Abstract
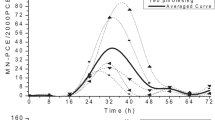
We designed in vitro study to determine possible genotoxic effects of tacrolimus (FK-506), which is used as a potent immunosuppressive drug, by using sister chromatid exchange (SCEs), chromosome aberration (CAs), micronuclei tests (MN) and cell growth kinetics such as mitotic index (MI) and replication index (RI) in human lymphocytes. The ceils were treated with 5, 25, 50, and 100 ng/ml concentrations of tacrolimus, for 24 and 48 h treatment periods. Tacrolimus induced CA and MN frequency at all concentrations for 24 and 48 h. In additon, it induced the SCE at the highest concentration for 24 h and at 25 and 100 ng/ml for 48 h. Tacrolimus decreased MI at all concentrations (except 5 ng/ml) for all treatment periods. It also inhibited the RI at 50 and 100 ng/ml concentrations for 24 h and at all concentrations for 48 h. Treatments given with tacrolimus result in the enhance of the different endpoints of genotoxicity, suggesting its mutagenic action on lymphocytes in vitro.
Similar content being viewed by others
References
Hiraoka, A., Ohashi, Y., Okamoto, S., et al., Japanese FK506 BMT (Bone Marrow Transplantation) Study Group: Phase III Study Comparing Tacrolimus (FK506) with Cyclosporine for Graft-versus-Host Disease Prophylaxis after Allogeneic Bone Marrow Transplantation, Bone Marrow Transplant., 2001, vol. 28, no. 2, pp.181–185.
Niwa, Y., Terashima, T., and Sumi, H., Topical Application of the Immunosuppressant Tacrolimus Accelerates Carcinogenesis in Mouse Skin, Br. J. Dermatol., 2003, vol. 149, no. 5, pp. 960–967.
Choi, S.J., You, H.S., and Chung, S.Y., Tacrolimus-Induced Apoptotic Signal Transduction Pathway, Transplant. Proc., 2008, vol. 40, no. 8, pp. 2734–2736.
Sommerer, C., Giese, T., Meuer, S., and Zeier, M., New Concepts to Individualize Calcineurin Inhibitor Therapy in Renal Allograft Recipients, Saudi J. Kidney Dis. Transplant., 2010, vol. 21, no. 6, pp. 1030–1037.
Li, H., Rao, A., and Hogan, P.G., Interaction of Calcineurin with Substrates and Targeting Proteins, Trends Cell Biol., 2011, vol. 21, no. 2, pp. 91–103.
Suzuki, T., Ikezumi, Y., Okubo, S., et al., Epstein-Barr Virus DNA Load and Seroconversion in Pediatric Renal Transplantation with Tacrolimus Immunosuppression, Pediatr. Transplant., 2007, vol. 11, no. 7, pp. 749–754.
Rauch, M.C., San Martín, A., Ojeda, D., et al., Tacrolimus Causes a Blockage of Protein Secretion which Reinforces Its Immunosuppressive Activity and also Explains Some of Its Toxic Side-Effects, Transplant. Immunol., 2009, vol. 22, nos. 1–2, pp. 72–81.
Penn, I., Cancers in Renal Transplant Recipients, Adv. Ren. Replace Ther., 2003, vol. 7, no. 2, pp. 147–156.
Anaya, F., Plaza, J., Sanz-Guajardo, D., et al., Cancer after Renal Transplantation, Transplant. Proc., 2003, vol. 35, no. 2, pp. 697–699.
Hui, R.L., Lide, W., Chan, J., et al., Association between Exposure to Topical Tacrolimus or Pimecrolimus and Cancers, Ann. Pharmacother., 2009, vol. 43, no. 12, pp. 1956–1963.
Bugelski, P.J., Volk, A., Walker, M.R., et al., Critical Review of Preclinical Approaches to Evaluate the Potential of Immunosuppressive Drugs to Influence Human Neoplasia, Int. J. Toxicol., 2010, vol. 29, no. 5, pp. 435–466.
Veness, M.J., Quinn, D.I., Ong, C.S., et al., Aggressive Cutaneous Malignancies Following Cardiothoracic Transplantation: The Australian Experience, Cancer, 1999, vol. 85, pp. 1758–1764.
Mistríkoví, J., Mrmusova’, M., Durmanova’, V., and Rajca’ni, J., Increased Neoplasm Development Due to Immunosuppressive Treatment with FK-506 in BALB/C Mice Persistently Infected with the Mouse Herpes Virus (MHV-72), Viral Immunol., 1999, vol. 12, no 3, pp. 237–247.
Penn, I., Lymphomas Complicating Organ Transplantation, Transplant. Proc., 1983, vol. 15, no. 4, suppl. 1.
Yarosh, D.B., Pena, A.V., et al., Calcineurin Inhibitors Decrease DNA Repair and Apoptosis in Human Keratinocytes Following Ultraviolet B Irradiation, J. Invest. Dermatol., 2005, vol. 125, no. 5, pp. 1020–1025.
Duncan, F.J., Wulff, B.C., Tober, K.L., et al., Clinically Relevant Immunosuppressants Influence UVB-Induced Tumor Size through Effects on Inflammation and Angiogenesis, Am. J. Transplant., 2007, vol. 7, no. 12, pp. 2693–2703.
Bonassi, S., Hagmar, L., Strömberg, U., et al., Chromosomal Aberrations in Lymphocytes Predict Human Cancer Independently of Exposure to Carcinogens: European Study Group on Cytogenetic Biomarkers and Health, Cancer Res., 2000, vol. 60, no. 6, pp. 1619–1625.
Yu, Y., Yuzawa, K., Otsuka, M., and Fukao, K., Mutagenicity of the New Immunosuppressive Drugs, FK 506, and Spergualin, Transplant. Proc., 1993, vol. 25, no. 2, pp. 2116–2118.
Özturk, Š., Ayna, T.K., Çefle, K., Palanduz, Ş., et al., Effect of Cyclosporin A and Tacrolimus on Sister Chromatid Exchange Frequency in Renal Transplant Patients, Genet. Test., 2008, vol. 12, no. 3, pp. 427–430.
Speit, G. and Haupter, S., On the Mechanism of Differential Giemsa Staining of Bromodeoxyuridine-Substituted Chromosomes, 2. Differences between the Demonstration of Sister Chromatid Differentiation and Replication Patterns, Hum. Genet., 1985, vol. 70, no. 2, pp. 126–129.
Shaffer, L.G., Slovak, M.L., and Campbell, L.J., An International System for Human Cytogenetic Nomenclature (ISCN), Basel: Karger, 2009.
Albertini, R.J., Anderson, D., Douglas, G.R., et al., IPCS Guidelines for the Monitoring of Genotoxic Effects of Carcinogens in Humans, Mutat. Res., 2000, vol. 463, pp. 111–117.
Fenech, M., Cytokinesis-Block Micronucleus Cytome Assay, Nat. Protoc., 2000, vol. 2, pp. 1084–1104.
Surralle’s, J. and Natarajan, A.T., Human Lymphocytes Micronucleus Assay in Europe: An International Survey, Mutat. Res., 1997, vol. 392, nos. 1–2, pp. 165–174.
Armstrong, V.W. and Oellerich, M., New Developments in the Immunosuppressive Drug Monitoring of Cyclosporine, Tacrolimus, and Azathioprine, Clin. Biochem., 2001, vol. 34, no. 1, pp. 9–16.
Gutierrez-Dalmau, A. and Campistol, J.M., Immunosuppressive Therapy and Malignancy in Organ Transplant Recipients: A Systematic Review, Drugs, 2007, vol. 67, pp. 1167–1198.
Dworkin, A.M., Tober, K.L., Duncan, J., et al., Chromosomal Aberrations in UVB-Induced Tumors of Immunosuppressed Mice, Genes, Chromosomes Cancer., 2009, vol. 48, pp. 490–501.
Palanduz, S., Sever, M.S., Oztürk, S., et al., Genotoxic Potential of Cyclosporin A in Patients with Renal Transplantation, Cell Biol. Toxicol., 1999, vol. 15, no. 1, pp. 13–17.
Yuzawa, K., Fukao, K., Iwasaki, Y., and Hamaguchi, H., Mutagenicity of Cyclosporine against Human Cells, Transplant. Proc., 1987, vol. 19, no. 1, part 2, pp. 1218–1220.
Japanese FK 506 Study Group: Japanese Study of FK 506 on Kidney Transplantation: The Benefit of Monitoring the Whole Blood FK 506 Concentration, Transplant. Proc., 1991, vol. 23, no. 6, pp. 3085–3088.
Oliveira, V.D., Zankl, H., and Rath, T., Mutagenic and Cytotoxic Effects of Immunosuppressive Drugs on Human Lymphocyte Cultures, Exp. Clin. Transplant., 2004, vol. 2, no. 2, pp. 273–279.
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Kurtoglu, E.L., Yuksel, S. Genotoxic effects of tacrolimus on human lymphocyte cells. Russ J Genet 48, 651–655 (2012). https://doi.org/10.1134/S1022795412050134
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1022795412050134