Abstract
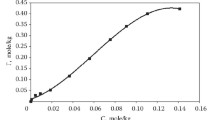
The sorption of aluminate from alkaline solutions on D-403 anion exchanger is studied. The sorption isotherm is described by the Langmuir and Freindlich classical equations and the Redlich-Peterson generalized equation. Thermodynamic parameters of sorption are determined using the Langmuir equation, modified to describe ion-exchange eqiulibria. A method for determining the type of the sorbed ion in the solid phases is proposed.
Similar content being viewed by others
References
H. M. F. Freundlich, Z. Phys. Chem. (Frankfurt) 57A, 385 (1906).
I. Langmuir, J. Am. Chem. Soc. 38, 2221 (1916).
St. Brunauer, P. H. Emmett, and E. Teller, J. Am. Chem. Soc. 60, 309 (1938).
O. Redlich and D. L. Peterson, J. Phys. Chem. 63, 1024 (1959).
R. Sips, J. Chem. Phys. 16, 490 (1948).
J. U. K. Oubagaranadin, Z. V. P. Murthy, and P. S. Rao, Int. Chem. Eng. 49, 196 (2007).
D. E. Chirkst, O. V. Cheremisina, and A. A. Chistyakov, Zh. Fiz. Khim. 82, 2382 (2008) [Russ. J. Phys. Chem. A 82, 2147 (2008)].
V. L. Raizman et al., Kompleks. Ispol’z. Mineral. Syr’ya, No. 3, 61 (1986).
N. I. Eremin, Yu. A. Volokhov, and V. E. Mironov, Usp. Khim. 43, 224 (1974).
G. M. Vol’dman and A. N. Zelikman, Theory of Hydrometallurgical Processes, the Manual for Higher School (Moscow, Intermet Inzhiniring, 2003).
Short Handbook of Physical Chemical Values, Ed. by A. A. Ravdel’ and A. M. Ponomareva (Ivan Fedorov, St. Petersburg, 2003) [in Russian].
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.A. Chistyakov, D.E. Chirkst, O.V. Cheremisina, 2011, published in Zhurnal Fizicheskoi Khimii, 2011, Vol. 85, No. 11, pp. 2137–2141.
Rights and permissions
About this article
Cite this article
Chistyakov, A.A., Chirkst, D.E. & Cheremisina, O.V. Sorption of aluminate from alkaline solutions on D-403 anion exchanger. Russ. J. Phys. Chem. 85, 1995–1999 (2011). https://doi.org/10.1134/S0036024411110069
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036024411110069