Abstract
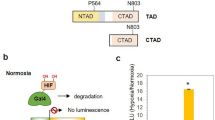
Uncontrolled growth in the cell mass of malignant tumors induces intensive angiogenesis. However, the demands of the cancer cells for nutrients and oxygen remain only partially met. Hypoxia is a process that accompanies malignant transformation and evokes changes in the DNA methylation profile in solid tumors. To a certain extent, these changes, including the hypermethylation of tumor suppressor gene promoters, are related to the decrease in the activity of Tet proteins under the conditions of oxygen and free radical deficit. Stabilization, accumulation, and nuclear translocation of the transcription factor HIF1α are the key molecular events in hypoxia. We modified the clear-cell renal cancer cell line Caki1 to stabilize the HIF1α protein and characterized a model cell line that will enable the studies of the mechanisms of changes of the DNA methylation level at a constant activity of Tet proteins and a gene transcription profile characteristic of hypoxia. The CRISPR/Cas9 DNA editing system was used to edit the VHL gene. The mutant VHL protein contained a disrupted alpha-helix at the C-terminus and could not participate in the molecular pathway of proteasomal degradation of the HIF1α factor; therefore, the latter accumulated in the nucleus and activated the specific target genes. An analysis of gene transcription revealed the induction of hypoxia-associated genes in the modified cell line. The developed Сaki-1/VHLmut model can be used to discriminate between the effects evoked by oxygen-suppressed hydroxylases of the Tet family and other hypoxia-associated mechanisms of DNA methylation/demethylation.
Similar content being viewed by others
Abbreviations
- VHL:
-
von Hippel–Lindau factor
- HIF1:
-
Hypoxiainducible factor 1
- Tet:
-
Ten-eleven translocation methylcytosine dioxygenase
- CRISPR:
-
Clustered Regularly Interspaced Short Palindromic Repeats
- CRISPR/Cas9:
-
a technique for genome editing in higher eukaryotes based on a bacterial system of adaptive immunity
References
Esteller M. 2008. Epigenetics in cancer. N. Engl. J. Med. 358, 1148–1159.
Thienpont B., Steinbacher J., Zhao H., et al. 2016. Tumour hypoxia causes DNA hypermethylation by reducing TET activity. Nature. 537, 63–68.
Kopera E., Schwerdtle T., Hartwig A., Bal W. 2004. Co(II) and Cd(II) substitute for Zn(II) in the zinc finger derived from the DNA repair protein XPA, demon strating a variety of potential mechanisms of toxicity. Chem. Res. Toxicol. 17, 1452–1458.
Zhigalova N., Artemov A., Mazur A., Prokhortchouk E. 2015. Transcriptome sequencing revealed differences in the response of renal cancer cells to hypoxia and CoCl2 treatment. F1000Res. 4, 1518.
Tanimoto K., Makino Y., Pereira T., Poellinger L. 2000. Mechanism of regulation of the hypoxia-inducible factor-1 alpha by the von Hippel–Lindau tumor suppressor protein. EMBO J. 19, 4298–4309.
Krieg M., Haas R., Brauch H., Acker T., Flamme I., Plate K.H. 2000. Up-regulation of hypoxia-inducible factors HIF-1alpha and HIF-2alpha under normoxic conditions in renal carcinoma cells by von Hippel–Lindau tumor suppressor gene loss of function. Oncogene. 19, 5435–5443.
Rechsteiner M.P., von Teichman A., Nowicka A., Sulser T., Schraml P., Moch H. 2011. VHL gene mutations and their effects on hypoxia inducible factor HIFalpha: Identification of potential driver and passenger mutations. Cancer Res. 71, 5500–5511.
Maher E.R., Kaelin W.G. 1997. von Hippel–Lindau disease. Medicine. 76, 381–391.
Ran F.A., Hsu P.D., Wright J., Agarwala V., Scott D.A., Zhang F. 2013. Genome engineering using the CRISPR–Cas9 system. Nat.Protoc. 8, 2281–2308.
Rathmell W.K., Hickey M.M., Bezman N.A., Chmielecki C.A., Carraway N.C., Simon M.C. 2004. In vitro and in vivo models analyzing von Hippel–Lindau disease-specific mutations. Cancer res. 64, 8595–8603.
Eustace A., Mani N., Span P.N., et al. 2013. A 26-gene hypoxia signature predicts benefit from hypoxia-modifying therapy in laryngeal cancer but not bladder cancer. Clin. Cancer Res. 19, 4879–4888.
Young R.M., Wang S.J., Gordan J.D., Ji X., Liebhaber S.A., Simon M.C. 2008. Hypoxia-mediated selective mRNA translation by an internal ribosome entry site-independent mechanism. J. Biol. Chem. 283, 16309–16319.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © N.A. Zhigalova, S.V. Zhenilo, A.V. Artemov, E.B. Prokhortchouk, 2017, published in Molekulyarnaya Biologiya, 2017, Vol. 51, No. 5, pp. 836–840.
Rights and permissions
About this article
Cite this article
Zhigalova, N.A., Zhenilo, S.V., Artemov, A.V. et al. CRISPR/Cas9-editing-based modeling of hypoxia in renal cancer cells. Mol Biol 51, 728–732 (2017). https://doi.org/10.1134/S0026893317050235
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0026893317050235