Abstract
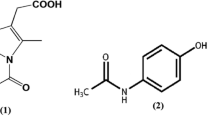
The crystalline states of cimetidine and piroxicam, when coprecipitated from solvents containing 1:1 mole ratio, were transformed to amorphous states as observed using powder X-ray diffraction (PXRD). Amorphous forms of drugs generally exhibit higher water solubility than crystalline forms. It is therefore interesting to investigate the interactions that cause the transformation of both the crystalline drugs. Intermolecular interactions between the drugs were determined using Fourier-transform infrared spectroscopy (FTIR) and solid-state 13C CP/MAS NMR. Molecular dynamic (MD) simulation was performed for the first time for this type of study to indicate the specific groups involved in the interactions based on radial distribution function (RDF) analyses. RDF is a useful tool to describe the average density of atoms at a distance from a specified atom. FTIR spectra revealed a shift of the C≡N stretching band of cimetidine. The 13C CP/MAS NMR spectra indicated downfield shifts of C11, C15 and C7 of piroxicam. RDF analyses indicated that intermolecular interactions occurred between the amide oxygen atom as well as the pyridyl nitrogen of piroxicam and H-N3 of cimetidine. The hydrogen atom (O–H) at C7 interacts with the N1 of cimetidine. Since the MD simulation results are consistent with, and complementary to the experimental analyses, such simulations could provide a novel strategy for investigating specific interacting groups of drugs in coprecipitates, or in amorphous mixtures.











Similar content being viewed by others
References
Morre DJ, Morre DM. tNOX, an alternative target to COX-2 to explain the anticancer activities of non-steroidal anti-inflammatory drugs (NSAIDS). Mol Cell Biochem. 2006;283:159–67.
Knottenbelt C, Chambers G, Gault E, Argyle DJ. The in vitro effects of piroxicam and meloxicam on canine cell lines. J Small Anim Pract. 2006;47:14–20.
Semble EL, Wu WC. Antiinflammatory drugs and gastric mucosal damage. Semin Arthritis Rheum. 1987;16:271–86.
Finkelstein W, Isselbacher KJ. Drug therapy: cimetidine. N Engl J Med. 1978;299:992–6.
Maciel HP, Cardoso LG, Ferreira LR, Perazzo FF, Carvalho JC. Anti-inflammatory and ulcerogenic effects of indomethacin and tenoxicam in combination with cimetidine. Inflammopharmacol. 2004;12:203–10.
Takahashi HK, Watanabe T, Yokoyama A, Iwagaki H, Yoshino T, Tanaka N, et al. Cimetidine induces interleukin-18 production through H2-agonist activity in monocytes. Mol Pharmacol. 2006;70:450–3.
Lefranc F, Yeaton P, Brotchi J, Kiss R. Cimetidine, an unexpected anti-tumor agent, and its potential for the treatment of glioblastoma (review). Int J Oncol. 2006;28:1021–30.
Milligan PA, McGill PE, Howden CW, Kelman AW, Whiting B. The consequences of H2 receptor antagonist-piroxicam coadministration in patients with joint disorders. Eur J Clin Pharmacol. 1993;45:507–12.
Said SA, Foda AM. Influence of cimetidine on the pharmacokinetics of piroxicam in rat and man. Arzneimittelforschung. 1989;39:790–2.
Guo W, Hamilton JA. Phase behavior and crystalline structures of cholesteryl ester mixtures:A C-13 MASNMR study. Biophys J. 1995;68:2376–86.
Yamamura S, Gotoh H, Sakamoto Y, Momose Y. Physicochemical properties of amorphous salt of cimetidine and diflunisal system. Int J Pharm. 2002;241:213–21.
Hancock BC, Parks M. What is the true solubility advantage for amorphous pharmaceuticals? Pharm Res. 2000;17:397–404.
Liu J, Qiu L, Gao J, Jin Y. Preparation, characterization and in vivo evaluation of formulation of baicalein with hydroxypropyl-beta-cyclodextrin. Int J Pharm. 2006;312:137–43.
Zmitek J, Smidovnik A, Fir M, Prosek M, Zmitek K, Walczak J, et al. Relative bioavailability of two forms of a novel water-soluble coenzyme Q10. Ann Nutr Metab. 2008;52:281–7.
Tantishaiyakul V, Kaewnopparat N, Ingkatawornwong S. Properties of solid dispersions of piroxicam in polyvinylpyrrolidone K-30. Int J Pharm. 1996;143:59–66.
Tantishaiyakul V, Kaewnopparat N, Ingkatawornwong S. Properties of solid dispersions of piroxicam in polyvinylpyrrolidone. Int J Pharm. 1999;181:143–51.
Valizadeh H, Zakeri-Milani P, Barzegar-Jalali M, Mohammadi G, Danesh-Bahreini MA, Adibkia K, et al. Preparation and characterization of solid dispersions of piroxicam with hydrophilic carriers. Drug Dev Ind Pharm. 2007;33:45–56.
Redenti E, Zanol M, Ventura P, Fronza G, Comotti A, Taddei P, et al. Raman and solid state 13C-NMR investigation of the structure of the 1:1 amorphous piroxicam:β-cyclodextrin inclusion compound. Biospectroscopy. 1999;5:243–51.
Tantishaiyakul V, Songkro S, Suknuntha K, Permkum P, Pipatwarakul P. Crystal structure transformations and dissolution studies of cimetidine–piroxicam coprecipitates and physical mixtures. AAPS PharmSciTech. 2009;10:789–95.
Okonogi S, Puttipipatkhachorn S. Dissolution improvement of high drug-loaded solid dispersion. AAPS PharmSciTech. 2006;7:E1–6.
Mashru RC, Sutariya VB, Sankalia MG, Yagnakumar P. Characterization of solid dispersions of rofecoxib using differential scanning calorimeter. JTherm Anal Calorim. 2005;82:167–70.
Schachter DM, Xiong J, Tirol GC. Solid state NMR perspective of drug-polymer solid solutions: a model system based on poly(ethylene oxide). Int J Pharm. 2004;281:89–101.
Suknuntha K, Tantishaiyakul V, Vao-Soongnern V, Espidel Y, Cosgrove T. Molecular modeling simulation and experimental measurements to characterize chitosan and poly(vinyl pyrrolidone) blend interactions. J Polym Sci Polym Phys. 2008;46:1258–64.
Sandoval C, Castro C, Gargallo L, Radic D, Freire J. Specific interactions in blends containing chitosan and functionalized polymers. Molecular dynamics simulations. Polymer. 2005;46:10437–42.
Suga Y, Takahama T. Application of molecular simulation to prediction of solubility parameter. Chem Lett. 1996;281–2.
Jacobson SH. Molecular modeling studies of polymeric transdermal adhesives: structure and transport mechanisms. Pharm Technol. 1999;122–30.
Li B, Pan F, Fang Z, Liu L, Jiang Z. Molecular dynamics simulation of diffusion behavior of benzene/water in PDMS-calix [4] arene hybrid pervaporation membranes. Ind Eng Chem Res. 2008;47:4440–7.
Heuchel M, Hofmann D, Pullumbi P. Molecular modeling of small-molecule permeation in polyimides and its correlation to free-volume distributions. Macromolecules. 2004;37:201–14.
Vrecer F, Vrbinc M, Meden A. Characterization of piroxicam crystal modifications. Int J Pharm. 2003;256:3–15.
Sheth AR, Bates S, Muller FX, Grant DJW. Polymorphism in piroxicam. Cryst Growth Des. 2004;4:1091–8.
Baranska M, Proniewicz LM. FT-IR and FT-Raman spectra of cimetidine and its metallocomplexes. J Mol Struct. 1999;511–512:153–62.
Shibata M, Kokubo H, Morimoto K, Morisaka K, Ishida T, Inoue M. X-ray structural studies and physicochemical properties of cimetidine polymorphism. J Pharm Sci. 1983;72:1436–42.
Sheth AR, Bates S, Muller FX, Grant DJW. Local structure in amorphous phases of piroxicam from powder X-ray diffractometry. Cryst Growth Des. 2005;5:571–8.
Rigby D. Fluid density predictions using the COMPASS force field. Fluid Phase Equilib. 2004;217:77–87.
Mazeau K, Heux L. Molecular dynamics simulations of bulk native crystalline and amorphous structures of cellulose. J Phys Chem B. 2003;107:2394–403.
Tantishaiyakul V, Permkam P, Suknuntha K. Use of DRIFTS and PLS for the determination of polymorphs of piroxicam alone and in combination with pharmaceutical excipients: a technical note. AAPS PharmSciTech. 2008;9:95–9.
Hegedus B, Gorog S. The polymorphism of cimetidine. J Pharm Biomed Anal. 1985;3:303–13.
Middleton DA, Duff CSL, Berst F, Reid DG. A cross-polarization magic-angle spinning 13C NMR characterization of the stable solid-state forms of cimetidine. J Pharm Sci. 1997;86:1400–2.
Sheth AR, Lubach JW, Munson EJ, Muller FX, Grant DJW. Mechanochromism of piroxicam accompanied by intermolecular proton transfer probed by spectroscopic methods and solid-phase changes. J Am Chem Soc. 2005;127:6641–51.
Geckle JM, Rescek DM, Whipple EB. Zwitterionic piroxicam in polar solution. Magn Reson Chem. 1989;27:150–4.
Karpinska G, Dobrowolski JC, Mazurek AP. Conformation and tautomerism of the cimetidine molecule: a theoretical study. J Mol Struct. 2003;645:37–43.
Taddei P, Torreggiani A, Simoni R. Influence of environment on piroxicam polymorphism: vibrational spectroscopic study. Biopolymers. 2000;62:68–78.
Middleton DA, Duff CSL, Peng X, Reid DG, Saunders D. Molecular conformations of the polymorphic forms of cimetidine from 13C solid-state NMR distance and angle measurements. J Am Chem Soc. 2000;122:1161–70.
Birkedal H, Bauer-Brandl A, Pattison P. The surprising polymorph C of cimetidine: synchrotron radiation to the rescue. XIXth European Crystallographic Meeting. Nancy, France: Abstracts. Acta Cryst. 2000;A56:s337. Supplement.
Acknowledgments
This work was supported by Prince of Songkla University, PSU-Grid and the National Nanotechnology Center (NANOTEC), National Science and Technology Development Agency (NSTDA), Ministry of Science and Technology through its National Nanoscience Consortium (CNC). The authors would like to thank Professor Terrence Cosgrove and Dr. Youssef Espidel for 13C CP/MAS NMR experiments.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tantishaiyakul, V., Suknuntha, K. & Vao-Soongnern, V. Characterization of Cimetidine–Piroxicam Coprecipitate Interaction Using Experimental Studies and Molecular Dynamic Simulations. AAPS PharmSciTech 11, 952–958 (2010). https://doi.org/10.1208/s12249-010-9461-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12249-010-9461-5