Abstract
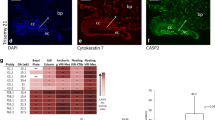
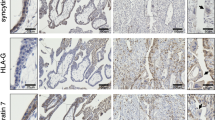
The outer layer of the human placenta is the multinucleated syncytiotrophoblast. The syncytiotrophoblast is formed by the fusion of mononuclear cytotrophoblasts, and aged syncytiotrophoblast nuclei are extruded into the maternal blood as membrane-enclosed “syncytial nuclear aggregates” that are then eliminated from the maternal circulation. Apoptosis proteins are hypothesized to be involved in both of these processes, but the mechanism of death in the syncytiotrophoblast is unclear and death processes in this multinucleated layer are likely to differ from related processes in mononuclear cells. We have used a combination of villous explant culture and immunohistochemical staining of semi-serial sections from the explants to study the changing expression of 4 proteins that are markers of apoptotic processes in first-trimester human placentae. These studies show that Bcl-2 expression is limited to the syncytiotrophoblast and syncytial nuclear aggregates, while conversely Bax is expressed in some cytotrophoblasts. Activated caspase 3 and the M30 cytokeratin neoepitope were localized to isolated regions of the syncytiotrophoblast and some syncytial nuclear aggregates but were never present in the same area. Combining our results with those of others, we suggest a refined scheme whereby proteins of the apoptosis cascade participate in both the processes of syncytial formation and death.
Similar content being viewed by others
References
Kaufmann KBaP. Pathology of the Human Placenta. New York, NY: Edited by Springer-Verlag; 2000.
Attwood HD, Park WW. Embolism to the lungs by trophoblast. J obstet gynaecol Br Commonw. 1961;68:611–617.
Mayhew TM, Leach L, McGee R, Ismail WW, Myklebust R, Lammiman MJ. Proliferation, differentiation and apoptosis in villous trophoblast at 13–41 weeks of gestation (including observations on annulate lamellae and nuclear pore complexes). Placenta. 1999;20(5–6):407–422.
Huppertz B, Frank HG, Kingdom JC, Reister F, Kaufmann P. Villous cytotrophoblast regulation of the syncytial apoptotic cascade in the human placenta. Histochem cell Biol. 1998;110(5):495–508.
Smith SC, Baker PN, Symonds EM. Placental apoptosis in normal human pregnancy. Am J obstet gynecol. 1997;177(1):57–65.
Nelson DM. Apoptotic changes occur in syncytiotrophoblast of human placental villi where fibrin type fibrinoid is deposited at discontinuities in the villous trophoblast. Placenta. 1996;17(7): 387–391.
Wyllie AH, Kerr JF, Currie AR. Cell death: the significance of apoptosis. Int Rev Cytol. 1980;68:251–306.
Straszewski-Chavez SL, Abrahams VM, Mor G. The role of apoptosis in the regulation of trophoblast survival and differentiation during pregnancy. Endocr Rev. 2005;26(7):877–897.
Huppertz B, Kadyrov M, Kingdom JC. Apoptosis and its role in the trophoblast. Am J obstet gynecol. 2006;195(1):29–39.
Huppertz B, Frank HG, Reister F, Kingdom J, Korr H, Kaufmann P. Apoptosis cascade progresses during turnover of human trophoblast: analysis of villous cytotrophoblast and syncytial fragments in vitro. Lab Invest; J Tech Methods Pathol. 1999;79(12):1687–1702.
Black S, Kadyrov M, Kaufmann P, Ugele B, Emans N, Huppertz B. Syncytial fusion of human trophoblast depends on caspase 8. Cell Death Differ. 2004;11(1):90–98.
Fadok VA, Chimini G. The phagocytosis of apoptotic cells. Semin Immunol. 2001;13(6):365–372.
Abumaree MH, Stone PR, Chamley LW. The effects of apoptotic, deported human placental trophoblast on macrophages: possible consequences for pregnancy. J Reprod Immunol. 2006;72(1–2): 33–45.
Mor G, Abrahams VM. Potential role of macrophages as immunoregulators of pregnancy. Reprod Biol Endocrinol: RB&E. 2003;1:119.
Sargent IL, Germain SJ, Sacks GP, Kumar S, Redman CW. Trophoblast deportation and the maternal inflammatory response in pre-eclampsia. J Reprod Immunol. 2003;59(2):153–160.
Boyd H. The Human Placenta. Cambridge, MA: W. Helfer & Sons; 1970.
WJ BJH. The Human Placenta. Cambridge, MA: W. Helfer & Sons; 1970.
Fox H. The significance of villous syncytial knots in the human placenta. J obstet gynaecol Br Commonw. 1965;72:347–355.
Bonney EA, Matzinger P. The maternal immune system’s interaction with circulating fetal cells. J Immunol. 1997;158(1):40–47.
Abumaree MH, Stone PR, Chamley LW. An in vitro model of human placental trophoblast deportation/shedding. Mol Hum Reprod. 2006;12(11):687–694.
Lea RG, al-Sharekh N, Tulppala M, Critchley HO. The immunolocalization of bcl-2 at the maternal-fetal interface in healthy and failing pregnancies. Hum Reprod. 1997;12(1):153–158.
Maruo T, Ishihara N, Samoto T, Murakoshi H, Laoag-Fernandez JB, Matsuo H. Regulation of human trophoblast proliferation and apoptosis during pregnancy. Early Pregnancy. 2001;5(1):28–29.
Ratts VS, Tao XJ, Webster CB, et al. Expression of BCL-2, BAX and BAK in the trophoblast layer of the term human placenta: a unique model of apoptosis within a syncytium. Placenta. 2000;21(4):361–366.
Marzioni D, Muhlhauser J, Crescimanno C, Banita M, Pierleoni C, Castellucci M. BCL-2 expression in the human placenta and its correlation with fibrin deposits. Hum Reprod. 1998;13(6): 1717–1722.
Ka H, Hunt JS. Temporal and spatial patterns of expression of inhibitors of apoptosis in human placentas. Am J pathol. 2003; 163(2):413–422.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Abumaree, M.H., Stone, P.R. & Chamley, L.W. Changes in the Expression of Apoptosis-Related Proteins in the Life Cycle of Human Villous Trophoblast. Reprod. Sci. 19, 597–606 (2012). https://doi.org/10.1177/1933719111428523
Published:
Issue Date:
DOI: https://doi.org/10.1177/1933719111428523