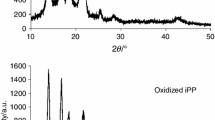
Abstract
The mechanism and kinetics of thermal oxidation of metallocene PP are investigated. It is shown that the rate of oxidation of the samples synthesized at a high temperature (40–70°C) is higher than that of the samples synthesized at a low temperature (20 and 30°C). The composition of oxidation products of PP samples; the kinetics of the accumulation of these products; and changes in structural, thermal, and thermophysical parameters during oxidation are analyzed in detail. Our data indicate that the oxidation of low-temperature samples and the oxidation of high-temperature samples obey different mechanisms. The oxidation of low-temperature samples corresponds to the radical-chain process, in which the intramolecular transfer of kinetic chains prevails. High-temperature samples are characterized by the intermolecular transfer of oxidation kinetic chains, which leads to the degradation of macromolecules. It is inferred that the rate and mechanism of thermal oxidation are determined by the microstructure of polymer chains.
Similar content being viewed by others
References
Metallocene-Based Polyolefins: Preparation, Properties and Technology, Ed. by J. Scheirs and W. Kaminsky (Wiley, Berlin, 2000).
W. Kaminsky, Macromol. Chem. Phys. 209, 459 (2008).
T. V. Monakhova, P. M. Nedorezova, V. I. Tsvetkova, and Yu. A. Shlyapnikov, Polymer Science, Ser. B 46, 113 (2004) [Vysokomol. Soedin., Ser. B 46, 744 (2004)].
H. Nakatani, Sh. Suzuki, T. Tanaka, and M. Terano, Polymer 46, 12366 (2005).
Yu. A. Shlyapnikov, S. G. Kiryushkin, and A. P. Mar’in, Antioxidative Stabilization of Polymers (Khimiya, Moscow, 1986) [in Russian].
L. A. Rishina, L. S. Shibryaeva, E. I. Vizen, et al., Polymer Science, Ser. A 43, 907 (2001) [Vysokomol. Soedin., Ser. A 43, 1472 (2001)].
J. H. Adams, J. Polym. Sci., Part A-1 8, 1077 (1970).
J. Dechant, R. Danz, W. Kimmer, and R. Schmolke, Ultrarotspektroskopische Untersuchungen an Polymeren (Akademie, Berlin, 1972; Nauka, Moscow, 1976).
V. L. Antonovskii and M. M. Buzlanova, Analytical Chemistry of Organic Peroxide Compounds (Khimiya, Moscow, 1978) [in Russian].
Y. V. Kissin, V. I. Tsvetkova, and N. M. Chirkov, Eur. Polym. J. 8, 529 (1972).
L. A. Rishina, Y. V. Kissin, and F. S. Dyachkovsky, Eur. Polym. J. 12, 727 (1976).
B. M. Shchedrin and L. A. Feigin, Kristallografiya 11, 159 (1966).
Yu. S. Lipatov, V. V. Shilov, Yu. P. Gomza, and N. E. Kruglyak, X-Ray Methods of Polymer System Study (Naukova Dumka, Kiev, 1982) [in Russian].
A. Turner-Jones, J. M. Aizewood, and D. R. Beckert, Makromol. Chem. 75, 134 (1964).
A. Turner-Jones, Polymer 12, 487 (1971).
D. I. Svergun and L. A. Feigin, Small-Angle X-Ray and Neutron Scattering (Nauka, Moscow, 1986) [in Russian].
N. M. Emanuel’ and A. L. Buchachenko, Chemical Physics of Polymer Molecular Degradation and Stabilization (Khimiya, Moscow, 1987) [in Russian].
N. Grassie and G. Scott, Polymer Degradation and Stabilization (Cambridge Univ. Press, Cambridge, 1985; Mir, Moscow, 1988).
L. S. Shibryaeva, O. V. Shatalova, A. V. Krivandin, et al., Polymer Science, Ser. A 45, 244 (2003) [Vysokomol. Soedin., Ser. A 45, 424 (2003)].
H. Nakatani, D. Kurniawan, T. Taniike, and M. Terano, Sci. Technol. Adv. Mater. 9, 4401 (2008).
D. Fisher and R. Mulhaupt, Macromol. Chem. Phys. 195, 1433 (1994).
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © L.S. Shibryaeva, L.A. Rishina, O.V. Shatalova, A.V. Krivandin, 2011, published in Russian in Vysokomolekulyarnye Soedineniya, Ser. B, 2011, Vol. 53, No. 12, pp. 2178–2187.
Rights and permissions
About this article
Cite this article
Shibryaeva, L.S., Rishina, L.A., Shatalova, O.V. et al. Thermal oxidation of isotactic polypropylene synthesized with a metallocene catalyst. Polym. Sci. Ser. B 53, 618–625 (2011). https://doi.org/10.1134/S1560090411120050
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1560090411120050