Abstract
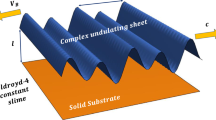
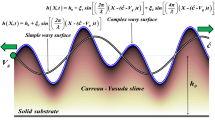
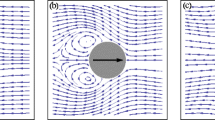
The self-propelling mechanism of rod-shaped bacteria over complex rheological slime is vital. These bacteria glide near the surface by producing waves in their body and secrete an extracellular polymeric substance (EPS) that allows them to move without flagella. During gliding motion, bacteria experience hydrodynamic interactions with complex rheological fluid attached to the surfaces which are either rigid or soft. Their natural response to such interactions affects their gliding speed and power required for propulsion. Motivated by this fact, we investigate the fundamental mechanics of bacterial locomotion by utilizing an undulating surface model combined with the Ellis fluid model. The substrate beneath the organism is considered a wavy (soft substrate) or a rigid surface. The equations of motion are reduced into a single ordinary differential equation under the lubrication approximation. A numerical solution of this equation is computed via the MATLAB bvp5c routine. The code is adjusted in such a way that it refines the unknowns, i.e., flow rate and gliding speed by employing a modified Newton–Raphson algorithm until the satisfaction of equilibrium conditions. The computed pairs of speed and flow rate are then utilized to compute the energy consumed by the glider. Work done by the glider, gliding speed, flow rate, velocity of the slime, and streamlines patterns are also visualized by graphs.




Similar content being viewed by others
Abbreviations
- X, Y :
-
Cartesian coordinate in the fixed frame
- x, y :
-
Cartesian coordinate in the wave frame
- \(V_{g}\) :
-
Gliding speed of the organism
- \(c' \) :
-
Wave speed
- \({\tilde a_G}\) :
-
Amplitude of the wave in glider
- \({\tilde a_S}\) :
-
Amplitude of the wave in substrate
- \(\hbar_{0}\) :
-
Mean distance between wavy sheet to the soft substrate
- \(\phi_{G} ,\,\,\phi_{S}\) :
-
Occlusion parameters
- \(\bar U\) :
-
Velocity vector
- \({\bar U_1},\,{\bar U_2}\) :
-
Components of velocity field in wave frame
- \({\text{A}}_{1}\) :
-
First Rivlin–Ericksen tensor
- \(\frac{d}{dt}\) :
-
Material derivative
- t :
-
Time
- \({\mathbf{T}}\) :
-
Extra stress tensor
- \(Re\) :
-
Reynolds number
- \(Q\) :
-
Flow rate of the fluid
-
 :
: -
Work done by the glider
- \(\rho\) :
-
Density
- \(\nabla\) :
-
Gradient operator
- \(\psi\) :
-
Stream function
- \(\mu\) :
-
Dynamic viscosity
- \(\delta\) :
-
Dimensionless wave number
- \(\lambda\) :
-
Wavelength
- \(\alpha\) :
-
Material constant
- \(\beta\) :
-
Dimensionless material parameter
- \(\Pi\) :
-
Second-order invariant
References
R.P. Burchard, Gliding motility and taxes. In Myxobacteria Springer, New York, NY (1984) 139–161.
J.L. Pate, Gliding motility in procaryotic cells. Can. J. Microbiol. 34(4), 459–465 (1988)
B. Nan, D.R. Zusman, Uncovering the mystery of gliding motility in the myxobacteria. Annu. Rev. Genet. 45, 21–39 (2011)
R.N. Doetsch, G.J. Hageage, Motility in procaryotic organisms: problems, points of view, and perspectives. Biol. Rev. 43(3), 317–362 (1968)
M.J. McBride, Bacterial gliding motility: multiple mechanisms for cell movement over surfaces. Ann. Rev. Microbiol. 55(1), 49–75 (2001)
K.F. Jarrell, M.J. McBride, The surprisingly diverse ways that prokaryotes move. Nat. Rev. Microbiol. 6(6), 466–476 (2008)
R.P. Burchard, Gliding motility of prokaryotes: ultrastructure, physiology, and genetics. Ann. Rev. Microbiol. 35(1), 497–529 (1981)
J.W.F. Costerton, R.G.E. Murray, C.F. Robinow, Observations on the motility and the structure of Vitreoscilla. Can. J. Microbiol. 7(3), 329–339 (1961)
L.N. Halfen, R.W. Castenholz, Gliding in a blue-green alga: a possible mechanism. Nature 225(5238), 1163–1165 (1970)
B.A. Humphrey, M.R. Dickson, K.C. Marshall, Physicochemical and in situ observations on the adhesion of gliding bacteria to surfaces. Arch. Microbiol. 120(3), 231–238 (1979)
N. Read, S. Connell, D.G. Adams, Nanoscale visualization of a fibrillar array in the cell wall of filamentous cyanobacteria and its implications for gliding motility. J. Bacteriol. 189(20), 7361–7366 (2007)
R.W. O’Brien, The gliding motion of a bacterium: Flexibacter strain BH3. ANZIAM J. 23(1), 2–16 (1981)
A.M. Siddiqui, R.P. Burchard, W.H. Schwarz, An undulating surface model for the motility of bacteria gliding on a layer of non-Newtonian slime. Int. J. Non-Linear Mech. 36(5), 743–761 (2001)
T. Hayat, Y. Wang, A.M. Siddiqui, S. Asghar, A mathematical model for the study of gliding motion of bacteria on a layer of non-Newtonian slime. Math. Methods Appl. Sci. 27(12), 1447–1468 (2004)
Y. Wang, T. Hayat, A.M. Siddiqui, Gliding motion of bacteria on power-law slime. Math. Methods Appl. Sci. 28(3), 329–347 (2005)
B. Nan, D.R. Zusman, Novel mechanisms power bacterial gliding motility. Mol. Microbiol. 101(2), 186–193 (2016)
N. Ali, Z. Asghar, O.A. Bég, M. Sajid, Bacterial gliding fluid dynamics on a layer of non-Newtonian slime: Perturbation and numerical study. J. Theor. Biol. 397, 22–32 (2016)
Z. Asghar, N. Ali, M. Sajid, Interaction of gliding motion of bacteria with rheological properties of the slime. Math. Biosci. 290, 31–40 (2017)
E. Lauga, Propulsion in a viscoelastic fluid. Phys. Fluids 19(8), 083104 (2007)
T.R. Ives, A. Morozov, The mechanism of propulsion of a model microswimmer in a viscoelastic fluid next to a solid boundary. Phys. Fluids 29(12), 121612 (2017)
Z. Asghar, N. Ali, O.A. Bég, T. Javed, Rheological effects of micropolar slime on the gliding motility of bacteria with slip boundary condition. Results Phys. 9, 682–691 (2018)
Z. Asghar, N. Ali, M. Sajid, Mechanical effects of complex rheological liquid on a microorganism propelling through a rigid cervical canal: swimming at low Reynolds number. J. Braz. Soc. Mech. Sci. Eng. 40(9), 1–16 (2018)
Z. Asghar, N. Ali, M. Sajid, Analytical and numerical study of creeping flow generated by active spermatozoa bounded within a declined passive tract. Euro. Phys. J. Plus 134(1), 1–15 (2019)
J. Tchoufag, P. Ghosh, C.B. Pogue, B. Nan, K.K. Mandadapu, Mechanisms for bacterial gliding motility on soft substrates. Proc. NAS USA 116(50), 25087–25096 (2019)
N. Ali, M.W.S. Khan, The Graetz problem for the Ellis fluid model. Zeitschrift für Naturforschung A 74(1), 15–24 (2019)
N. Ali, S. Hussain, K. Ullah, & O. A. Bég, Mathematical modelling of two-fluid electro-osmotic peristaltic pumping of an Ellis fluid in an axisymmetric tube. Euro. Phys. J. Plus, 134, 141 (2019).
M. Sajid, M. Rooman, N. Ali, M.N. Sadiq, Flow of the Ellis Fluid in the Renal Tubule. J. Appl. Mech. Tech. Phys. 62(2), 292–299 (2021)
S. Matsuhisa, R.B. Bird, Analytical and numerical solutions for laminar flow of the non-Newtonian ellis fluid. AIChE J. 11(4), 588–595 (1965)
M. M. Bhatti, M.A. Abbas, & M.M. Rashidi, Entropy generation in blood flow with heat and mass transfer for the Ellis fluid model. Heat Transfer Res. 49(8) (2018).
N. Ali, A. Abbasi, I. Ahmad, Channel flow of Ellis fluid due to peristalsis. AIP Adv. 5(9), 097214 (2015)
M. Celli, A. Barletta, P.V. Brandão, Rayleigh-Bénard Instability of an Ellis Fluid Saturating a Porous Medium. Transp. Porous Media 138(3), 679–692 (2021)
M.A. Javed, N. Ali, M. Sajid, A theoretical analysis of the calendering of Ellis fluid. J. Plast. Film Sheeting 33(2), 207–226 (2017)
M.K. Alam, K.N. Memon, A.M. Siddiqui, S.F. Shah, M. Farooq, M. Ayaz, H. Ahmad, Modeling and analysis of high shear viscoelastic Ellis thin liquid film phenomena. Phys. Scr. 96(5), 055201 (2021)
P. Shivapooja, Q. Wang, B. Orihuela, D. Rittschof, G.P. López, X. Zhao, Bioinspired surfaces with dynamic topography for active control of biofouling. Adv. Mater. 25(10), 1430–1434 (2013)
S.I. Abdelsalam, J.X. Velasco-Hernández, A.Z. Zaher, Electro-magnetically modulated self-propulsion of swimming sperms via cervical canal. Biomech. Model. Mechanobiol. 20(3), 861–878 (2021)
K. Ishimoto, E. Lauga, The N-flagella problem: elastohydrodynamic motility transition of multi-flagellated bacteria. Proc. Roy. Soc. A 475(2225), 20180690 (2019)
B. Chan, N.J. Balmforth, A.E. Hosoi, Building a better snail: Lubrication and adhesive locomotion. Phys. Fluids 17(11), 113101 (2005)
Y. Abd Elmaboud, K.S. Mekheimer, S.I. Abdelsalam, A study of nonlinear variable viscosity in finite-length tube with peristalsis. Appl. Bionics Biomech. 11(4), 197–206 (2014)
R. Raza, F. Mabood, R. Naz, S.I. Abdelsalam, Thermal transport of radiative Williamson fluid over stretchable curved surface. Thermal Sci. Eng. Progress 23, 100887 (2021)
Y. Abd Elmaboud, S.I. Abdelsalam, DC/AC magnetohydrodynamic-micropump of a generalized Burger’s fluid in an annulus. Phys. Scr. 94(11), 115209 (2019)
S.R. Elkoumy, E.I. Barakat, S.I. Abdelsalam, Hall and transverse magnetic field effects on peristaltic flow of a Maxwell fluid through a porous medium. Global J. Pure Appl. Math 9(2), 187–203 (2013)
M.M. Bhatti, M. Marin, A. Zeeshan, S.I. Abdelsalam, Recent trends in computational fluid dynamics. Front. Phys. 8, 593111 (2020)
S.I. Abdelsalam, A.Z. Zaher, Leveraging elasticity to uncover the role of rabinowitsch suspension through a wavelike conduit: Consolidated blood suspension application. Mathematics 9(16), 2008 (2021)
I. M., Eldesoky, S. I. Abdelsalam, W. A. El-Askary, & M. M. Ahmed, The integrated thermal effect in conjunction with slip conditions on peristaltically induced particle-fluid transport in a catheterized pipe. J. Porous Media, 23(7) (2020).
M. M. Bhatti, & S. I. Abdelsalam, Bio-inspired peristaltic propulsion of hybrid nanofluid flow with Tantalum (Ta) and Gold (Au) nanoparticles under magnetic effects. Waves Random Complex Media, 1–26 (2021).
K.S. Mekheimer, R.E. Abo-Elkhair, S.I. Abdelsalam, K.K. Ali, A.M.A. Moawad, Biomedical simulations of nanoparticles drug delivery to blood hemodynamics in diseased organs: Synovitis problem. Int. Commun. Heat Mass Transfer 130, 105756 (2022)
Acknowledgements
The constructive comments of the worthy reviewers are greatly appreciated. N. Ali and R. A Shah are thankful to the HEC Pakistan for financial assistance through Grant No: 7671/ Federal/NRPU/ R&D/HEC/2017.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shah, R.A., Asghar, Z. & Ali, N. Mathematical modeling related to bacterial gliding mechanism at low Reynolds number with Ellis Slime. Eur. Phys. J. Plus 137, 600 (2022). https://doi.org/10.1140/epjp/s13360-022-02796-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1140/epjp/s13360-022-02796-3


 :
: