Abstract
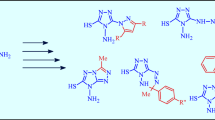
Acylation of 1-methyl-3,3-dialkyl-3,4-dihydroisoquinolines with acid chlorides afforded enaminoketones. Enaminophenylketones from the obtained series were reacted with oxalyl chloride to provide 1-benzoyl-5,5-dialkyl-2,3,5,6- tetrahydropyrrolo[2,1-a]isoquinoline- 2,3-diones. The reaction of the latter with binucleophiles leads to the opening of the pyrrole ring and to the heterocyclization: Under the effect of o-phenyldiamine qinoxaline is formed, 1,2-cyclohexanediamine provides the hexahydro-benzimidazole system, о-aminophenol gives benzoxazole, 3-hydroxy-2-amino-pyridine furnishes oxazolo[4,5-b]pyridine. In the reaction with hydrazine hydrate the acylation occurs with hydrazide formation.
Similar content being viewed by others
References
Shklyaev, V.S., Aleksandrov, B.B., Mikhailovskii, A.G., and Vakhrin, M.I., Chem. Heterocycl. Compd., 1987, vol. 23, p. 790.
Mikhailovskii, A.G., Shklyaev, V.S., and Aleksandrov, B.B., Chem. Heterocycl. Compd., 1990, vol. 26, p. 674.
Mikhailovskii, A.G. and Shklyaev, V.S., Chem. Heterocycl. Compd., 1994, vol. 30, p. 818.
Mikhailovskii, A.G. and Shklyaev, V.S., Chem. Heterocycl. Compd., 1997, vol. 33, p. 243.
Polygalova, N.N. and Mikhailovskii, A.G., Chem. Heterocycl. Compd., 2005, vol. 41, p. 1173.
Polygalova, N.N. and Mikhailovskii, A.G., Chem. Heterocycl. Compd., 2005, vol. 41, p. 1178.
Surikova, O.V., Aliev, Z.G., and Mikhailovskii, A.G., Chem. Heterocycl. Compd., 2008, vol. 44, p. 1500.
Surikova, O.V., Mikhailovskii, A.G., Aleksandrova, G.A., Kir’yanova, I.N., and Vakhrin, M.I., Pharm. Chem. J., 2010, vol. 44, p. 58.
Polygalova, N.N. and Mikhailovskii, A.G., Chem. Heterocycl. Compd., 2004, vol. 40, p. 1220.
Mashevskaya, I.V. and Maslivets, A.N., Chem. Heterocycl. Compd., 2006, vol. 42, p. 1.
Surikova, O.V., Mikhailovskii, A.G., Polygalova, N.N., and Vakhrin, M.I., Russ. J. Org. Chem., 2008, vol. 44, p. 840.
Mikhailovskii, A.G., Surikova, O.V., Chugainov, P.A., and Vakhrin, M.I., Chem. Heterocycl. Compd., 2013, vol. 48, p. 974.
Shklyaev, V.S., Aleksandrov, B.B., Legotkina, G.I., Vakhrin, M.I., Gavrilov, M.S., and Mikhailovskii, A.G., Chem. Heterocycl. Compd., 1983, vol. 19, p. 1242.
Surikova, O.V., Aliev, Z.G., Polygalova, N.N., Mikhailovskii, A.G., and Vakhrin, M.I., Russ. J. Org. Chem., 2008, vol. 44, p. 901.
Surikova, O.V., Mikhailovskii, A.G., Polygalova, N.N., Neifel’d, P.G., and Vakhrin, M.I., Russ. J. Org. Chem., 2007, vol. 43, p. 1413.
Mikhailovskii, A.G. and Dekaprilevich, M.O., Chem. Heterocycl. Compd., 1998, vol. 34, p. 957.
Feuer, H. and Pier, S.M., J. Am. Chem. Soc., 1954, vol. 76, p. 105.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © А.G. Mikhailovskii, А.S. Yusov, О.V. Gashkova, 2016, published in Zhurnal Organicheskoi Khimii, 2016, Vol. 52, No. 2, pp. 240–244.
Rights and permissions
About this article
Cite this article
Mikhailovskii, A.G., Yusov, A.S. & Gashkova, O.V. Synthesis and properties of 1-aroyl-5,5-dialkyl-2,3,5,6-tetrahydropyrrolo[ 2,1-a]isoquinoline-2,3-diones. Russ J Org Chem 52, 223–227 (2016). https://doi.org/10.1134/S1070428016020111
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070428016020111