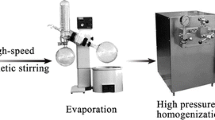
Abstract
The patient-centric strategy urges the pharmaceutical companies to develop orodispersible films (ODF) as a new approach for pediatrics. However, the most common ODF-fabricated method, solvent casting, is facing the safety challenges of safety during manufacturing. To obtain favorable formulations with the ease of use and rapid dissolution, nanotechnology has been accounted for the development process. In this work, we investigated the wet-milling technique in preparing nanocarriers for loratadine—a hydrophobic anti-histamine drug. The results showed that the wet-milling technique could produce nanocarriers at the size of 400 nm. The reduction of particle size induced the increase of solubility and the dissolution rate of loratadine. Moreover, the pre-formulation of nanosized materials could adapt to the preparation of orodispersible films that disintegrated (less than 60s) and dissolved quickly. The DSC results showed that after the milling process, the crystallinity of loratadine was unchanged; however, the reduction in size induced an enhancement of drug bioavailability. After orally administrated to rats, the drug was quickly reached to the blood circulation, just after 30 min. Cmax increased from 44.97 ng/mL for the raw drug to 101.02 ng/mL for the nanocrystal leading to an enhancement of the AUC0-24h by 5.69-fold when the nanocrystal ODF was administrated. The ease of formulation and the improvement of drug solubility as well as bioavailability potentiated orodispersible films as a promising drug delivery for loratadine.

Graphical abstract






Similar content being viewed by others
References
Drumond N. Future perspectives for patient-centric pharmaceutical drug product design with regard to solid oral dosage forms. J Pharm Innov. 2020;15:318–24.
Menditto E, Orlando V, De Rosa G, Minghetti P, Musazzi UM, Cahir C, et al. Patient centric pharmaceutical drug product design—the impact on medication adherence. Pharmaceutics. Multidisciplinary Digital Publishing Institute. 2020;12:44.
Musazzi UM, Khalid GM, Selmin F, Minghetti P, Cilurzo F. Trends in the production methods of orodispersible films. Int J Pharm. 2020;576:118963.
Klingmann V, Pohly CE, Meissner T, Mayatepek E, Möltner A, Flunkert K, et al. Acceptability of an orodispersible film compared to syrup in neonates and infants: a randomized controlled trial. Eur J Pharm Biopharm. 2020;151:239–45.
Orlu M, Ranmal SR, Sheng Y, Tuleu C, Seddon P. Acceptability of orodispersible films for delivery of medicines to infants and preschool children. Drug Deliv. Taylor & Francis. 2017;24:1243–8.
Visser JC, Woerdenbag HJ, Hanff LM, Frijlink HW. Personalized medicine in pediatrics: the clinical potential of orodispersible films. AAPS PharmSciTech. 2017;18:267–72.
Kapoor Y, Kumar K. Structural and clinical impact of anti-allergy agents: an overview. Bioorganic Chem. 2020;94:103351.
Li J, Zhou Y, Aisha M, Wu J, Wang H, Huang F, et al. Preparation of loratadine nanocrystal tablets to improve the solubility and dissolution for enhanced oral bioavailability. J Pharm Pharmacol. 2021;73:937–46.
Madhav KV, Kishan V. Self microemulsifying particles of loratadine for improved oral bioavailability: preparation, characterization and in vivo evaluation. J Pharm Investig. 2018;48:497–508.
Arya A, Sharma V, Pathak K. Pharmaceutical evaluation and dynamic vapor sorption studies of fast dissolving intraoral films of loratadine. Pharm Dev Technol. Taylor & Francis. 2013;18:1329–38.
Centkowska K, Ławrecka E, Sznitowska M. Technology of orodispersible polymer films with micronized loratadine—influence of different drug loadings on film properties. Pharmaceutics. Multidisciplinary Digital Publishing Institute. 2020;12:250.
Steiner D, Finke JH, Kwade A. Efficient production of nanoparticle-loaded orodispersible films by process integration in a stirred media mill. Int J Pharm. 2016;511:804–13.
Oh B-C, Jin G, Park C, Park J-B, Lee B-J. Preparation and evaluation of identifiable quick response (QR)-coded orodispersible films using 3D printer with directly feeding nozzle. Int J Pharm. 2020;584:119405.
Musazzi UM, Selmin F, Ortenzi MA, Mohammed GK, Franzé S, Minghetti P, et al. Personalized orodispersible films by hot melt ram extrusion 3D printing. Int J Pharm. 2018;551:52–9.
Alsofany JM, Hamza MY, Abdelbary AA. Fabrication of nanosuspension directly loaded fast-dissolving films for enhanced oral bioavailability of olmesartan medoxomil: in vitro characterization and pharmacokinetic evaluation in healthy human volunteers. AAPS PharmSciTech. 2018;19:2118–32.
Foo WC, Khong YM, Gokhale R, Chan SY. A novel unit-dose approach for the pharmaceutical compounding of an orodispersible film. Int J Pharm. 2018;539:165–74.
Abbasnezhad N, Zirak N, Shirinbayan M, Tcharkhtchi A, Bakir F. On the importance of physical and mechanical properties of PLGA films during drug release. J Drug Deliv Sci Technol. 2021;63:102446.
Nguyen OOT, Tran KD, Ha NT, Doan SM, Dinh TTH, Tran TH. Oral cavity: an open horizon for nanopharmaceuticals. J Pharm Investig. 2021;51:413–24.
Muller RH, Keck CM. Challenges and solutions for the delivery of biotech drugs – a review of drug nanocrystal technology and lipid nanoparticles. J Biotechnol. 2004;113:151–70.
Desai P, Thakkar A, Ann D, Wang J, Prabhu S. Loratadine self-microemulsifying drug delivery systems (SMEDDS) in combination with sulforaphane for the synergistic chemoprevention of pancreatic cancer. Drug Deliv Transl Res. 2019;9:641–51.
Sarheed O, Shouqair D, Ramesh K, Amin M, Boateng J, Drechsler M. Physicochemical characteristics and in vitro permeation of loratadine solid lipid nanoparticles for transdermal delivery. Ther Deliv. Future Science. 2020;11:685–700.
Alshweiat A, Csóka I, Tömösi F, Janáky T, Kovács A, Gáspár R, et al. Nasal delivery of nanosuspension-based mucoadhesive formulation with improved bioavailability of loratadine: preparation, characterization, and in vivo evaluation. Int J Pharm. 2020;579:119166.
Zhu Y, You X, Huang K, Raza F, Lu X, Chen Y, et al. Effect of taste masking technology on fast dissolving oral film: dissolution rate and bioavailability. Nanotechnology. IOP Publishing. 2018;29:304001.
Funding
This research is funded by PHENIKAA University under grant number 2-01.2020.01
Author information
Authors and Affiliations
Contributions
Khanh Van Nguyen: design of the work; acquisition, analysis, and interpretation of data for the work
Huyen Thi Nguyen: acquisition, analysis, and interpretation of data for the work
Lien Thi Ha Nghiem: acquisition, analysis, and interpretation of data for the work
Mao Van Can: acquisition, analysis, and interpretation of data for the work
Tuan Hiep Tran: design of the work; analysis, interpretation of data for the work; drafting the work
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 50 kb)
Rights and permissions
About this article
Cite this article
Van Nguyen, K., Nguyen, H.T., Nghiem, L.H.T. et al. Nanosized-Loratadine Embedded Orodispersible Films for Enhanced Bioavailability: Scalable Preparations and Characterizations. AAPS PharmSciTech 23, 78 (2022). https://doi.org/10.1208/s12249-022-02230-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1208/s12249-022-02230-y