Abstract
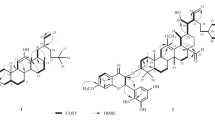
To investigate the chemical constituents of fruits of wild species of Crataegus scabrifolia and their lipid-lowering activities. The compounds were isolated and purified by various column chromatography, and their structures were identified based on the comprehensive analyses of the physicochemical properties and spectroscopic data. The lipid-lowering activities of isolated compounds were evaluated by a cellular model of HepG2 in vitro. Ten triterpenoids were obtained from the 95% ethanol extract of the fruits of wild species of C. scabrifolia and identified as α-amyrin (I), ursolic acid (II), pomolic acid (III), tormentic acid (IV), euscaphic acid (V), 2α,19α-dihydroxy-3-oxo-urs-12-en-28-oic acid (VI), fupenzic acid (VII), oleanolic acid (VIII), β-amyrin (IX), and 3-epifriedelinol (X). Compounds (I), (IX), and (X) were isolated from the genus Crataegus for the first time, and compounds (III−VIII) were isolated from the C. scabrifolia for the first time. Compounds (V‒VII) showed significant lipid-lowering activities in vitro with lipid-lowering rates of 8.9 ± 4.3%, 12.0 ± 4.6%, and 6.5 ± 5.5% at a concentration of 200 μmol/L. Molecular docking results suggested that the ACC may be a target of lipid-lowering activity for Crataegus triterpenoids.



Similar content being viewed by others
REFERENCES
Chinese Pharmacopeia Commission, Chinese Pharmacopoeia, vol. 1, Beijing: China Medical Science Press, 2020.
Dong, J., Chen, J., Gong, S., Xu, J., Xu, X., and Zhang, T., Chin. Tradit. Herb. Drugs, 2021, vol. 52, pp. 2801–2818.
Wu, J., Peng, W., Qin, R., and Zhou, H., Molecules, 2014, vol. 19, pp. 1685–1712. https://doi.org/10.3390/molecules19021685
Kumar, D., Arya, V., Bhat, Z.A., Khan, N.A., and Prasad, D.N., Rev. Bras. Farmacogn., 2012, vol. 22, pp. 1187–1200. https://doi.org/10.1590/S0102-695X2012005000094
Vera-Sánchez, K.S., Parra-Quijano, M., Nieto-Ángel, R., and Barrientos-Priego, A.F., Plants, 2021, vol. 10, p. 2561. https://doi.org/10.3390/plants10122561
Kunming Institute of Botany and Chinese Academy of Sciences, Flora Yunnanica, vol. 12, Beijing: Science Press, 2006.
Zhu, Q.-J., Lang, L.-J., Jiang, B., Shen, Y., Wang, Y., and Xiao, C.-J., J. Dali Univ., 2021, vol. 6, pp. 7–9.
Si, J.-Y., Chen, D.-H., and Gau, G.-Y., China J. Chin. Mater. Med., 1998, vol. 23, pp. 422–423.
Si, J.-Y., Gau, G.-Y., and Chen, D.-H., Nat. Prod. Res. Dev., 1994, vol. 6, pp. 49–51.
Kardar, M.N., Zhang, T., Coxon, G.D., Watson, D.G., Fearnley, J., and Seidel, V., Phytochemistry, 2014, vol. 106, pp. 156–163. https://doi.org/10.1016/j.phytochem.2014.07.016
Lee, Y.-G., Kang, K.W., Hong, W., Kim, Y.H., Oh, J.T., Park, D.W., Ko, M., Bai, Y.-F., Seo, Y.-J., Lee, S.-M., Kim, H., and Kang, S.C., Bioorg. Med. Chem., 2021, vol. 45, p. 116329. https://doi.org/10.1016/j.bmc.2021.116329
Isobe, T., Noda, Y., Ohsaki, A., Sakanaka, S., Kim, M., and Taniguchi, M., Yakugaku Zasshi, 1989, vol. 109, pp. 175–178. https://doi.org/10.1248/yakushi1947.109.3_175
Gai, C., Kong, D., and Wang, S., Chin. J. Pharm., 2010, vol. 41, pp. 580–582.
Xu, H.-X., Zeng, F.-Q., Wan, M., and Sim, K.-Y., J. Nat. Prod., 1996, vol. 59, pp. 643–645. https://doi.org/10.1021/np960165e
Hattori, M., Kuo, K.-P., Shu, Y.-Z., Tezuka, Y., Kikuchi, T., and Namba, T., Phytochemistry, 1988, vol. 27, pp. 3975–3976. https://doi.org/10.1016/0031-9422(88)83061-9
Acebey-Castellon, I.L., Voutquenne-Nazabadioko, L., Doan Thi Mai, H., Roseau, N., Bouthagane, N., Muhammad, D., Le Magrex Debar, E., Gangloff, S.C., Litaudon, M., Sevenet, T., Hung, N.V., and Lavaud, C., J. Nat. Prod., 2011, vol. 74, pp. 163–168. https://doi.org/10.1021/np100502y
Yu, B.-L., Liu, S.-B., Huang, N.-L., Yang, L., Yang, M.-C., Fan, H.-F., and Dai, H.-F., Chin. J. Exp. Tradit. Med. Form., 2017, vol. 23, pp. 96–100.
Shen, Y., Chen, H., Lang, L.-J., Dong, X., Xiao, C.-J., and Jiang, B., Phytochem. Lett., 2021, vol. 46, pp. 172–175. https://doi.org/10.1016/j.phytol.2021.10.017
Pu, S., Liu, Y., Liang, S., Liu, P., Qian, H., Wu, Q., and Wang, Y., Molecules, 2020, vol. 25, pp. 1392. https://doi.org/10.3390/molecules25061392
Luo, H.-Q., Shen, J., Chen, C.-P., Ma, X., Lin, C., Ouyang, Q., Xuan, C.-X., Liu, J., Sun, H.-B., and Liu, J., Chin. J. Nat. Med., 2018, vol. 16, pp 339–346. https://doi.org/10.1016/S1875-5364(18)30065-7
Nogueira, A.O., Oliveira, Y.I.S., Adjafre, B.L., De Moraes, M.E.A., and Aragão, G.F., Fund. Clin. Pharmacol., 2019, vol. 33, pp. 4–12. https://doi.org/10.1111/fcp.12402
Lang, L.-J., Wang, M., Lei, C., Shen, Y., Zhu, Q.-J., Diao, H.-M., Chen, H., Shen, L., Dong, X., Jiang, B., and Xiao, C.-J., Planta Med., 2022. https://doi.org/10.1055/a-1716-0958
Funding
The work was financially supported by the Special Basic Cooperative Research Programs of Yunnan Provincial Undergraduate Universities’ Association (grant no. 2019FH001-050), the Yunnan Key Laboratory of Screening and Research on Anti-pathogenic Plant Resources from Western Yunnan (grant no. 202105AG070003), the Yunnan Fundamental Research Projects (grant no. 202201AT070005), and the Scientific Research Initiation Fund of Dali University for Doctor (grant no. KYBS201734).
Author information
Authors and Affiliations
Contributions
Chao-Jiang Xiao and Bei Jiang conceptualized the work. Material preparation, extraction and isolation were performed by Qi-Jie Zhu, Ying Wang, and De-Quan Zhang. Biological activity assay was performed by Li-Juan Lang. Molecular docking and the draft of the manuscript were done by Chao-Jiang Xiao.
Corresponding authors
Ethics declarations
COMPLIANCE WITH ETHICAL STANDARDS
This article does not contain any studies involving animals or human participants performed by any of the authors.
Conflict of Interests
The authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Qi-Jie Zhu, Lang, LJ., Wang, Y. et al. Triterpenoids from the Fruits of Wild Species of Crataegus scabrifolia and Their Lipid-Lowering Activities. Russ J Bioorg Chem 48, 1291–1298 (2022). https://doi.org/10.1134/S1068162022060292
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1068162022060292