Abstract
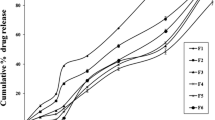
The purpose of the present research was to produce a quick/slow biphasic delivery system for ibuprofen. A dual-component tablet made of a sustained release tableted core and an immediate release tableted coat was prepared by direct compression. Both the core and the coat contained a model drug (ibuprofen). The sustained release effect was achieved with a polymer (hydroxypropyl methylcellulose [HPMC] or ethylcellulose) to modulate the release of the drug. The in vitro drug release profile from these tablets showed the desired biphasic release behavior: the ibuprofen contained in the fast releasing component was dissolved within 2 minutes, whereas the drug in the core tablet was released at different times (⊂16 or >24 hours), depending on the composition of the matrix tablet. Based on the release kinetic parameters calculated, it can be concluded that the HPMC core was suitable for providing a constant and controlled release (zero order) for a long period of time.
Similar content being viewed by others
References
Maggi L, Machiste EO, Torre ML, Conte U. Formulation of biphasic release tablets containing slightly soluble drugs.Eur J Pharm Biopharm. 1999;48:37–42.
Colombo P, Conte U, Gazzaniga A, et al. Drug release modulation by physical restrictions of matrix swelling.Int J Pharm. 1990;63:43–48.
Qiu Y, Chidambaram N, Flood K. Design and evaluation of layered diffusional matrices for zero-order sustained release.J Control Release. 1998;52:123–130.
Chidambaram N, Porter W, Flood K, Qui Y. Formulation and characterization of new layered diffusional matrices for zero-order sustained release.J Control Release 1998;52:149–158.
Conte U, Maggi L. A flexible technology for the linear, pulsative and delayed release drugs, allowing for easy accommodation of difficult in vitro targets.J Control Release 2000;64:263–268.
Uekama K, Matsubara K, Abe K, Horiuchi Y, Hirayamma F, Suzuki N. Design and in vitro evaluation of slow-release dosage form of piretanide: utility of beta-cyclodextrin: cellulose derivative combination as a modified-release drug carrier.J Pharm Sci. 1990;79:244–248.
Li Y, Zhu J. Modulation of combined-release behaviours from a novel “tablets-in-capsule system.”J Control Release 2004;95:381–389.
De Brabander C, Vervaet C, Görtz JP, Remon JP, Berlo JA. Bioavailability of ibuprofen from matrix mini-tablets based on a mixture of starch and microcrystallien wax.Int J Pharm. 2000;208:81–86.
Colombo P, Bettini R, Santi P, Peppas NA. Swellable matrices for controlled drug delivery: gel-layer behavior, mechanisms and optimal performance.Pharm Sci Technol Today. 2000;3:198–204.
Kiil S, Dam-Johansen K. Controlled drug delivery from swellable hydroxypropylmethylcellulose matrices: model-based analysis of observed radial front movements.J Control Release. 2003;90:1–21.
Fell JT, Newton JM. The tensile strength of lactose tablets.J Pharm Pharmacol. 1968;20:657–659.
Morre JW, Flanner HH. Mathematical comparison of dissolution profiles.Pharm Technol. 1996;20:64–74.
CDER. Center for Drug Evaluation and Research, Guidance for Industry, Dissolution Testing of Immediate Release Solid Oral Dosage, 1997. Available at:http://www.fda.gov/cder/Guidance/1713bpl.pdf. Accessed: September 19, 2006.
EMEA. European Agency for the Evaluation of Medicinal Products, Human Medicines Evaluation Unit, Note for Guidance on Quality of Modified Release Products: (A) Oral Dosage Forms; (B) Transdermal Dosage Forms; Section I (Quality), CPMP/QWP/604/96 (1999). Available at: http://www.emea.eu.int/pdfs/human/qwp/060496en.pdf. Accessed: September 19, 2006.
Costa P, Sousa Lobo JM. Modelling and comparison of dissolution profiles.Eur J Pharm Sci. 2001;13:123–133.
Ritger PL, Peppas NA. A simple equation for description of solute release. I. Fickian and non-Fickian release from non-swellable devices in the form of slabs, spheres, cylinders or discs.J Control Release. 1987;5:23–36.
Donbrow M, Samuelov Y. Zero order drug delivery from double-layered porous films: release rate profiles from ethylcellulose, hydroxypropylcellulose and polyethylene glycol mixtures.J Pharm Pharmacol. 1980;32:463–470.
Higuchi T. Rate of release of medicaments from ointment bases containing drugs in suspension.J Pharm Sci. 1961;50:874–875.
Higuchi T. Mechanism of sustained-action medication: theoretical analysis of rate of release of soild drugs dispersed in solid matrices.J Pharm Sci. 1963;52:1145–1149.
Korsmeyer RW, Gurny R, Doelker EM, Buri P, Peppas NA. Mechanism of solute release from porous hydrophilic polymers.Int J Pharm. 1983;15:25–35.
Peppas NA. Analysis of Fickian and non-Fickian drug release from polymers.Pharm Acta Helv. 1985;60:110–111.
Jacques CHM, Hopfenberg HB, Stannett V. Super case II transport of organic vapors in glassy polymers. In: Hopfenberger HB, ed.Permeability of Plastic Films and Coatings to Gases, Vapors, and Liquids, New York, NY: Plenum Press; 1974:73–86.
Waterman KC, Fergione MB. Press-coating of immediate release powders onto coated controlled release tablets with adhesives.J Control Release. 2003;89:387–395.
Johansson B, Wikberg M, Ek R, Alderborn G. Compression behaviour and compactability of microcrystalline cellulose pellets in relationship to their pore structure and mechanical properties.Int. J. Pharm. 1995;117:57–73.
Johansson B, Nicklasson F, Alderborn G. Effect of pellet size on degree of deformation and densification during compression and on compactability of microcrystalline cellulose pellets.Int J Pharm. 1998;163:35–48.
Johansson B, Alderborn G. The effect of shape and porosity on the compression behaviour and tablet forming ability of granular materials formed from microcrystalline cellulose.Eur J Pharm Biopharm. 2001;52:347–357.
Tunón A, Borjesson E, Frenning G, Alderborn G. Drug release from reservoir pellet compacted with some excipients of different physical properties.Eur J Pharm Sci. 2003;20:469–479.
Lopes CM, Sousa Lobo JM, Pinto JF, Costa P. Compressed mini-tablets as a biphasic delivery system.Int J Pharm. 2006;323:93–100.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published: September 21, 2007
Rights and permissions
About this article
Cite this article
Lopes, C.M., Sousa Lobo, J.M., Pinto, J.F. et al. Compressed matrix core tablet as a quick/slow dual-component delivery system containing ibuprofen. AAPS PharmSciTech 8, 76 (2007). https://doi.org/10.1208/pt0803076
Received:
Revised:
Accepted:
DOI: https://doi.org/10.1208/pt0803076