Abstract
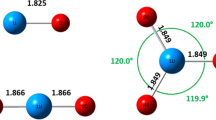
In the framework of Bader’s atoms in molecules theory a complete analysis of the distribution function of electron density in molecules of complexes of Ca2+ and Mg2+ with methylidene rhodanine and its anion was carried out. The role of mutual polarization of the metal cation and the ligand in the formation of coordination bonds was demonstrated. The accumulation of electron density in the interatomic space of coordination bonds is assumed to be a consequence of the deformation of the ligand electron shell under the influence of the cation electric field. Based on the magnitude and sign of the Laplacian and the electron energy density at the critical points of coordination bonds the interactions were classified the in terms of the atoms in molecules theory. The energy of the coordination bonds was evaluated using the Espinoza’s formula. The stability of metal-containing rings was considered basing on the values of the bond ellipticity.
Similar content being viewed by others
References
Lesyk, R.B., Zimenkovsky, B.S., and Troc’ko, N.Y., Ukrainica Bioorganica Acta, 2004, vol. 1, nos. 1–2, p. 29.
Fabretti, A.C., Franchini, G.C., and Peyronel, G., Transition Met. Chem., 1978, vol. 3, no. 1, p. 355.
Fabretti, A.C. and Peyronel, G., Transition Met. Chem., 1977, vol. 2, no. 1, p. 224.
Fabretti, A.C., Peyronel, G., and Franchini, G.C., Transition Met. Chem., 1978, vol. 3, no. 1, p. 125.
Fabretti, A.C. and Peyronel, G., Transition Met. Chem., 1977, vol. 2, no. 1, p. 207.
Tang, E., Yang, G., and Yin, J., Spectrochim. Acta (A)., 2003, vol. 59, no. 3, p. 651.
Loncharich, R.J., Nissen, J.S., and Boyd, D.B., Struct. Chem., 1996, vol. 7, no. 1, p. 37.
Baryshnikov, G.V., Minaev, B.F., and Minaeva, V.A., Zh. Obshch. Khim., 2011, vol. 81, no. 3, p. 481.
Irvine, M.W., Patrick, G.L., Kewney, J., Hastings, S.F., and MacKenzie, S.J., Bioorg. & Med. Chem. Lett., 2008, vol. 18, no. 6, p. 2032.
Boyd, D.B., J. Mol. Struct.: Theochem., 1997, vol. 401, no. 3, p. 227.
Bader, R.W.F., Atoms in Molecules. A Quantum Theory, Oxford: Calendon Press, 1990.
Becke, A.D., J. Chem. Phys., 1993, vol. 98, no. 7, p. 5648.
Lee, C., Yang, W., and Parr, R.G., Phys. Rev. (B), 1988, vol. 37, no. 2, p. 785.
Francl, M.M., Petro, W.J., Hehre, W.J., Binkley, J.S., Gordon, M.S., DeFrees, D.J., and Pople, J.A., J. Chem. Phys., 1982, vol. 77, no. 7, p. 3654.
Bader, R.F.W., Acc. Chem. Res., 1985, vol. 18, no. 1, p. 9.
Bader, R.F.W., Chem. Rev., 1991, vol. 91, no. 5, p. 893.
Frisch, M.J., Trucks, G.W., Schlegel, H.B., Scuseria, G.E., Robb, M.A., Cheeseman, J.R., Montgomeri, J.A., Jr., Vreven, T., Kudin, K.N., Burant, J.C., Millam, J.M., Iyengar, S.S, Tomasi, J., Barone, V., Mennucci, B., Cossi, M., Scalmani, G., Rega, N., Peters-son, G.A., Nakatsuji, H., Hada, M., Ehara, M., Toyota, K., Fukuda, R., Hasegawa, J., Ishida, M., Nakajima, T., Honda, Y., Kitao, O., Nakai, H., Klene, M., Li, X., Knox, J.E., Hratchian, H.P., Cross, J.B., Adamo, C., Jaramillo, J., Gomperts, R., Stratmann, R.E., Yazyev, O., Austin, A.J., Cammi, R., Pomelli, C., Ochterski, J.W., Ayala, P.Y., Morokuma, K., Voth, G.A., Salvador, P., Dannenberg, J.J., Zakrzewski, V.G., Dapprich, S., Daniels, A.D., Strain, M.C., Farkas, O., Malick, D.K., Rabuck, A.D., Raghavachari, K., Foresman, J.B., Ortiz, J.V., Cui, Q., Baboul, A.G., Clifford, S., Cioslowski, J., Stefanov, B.B., Liu, G., Liashenko, A., Piskorz, P., Komaromi, I., Martin, R.L., Fox, D.J., Keith, T., Al-Laham, M.A., Peng, C.Y., Nanayakkara, A., Challacombe, M., Gill, P.M.W., Johnson, B., Chen, W., Wong, M.W., Gonzalez, C., and Pople, J.A., GAUSSIAN 03, Revision, C.02, Gaussian, Inc., Wallingford, CT, 2004.
Keith, T.A., AIMAll (Version 10.07.25); www.aim.tkgristmill.com, 2010.
Abramov, Yu.A., Acta Crystallogr. (A), 1997, vol. 53, no. 3, p. 264.
Bader, R.F.W. and Essen, H., J. Chern. Phys., 1984, vol. 80, no. 5, p. 1943.
Bader, R.F.W., Slee, T.S., Cremer, D., and Kraka, E., J. Am. Chem. Soc., 1983, vol. 105, no. 15, p. 5061.
Cremer, D. and Kraka, E., Croat. Chem. Acta., 1984, vol. 57, no. 6, p. 1259.
Firme, C.L., Antunes, O.A.C., and Esteves, P.M., Chem. Phys. Lett., 2009, vol. 468, nos. 4–6, p. 129.
Espinosa, E., Molins, E., and Lecomte, C., Chem. Phys. Lett., 1998, vol. 285, nos. 3–4, p. 170.
Borissova, A.O., Korlyukov, A.A., Antipin, M.Yu., and Lyssenko, K.A., J. Phys. Chem. (A), 2008, vol. 112, no. 46, p. 11519.
Puntus, L.N., Lyssenko, K.A., Antipin, M.Yu., and Bünzli, J.-C.G., Inorg. Chem., 2008, vol. 47, no. 23, p. 11095.
Lysenko, K.A., Antipin, M.Yu., and Khrustalev, B.H., Izv. Akad. Nauk, Ser. Khim., 2001, vol. 50, no. 9, p. 1465.
Bushmarinov, I.S., Lysenko, K.A., and Antipin, M.Yu., Usp. Khim., 2009, vol. 78, no. 4, p. 307.
Nelyubina, Yu.V., Antipin, M.Yu., and Lysenko, K.A., Usp. Khim., 2010, vol. 79, no. 3, p. 195.
Macchi, P. and Sironi, A., Coord. Chem. Rev., 2003, vols. 238–239, p. 383.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © G.V. Baryshnikov, B.F. Minaev, V.A. Minaeva, A.T. Podgornaya, H. Ågren, 2012, published in Zhurnal Obshchei Khimii, 2012, Vol. 82, No. 7, pp. 1153–1162.
Rights and permissions
About this article
Cite this article
Baryshnikov, G.V., Minaev, B.F., Minaeva, V.A. et al. Application of Bader’s atoms in molecules theory to the description of coordination bonds in the complex compounds of Ca2+ and Mg2+ with methylidene rhodanine and its anion. Russ J Gen Chem 82, 1254–1262 (2012). https://doi.org/10.1134/S1070363212070122
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363212070122