Abstract
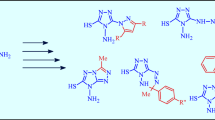
The use of hydrazine as reagent in nucleophilic substitution and reduction in the 1,2,4-triazole series in combination with introduction of labile protecting groups makes it possible to synthesize 5-hydrazino-3-nitro-1H-1,2,4-triazole and 3-chloro-5-hydrazino-1H-1,2,4-triazol-5-ylhydrazine which were difficultly accessible previously, as well as to extend the series of 3-substituted 5-amino-1H-1,2,4-triazoles.
Similar content being viewed by others
References
Garratt, P.J. and Thorn, S.N., Tetrahedron, 1993, vol. 49, no. 1, p. 165.
Caulkett, P.W.R., Jones, G., McPartlin, M., Renshaw, N.D., Stewart, S.K., and Wright, B., J. Chem. Soc., Perkin Trans. 1, 1995, no. 7, p. 801.
Knight, P.D., deMauriac, R.A., and Graham, P.A., US Patent 4128557, 1977; Chem. Abstr., 1979, vol. 90, no. 79146s.
Tolstyakov, V.V. and Tselinskii, I.V., Russ. J. Gen. Chem., 2004, vol. 74, no. 3, p. 399.
Sinegibskaya, A.D., Kovalev, E.G., and Postovskii, I.Ya., Khim. Geterotsikl. Soedin., 1973, no. 12, p. 1708.
Bagal, L.I., Pevzner, M.S., Egorov, A.P., and Samarenko, V.Ya., Khim. Geterotsikl. Soedin., 1970, no. 7, p. 997.
Robins, R.K., Godefroi, E.F., Taylor, E.C., Lewis, L.R., and Jackson, A., J. Am. Chem. Soc., 1961, vol. 83, no. 11, p. 2574.
Bradshow, J.S., Nielsen, R.B., Tse, P.-K., Arena, G., Wilson, B.E., Dalley, N.K., Lamb, J.D., Christensen, J.J., and Izatt, R.M., J. Heterocycl. Chem., 1986, vol. 23, no. 2, p. 361.
Emilsson, H., Lewisson, A., and Selander, H., Acta Pharm. Suec., 1983, vol. 20, no. 3, p. 161.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text V.V. Tolstyakov, I.V. Tselinskii, N.A. Dreving, 2007, published in Zhurnal Obshchei Khimii, 2007, Vol. 77, No. 12, pp. 2034-2040.
Rights and permissions
About this article
Cite this article
Tolstyakov, V.V., Tselinskii, I.V. & Dreving, N.A. Diphenylmethyl and tetrahydropyranyl protecting groups in the synthesis of 3-substituted 5-amino- and 5-hydrazino-1,2,4-triazoles. Russ J Gen Chem 77, 2179–2185 (2007). https://doi.org/10.1134/S107036320712016X
Received:
Issue Date:
DOI: https://doi.org/10.1134/S107036320712016X