Abstract
Background
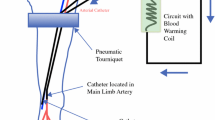
Isolated limb infusion (ILI) is a recently described minimally invasive technique developed in Australia for delivering regional chemotherapy. This study examined the efficacy and toxicity of ILI, compared to hyperthermic isolated limb perfusion (HILP), in treating extremity in-transit melanoma.
Methods
Variables from a prospective single institution database of 120 regionally treated melanoma patients (1995–2007) were compared using chi-square analysis. This included 61 consecutive ILI treatments in 58 patients and 59 HILP treatments in 54 patients. Response was defined at 3 months using the response evaluation criteria in solid tumors (RECIST). ILI was performed using melphalan (LPAM) and dactinomycin for 30 min after limb temperature reached 37°C. HILP was performed using LPAM for 60 min after limb temperature reached 38.5°C.
Results
For ILI (n = 61), the complete response (CR) rate was 30%, the partial response (PR) rate was 14%, and there was no response (NR) in 56% of patients. The median duration of CR was 12 months and 18% of patients experienced (grade ≥3) toxicity. HILP (n = 59) was associated with a better (P < 0.001) response rate (CR 57%, PR 31%, and NR 12%) however, more patients (32%) experienced grade ≥3 toxicity (P = 0.037). The dose of LPAM was corrected for ideal body weight (IBW) in 40 out of 61 ILI procedures, and 13 of 59 HILP procedures. This dosing modification was associated with decreased toxicity (P = 0.024) without diminishing response.
Conclusion
ILI was found to be a well-tolerated alternative to HILP. While ILI does not appear to be as effective as HILP, it does seem to be associated with less morbidity.





Similar content being viewed by others
References
Balch CM, Houghton AN, Peters LJ. (1993) Cutaneous melanoma. In: DeVita VT, Hellman S, Rosenberg SA, eds. Cancer: Principles and Practice of Oncology, 4th edition. JB Lippincott, Philadelphia. p. 1612
Pawlik TW, Ross MI, Johnson MM, et al. Predictors and natural history of in-transit melanoma after sentinel lymphadenectomy. Ann Surg Oncol 2004; 11:1612—61
Minor DR, Allen RE, Alberts D, et al. A clinical and pharmacokinetic study of isolated limb perfusion with heat and melphalan for melanoma. Cancer 1985; 55:2638–44
Di Filippo F, Calabro A, Giannarelli D, et al. Prognostic variables in recurrent limb melanoma treated with hyperthermic aniblastic perfusion. Cancer 1989; 63:2551–61
Klaase JM, Kroon BBR, van Geel AN, et al. Prognostic factors for tumor response and limb recurrence-free interval in patients with advanced melanoma of the limbs treated with regional isolated perfusion with melphalan. Surgery 1994; 115:39–45
Kroon BR, van Geel AN, Benckhuijsen C, et al. Normothermic isolation perfusion with melphalan for advanced melanoma of the limbs. Anticancer Res 1987; 7:4411–2
Fraker DL, Alexader HR, Ross M, et al. A phase III trial of isolated limb infusion perfusion for extremity melanoma comparing melphalan alone versus melphalan plus tumor necrosis factor (TNF-α) plus interferon-gamma (IFN). Ann Surg Oncol 2002; 9:S8
Skene AL, Bulman AS, Williams TF, et al. Hyperthermic isolated limb perfusion with melphalan in the treatment of advanced malignant melanoma of the lower limb. Br J Surg 1990; 77:765–7
Fraker DL, Eggermont AMM. (2003) Hyperthermic regional perfusion for melanoma of the limbs. In: Balch H, Sober S, eds. Cutaneous Melanoma, 14th edition. Quality Medical, St. Louis, MO
Sanki A, Kam PCA, Thompson JF. Long-term results of hyperthermic, isolated limb perfusion for melanoma. Ann Surg 2007; 245:591–6
Thompson JF, Kam PC, Waugh RC, et al. Isolated limb infusion with cytotoxic agents: a simple alternative to isolated limb perfusion. Semin Surg Oncol 1998;14:238–47
Thompson JF, Waugh RC, Saw RPM, et al. Isolated limb infusion with melphalan for recurrent limb melanoma: a simple alternative to isolated limb perfusion. Regional Cancer Treat 1994;7:188–92
Lindner P, Doubrovsky A, Kam PCA, et al. Prognostic factors after isolated limb infusion with cytotoxic agents for melanoma. Ann Surg Oncol 2002;9:127–36
Cornett WR, McCall LM, Petersen RP, et al. Prospective randomized multicenter trial of hyperthermic isolated limb perfusion with melphalan alone versus melphalan plus tumor necrosis factor: American College of Surgeons Oncology Group Trial Z0020. J Clin Oncol 2006; 24:4196–201
Brady MS, Brown K, Patel A, et al. A phase II trial of isolated limb infusion with melphalan and dactinomycin for regional melanoma and soft tissue sarcoma of the extremity. Ann Surg Oncol 2006;13:1123–9
Aloia TA, Grubbs E, Onaitis M, et al. Predictors of outcome after hyperthermic limb perfusion: role of tumor response. Arch Surg 2005; 140:1115–20
Ehrsson H, Eksborg S, Lindfors A. Quantitative determination of melphalan in plasma by liquid chromatography after derivatization with N-acetylcysteine. J Chromatogr Biomed Appl 1986; 380:220
Balch CM, Buzaid AC, Soong SJ, et al. Final version of the American Joint Committee on Cancer staging system for cutaneous melanoma. J Clin Oncol 2001;19:3635–48
Cheng T-Y, Grubbs E, Abdul-Wahab O, et al. Marked variability of melphalan plasma drug levels during regional hyperthermic isolated limb perfusion. Am J Surg 2003; 186:460–7
Zager JS, Gershenwald JE, Aldrink J, et al. Isolated limb infusion for locally recurrent and in-transit extremity melanoma: combined institutional initial experience. Presented at Society of Surgical Oncology 59th annual cancer symposium. San Diego, CA March 23–26, 2006
Brady MS, Patel A, Brown K, et al. Isolated limb infusion for patients with advanced extremity melanoma and soft tissue sarcoma. Final report of a Phase II trial. Presented at Society of Surgical Oncology 60th annual cancer symposium. Washington, DC March 15–18, 2007
Siemann DW, Chapman M, Beikrich A. Effects of oxygenation and pH on tumor cell response to aklylating chemotherapy. Int J Radiat Oncol Biol Phys 1991; 20:287–9
Stehlin JS. Hyperthermic perfusion with chemotherapy for cancers of the extremity. Surg Gynecol Obstet 1969; 129:305
Skene AL, Bulman AS, Williams TF, et al. Hyperthermic isolated limb perfusion with melphalan in the treatment of advanced malignant melanoma of the lower limb. Br J Surg 1990; 77:765–7
Storm FK, Morton DL. Value of therapeutic hyperthermic limb perfusion in advanced recurrent melanoma of the lower extremity. Am J Surg 1985; 15:32–5
Minor DR, Allen RE, Alberts D, et al. A clinical and pharmacokinetic study of isolated limb perfusion with heat and melphalan for melanoma. Cancer 1985; 55:2638–44
Kroon BB. Regional isolated perfusion in melanoma of the limbs: accomplishments, unsolved problems, future. Eur J Surg Oncol 1998;14:101–10
Abdel-Wahab OI, Grubbs E, Viglianti B. The role of hyperthermia in regional alkylating agent chemotherapy. Clin Can Res 2004;10:5919–29
Hildebrandt B, Wust P, Ahlers O. The cellular and molecular basis of hyperthermia. Crit Rev Oncol/Hematol 2002; 43:33–56
Thompson JF, Hunt JA, Shannon KF, et al. Frequency and duration of remission after isolated limb perfusion for melanoma. Arch Surg 1997; 132:903–7
Wieberdink J, Benckhuijsen C, Braat RP. Dosimetry in isolated perfusion of the limbs by assessment of perfused tissue and grading of toxic tissue reactions. Eur J Cancer Clin Oncol 1982; 18:905–10
Acknowledgements
The authors would like to acknowledge Dr. John Thompson of the Sydney Melanoma Unit for teaching the technique of ILI to our group. Grant Support: This work was supported in part by the Duke Melanoma Research Fund and a Grant from the Institute of Genomic Sciences and Policy at Duke University Medical Center.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Beasley, G.M., Petersen, R.P., Yoo, J. et al. Isolated Limb Infusion for In-Transit Malignant Melanoma of the Extremity: A Well-Tolerated but Less Effective Alternative to Hyperthermic Isolated Limb Perfusion. Ann Surg Oncol 15, 2195–2205 (2008). https://doi.org/10.1245/s10434-008-9988-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-008-9988-9