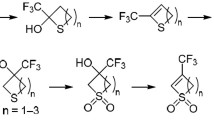
Abstract
In reactions of 4,5-diacetoxy-2-(dinitromethylene)imidazolidine, 4,5-diacetoxy-2-nitriminoimidazolidine, and 1,2-diacetoxy-1,2-bis(chloroacetylamino)ethane with thionyl chloride at room temperature the corresponding cyclic sulfites were obtained. Treating the sulfites with methanol, ethanol, and 2-chloroethanol at room temperature we prepared acyclic ethers in 80–90% yields. Similarly cyclic ethers were synthesized from ethylene glycol and 1,3-propanediol in 50–60% yields.
Similar content being viewed by others
References
Sizova, E.V., Sizov, V.V., and Tselinskii, I.V., Zh. Org. Khim., 2007, vol. 43, p. 1235.
Masaya, I. and Atsushi, M., Japan Patent 2000-159734, 2000; Chem. Abstr., 2000, vol. 133, 17467v.
Ota Seiyaku Co., Ltd., Japan Patent 58 103 343, 1981; Chem. Abstr., 1981, vol. 99, 122022y.
Bolotov, A.A., Rodin, A.A., V’yunov, K.A., Ginak, A.I., and Sarkisov, Yu.S., Zh. Org. Khim., 1982, vol. 18, p. 2060.
Bolotov, A.A., Rodin, A.A., V’yunov, K. A., and Ginak, A.I., Zh. Org. Khim., 1984, vol. 20, p. 1185.
Azuma, N., Takata, T., Sanda, F., and Endo, T. Macromolecules, 1995, vol. 28, p. 7331.
Van Woenden, H.F. Chem. Rev., 1963, vol. 63, no. 6, p. 557.
Vail, S.L., Moran, C.M., and Barker, R.H., J. Org. Chem., 1965, vol. 30, p. 1195.
Tanimoto, S., Imazu, Y., Imuta, M., and Okano, M., Bull. Inst. Chem. Res., Kyoto, Univ., 1972, vol. 50, no. 4, p. 368.
Terpigorev, A.N. and Rudakova, S.B., Zh. Org. Khim., 1998, vol. 34, p. 1078.
Sizova, E.V., Sizov, V.V., Zelenov, M.P., and Tselinskii, I.V., Zh. Org. Khim., 2007, vol. 43, p. 178.
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated to the Full Member of the Russian Academy of Sciences V.A.Tartakovsky on occasion of his 75th birthday
Original Russian Text © E.V. Sizova, V.V. Sizov, I.V. Tselinskii, 2007, published in Zhurnal Organicheskoi Khimii, 2007, Vol. 43, No. 8, pp. 1217–1221.
Rights and permissions
About this article
Cite this article
Sizova, E.V., Sizov, V.V. & Tselinskii, I.V. Synthesis of 1,2-diols ethers, condensation products of glyoxal with nitrogen-containing nucleophiles: I. Reaction of cyclic sulfites with primary alcohols and glycols. Russ J Org Chem 43, 1213–1217 (2007). https://doi.org/10.1134/S1070428007080222
Received:
Issue Date:
DOI: https://doi.org/10.1134/S1070428007080222