall J, et al. Factors predicting survival after intraperitoneal hyperthermic chemotherapy with mitomycin C after cytoreductive surgery for patients with peritoneal carcinomatosis. Arch Surg. 2003;138:26–33.
Sugarbaker PH, et al. Management of peritoneal-surface malignancy: the surgeon’s role. Langenbecks Arch Surg. 1999;384:576–87.
Ragnhammar P, Hafstrom L, et al.; SBU-Group. Swedish Council of Technology assessment in health care. A systematic overview of chemotherapy effects in colorectal cancer. Acta Oncol. 2001;40:282–308.
Pestieau SR, Sugarbaker PH, et al. Pharmacokinetics of intraperitoneal oxaliplatin: experimental studies. J Surg Oncol. 2001;76:106–14.
Mahteme H, Pahlman L, et al. Systemic exposure of the parent drug oxaliplatin during hyperthermic intraperitoneal perfusion. Eur J Clin Pharmacol. 2008;64:907–11.
Elias D, Bonnay M, et al. Heated intra-operative intraperitoneal oxaliplatin after complete resection of peritoneal carcinomatosis: pharmacokinetics and tissue distribution. Ann Oncol. 2002;13:267–72.
Jacquet P, Sugarbaker PH, et al. Clinical research methodologies in diagnosis and staging of patients with peritoneal carcinomatosis. In: Sugarbaker PH, editor. Peritoneal carcinomatosis: principles of management. Boston: Kluwer Academic; 1996. p 359–37.
Sugarbaker PH, et al. Peritonectomy procedures. Surg Oncol Clin N Am. 2003;12:703–27.
Harrell F, et al. Regression modeling strategies. New York: Springer; 2001.
Heinze G, Gnant M, Schemper M, et al. Exact logrank test for unequal follow-up. Biometrics. 2003;59:1151–7.
Elias D, Blot F, et al. Curative treatment of peritoneal Carcinomatosis arising from colorectal cancer by complete resection and intraperitoneal chemotherapy. Cancer. 2001;92:71–6.
van Leeuwen B, Graf W, Pahlman L, et al. Swedish experience with peritonectomy and HIPEC. HIPEC in peritoneal carcinomatosis. Ann Surg Oncol. 2007;15:745–53.
Elias D, Glehen O, Pocard M, et al. A comparative study of complete cytoreductive surgery plus intraperitoneal chemotherapy to treat peritoneal dissemination from colon, rectum, small bowel and nonpseudomyxoma appendix. Ann Surg. 2010;251:896–901.
Elias D, Gilly F, Boutitie F, et al. Peritoneal carcinomatosis treated with surgery and perioperative intraperitoneal chemotherapy: retrospective analysis of 523 patients from a multicentric French study. J Clin Oncol. 2010;28:63–8.
Glehen O, Elias D, Deraco M, et al. Cytoreductive surgery combined with perioperative intraperitoneal chemotherapy for the management of peritoneal carcinomatosis from colorectal cancer: a multiinstitutional study. J Clin Oncol. 2004;22:3284–92.
Elias D, Lefevre J, Chevalier J, et al. Complete cytoreductive surgery plus intraperitoneal chemohyperthermia with oxaliplatin for peritoneal carcinomatosis of colorectal origin. J Clin Oncol. 2009;27:681–5.
Elias D, Pocard M, et al. Treatment and prevention of peritoneal carcinomatosis from colorectal cancer. Surg Oncol Clin N Am. 2003;12:543–59.
Cao C, Yan T, Black D, et al. A systematic review and meta-analysis of cytoreductive surgery with perioperative intraperitoneal chemotherapy for peritoneal carcinomatosis of colorectal origin. Ann Surg Oncol. 2009;16:2152–65.
Elias D, Pocard M, Goere D, et al. HIPEC with oxaliplatin in the treatment of peritoneal carcinomatosis of colorectal origin. Cancer Treat Res. 2007;134:303–18.
Andres A, Majno P, Morel P, et al. Improved long-term outcome of surgery for advanced colorectal liver metastases: reasons and implications for management on the basis of a severity score. Ann Surg Oncol. 2008;15:134–43.
Arru M, Aldrighetti L, Castoldi R, et al. Analysis of prognostic factors influencing long-term survival after hepatic resection for metastatic colorectal cancer. World J Surg. 2008;32:93–103.
Choti M, Sitzmann J, Tiburi M, et al. Trends in long-term survival following liver resection for hepatic colorectal metastases. Ann Surg. 2002;235:759–66.
Zakaria S, Donohue JH, Que FG, et al. Hepatic resection for colorectal metastases: value for risk scoring systems? Ann Surg. 2007;246:183–91.
House M, Ito H, Fong Y, et al. Survival after hepatic resection for metastatic colorectal cancer: trends in outcomes for 1,600 patients during two decades at a single institution. J Am Coll Surg. 2010;210:744–52.
Maggiori L, Elias D, et al. Curative treatment of colorectal peritoneal carcinomatosis: current status and future trends. Eur J Surg Oncol. 2010;36:599–603.
Kusamura S, Elias D, Baratti D, et al. Drugs, carrier solutions and temperature in hyperthermic intraperitoneal chemotherapy. J Surg Oncol. 2008;98:247–52.
Verwaal V, van Ruth S, De Bree E, et al. Randomized trial of cytoreduction and hyperthermic intraperitoneal chemotherapy versus systemic chemotherapy and palliative surgery in patients with peritoneal carcinomatosis of colorectal cancer. J Clin Oncol. 2003;21:3737–47.
Verwaal V, Bruijn S, Boot H, et al. 8-Year follow-up of randomized trial: cytoreduction and hyperthermic intraperitoneal chemotherapy versus systemic chemotherapy in patients with peritoneal carcinomatosis of colorectal cancer. Ann Surg Oncol. 2008;15:2426–32.
Franko J, Ibrahim Z, Gusani N, et al. Cytoreductive surgery and hyperthermic intraperitoneal chemoperfusion versus systemic chemotherapy alone for colorectal peritoneal carcinomatosis. Cancer. 2010;116:3756–62.
Hompes D, Boot H, van Tinteren H, et al. Unresectable peritoneal carcinomatosis from colorectal cancer: a single center experience. J Surg Oncol. 2011;104:269–73.
van Ruth S, Mathôt R, Sapridans R, et al. Population pharmacokinetics and pharmacodynamics of mitomycin C during intra-operative hyperthermic intraperitoneal chemotherapy. Clin Pharmacokinet. 2004;43:131–43.
van Ruth S, Verwaal V, Zoetmulder F, et al. Pharmacokinetics of intraperitoneal mitomycin C. Surg Oncol Clin N Am. 2003;12:771–80.
Lambert L, Armstrong T, Mansfield F, et al. Incidence, risk factors and impact of severe neutropenia after hyperthermic intraperitoneal mitomycin C. Ann Surg Oncol. 2009;16:2181–7.
Verwaal VJ, van Tinteren H, Ruth SV, et al. Toxicity of cytoreductive surgery and hyperthermic intra-peritoneal chemotherapy. J Surg Oncol. 2004;85:61–7.
Graham M, Lockwood G, Greenslade D. Clinical pharmacokinetics of oxaliplatin: a critical review. Clin Cancer Res. 2000;6:1205–18.
Soulié P, Raymond E, Brienza S, et al. Oxaliplatin: the first DACH platinum in clinical practice. Bull Cancer. 1997;84:665–73.
de Gramont A, Figer A, Seymour M. Leucovorin and fluorouracil with or without oxaliplatin as first-line treatment in advanced colorectal cancer. J Clin Oncol. 2000;18:2938–47.
Makatsoris T, Kalafonos HP, Aravantinos G. A phase II study of capecitabine plus oxaliplatin (XELOX): a new first-line option in metastatic colorectal cancer. Int J Gastrointest Cancer. 2005;35:103–9.
Scheithauer W, Kornek G, Raderer M. Randomized multicenter phase II trial of two different schedules of capecitabine plus oxaliplatin as first-line treatment in advanced colorectal cancer. J Clin Oncol. 2003;21:1307–12.
Acknowledgment
This study was supported by a research grant from Sanofi-Aventis.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hompes, D., D’Hoore, A., Van Cutsem, E. et al. The Treatment of Peritoneal Carcinomatosis of Colorectal Cancer with Complete Cytoreductive Surgery and Hyperthermic Intraperitoneal Peroperative Chemotherapy (HIPEC) with Oxaliplatin: A Belgian Multicentre Prospective Phase II Clinical Study. Ann Surg Oncol 19, 2186–2194 (2012). https://doi.org/10.1245/s10434-012-2264-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-012-2264-z