Abstract
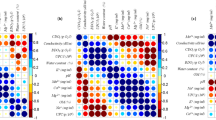
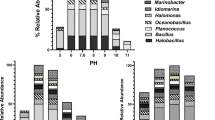
Investigations on the microbial life in several coastal solar salterns have revealed the presence of novel organisms and synthesis of unusual molecules active in extreme conditions which might be useful in different biotechnological industries. Biodiversity of heterotrophic aerobic bacteria isolated from two salterns, Pomorie salterns and Burgas salterns located at Burgas Bay, Black Sea coast, Bulgaria, as well as ability of the isolates to synthesize biotechnologically valuable compounds were investigated. The results revealed high taxonomic and metabolic bacterial diversity—we isolated 20 morphologically different moderately halophilic and two halotolerant strains affiliated with 11 species from eight genera referred to the phyla Proteobacteria, Firmicutes, and Actinobacteria. Gram-negative bacteria belonged to the genera Halomonas, Chromohalobacter, Salinivibrio, Cobetia, and Nesiotobacter, and gram-positive strains were representatives of the genera Virgibacillus, Salinicoccus, and Brevibacterium. All isolates were found to be alkalitolerant, and 41% of them were psychrotolerant. The strains degraded nine of the tested 18 substrates; polygalacturonase, catalase, phytase, and lipase producers were predominant. This is the first reported detection of xanthan lyase, gellan lyase, arabinase, and phytase activities in halophilic bacteria. Nine of the strains belonging to five different genera were found to produce exopolysaccharides (EPS). The highest level of EPS was observed in Chromohalobacter canadensis strain 28. More than a half of the strains displayed antimicrobial activity against one to five test bacteria and yeasts. The present study is the first report on halophilic bacteria isolated from salterns at the Black Sea coast indicating that the investigated area is an untapped resource of halophilic bacteria with biotechnological potential.
Similar content being viewed by others
References
Altschul, S.F., Gish, W., Miller, W., Myers, E.W., and Lipman, D.J., Basic local alignment search tool, J. Mol. Biol., 1990, vol. 215, no. 3, pp. 403–410.
Antón, J., Rosselo-Mora, R., Rodriguez-Valera, F., and Amann, R., Extremely halophilic bacteria in crystallizer ponds from solar salterns, Appl. Environ. Microbiol., 2000, vol. 66, no. 7, pp. 3052–3057.
Atanasova, N.S., Pietilä, M.K., and Oksanen, H.M., Diverse antimicrobial interactions of halophilic archaea and bacteria extend over geographical distances and cross the domain barrier, Microbiol. Open, 2013, vol. 2, no. 5, pp. 811–825.
Baati, H., Amdouni, R., Gharsallah, N., Sghir, A., and Ammar, E., Isolation and characterization of moderately halophilic bacteria from Tunisian solar saltern, Curr. Microbiol., 2010, vol. 60, no. 3, pp. 157–161.
Ballav, S., Kerkar, S., Thomas, S., and Augustine, N., Halophilic and halotolerant actinomycetes from a marine saltern of Goa, India producing antibacterial metabolites, J. Biosci. Bioeng., 2015, vol. 119, no 3, pp. 323–330.
Berrada, I., Willems, A., De Vos, P., El Fahime, E.M., Swings, J., Bendaou, N., Melloul, M., and Amar, M., Diversity of culturable moderately halophilic and halotolerant bacteria in a marsh and two salterns a protected ecosystem of Lower Loukkos (Morocco), Afr. J. Microbiol. Res., 2012, vol. 6, no. 10, pp. 2419–2434.
Biswas, J. and Paul, A.K., Production of extracellular enzymes by halophilic bacteria isolated from solar salterns, Int. J. Appl. Biol. Pharm., 2013, vol. 4, no. 4, pp. 30–36.
Biswas, J. and Paul, A.K., Production of extracellular polymeric substances by halophilic bacteria of solar salterns, Chinese J. Biol., 2014, article ID 205731, 12 pages.
Chen, L., Wang, G., Bu, T., Zhang, Y., Wang, Y., Liu, M., and Lin, X., Phylogenetic analysis and screening of antimicrobial and cytotoxic activities of moderately halophilic bacteria isolated from the Weihai Solar Saltern (China), World J. Microbiol. Biotechnol., 2010, vol. 26, no. 5, pp. 879–888.
Choi, D.H. and Cho, B.C., Citreimonas salinaria gen. nov., sp. nov., a member of the Roseobacter clade isolated from a solar saltern, Int. J. Syst. Evol. Microbiol., 2006, vol. 56, pp. 2799–2803.
Cosa, S., Mabinya, L.V., Olaniran, O.A., Okoh, O.O., Bernard, K., Deyzel, S., and Okoh, A.I., Bioflocculant production by Virgibacillus sp. Rob isolated from the bottom sediment of Algoa Bay in the Eastern Cape, South Africa, Molecules, 2011, vol. 16, no. 3, pp. 2431–2442.
De la Haba, R.R., Arahal, D.R., Márquez, M.C., and Ventosa, A., Phylogenetic relationships within the family Halomonadaceae based on comparative 23S and 16S rRNA gene sequence analysis, Int. J. Syst. Evol. Microbiol., 2010, vol. 60, no. 4, pp. 737–748.
De la Haba, R.R., Márquez, M.C., Papke, R.T., and Ventosa, A., Multilocus sequence analysis of the family Halomonadaceae, Int. J. Syst. Evol. Microbiol., 2012, vol. 62, no. 3, pp. 520–538.
Gupta, S., Sharma, P., Dev, K., and Sourirajan, A., Halophilic bacteria of Lunsu produce an array of industrially important enzymes with salt tolerant activity, Biochem. Res. Int., 2016, article ID 9237418, 10 pages.
Hidri, D.E., Guesmi, A., Najjari, A., Cherif, H., Ettoumi, B., Hamdi, C., Boudabous, A., and Cherif, A., Cultivationdependrnt assessment, diversity, and ecology of haloalkaliphilic bacteria in arid saline systems of southern Tunisia, Biomed. Res. Int., 2013, article 648141.
Jacob, H.J., Classification of halophilic heterotrophic bacteria thriving in the Jordanian Dead Sea littoral zone, J. Biol. Sci., 2012, vol. 12, no. 4, pp. 246–252.
Jakobsen, T.F., Kjeldsen, K.U., and Ingvorsen, K., Desulfohalobium utahense sp. nov., a moderately halophilic, sulfate-reducing bacterium isolated from Great Salt Lake, Int. J. Syst. Evol. Microbiol., 2006, vol. 56, pp. 2063–2069.
Jose, P.A. and Jebakumar, R.D.J., Phylogenetic appraisal of antagonistic, slow growing actinomycetes isolated from hypersaline inland solar salterns at Sambhar salt lake, India, Front. Microbiol., 2013, vol. 4, article 190.
Joshi, A.A., Kanekar, P.P., Kelkar, A.S., Shouche, Y.S., Vani, A.A., Borgave, S.B., and Sarnaik, S.S., Cultivable bacterial diversity of alkaline Lonar Lake, India, Microb. Ecol., 2008, vol. 55, no. 2, pp. 163–172.
Kambourova, M., Tomova, I., Boyadzhieva, I., Radchenkova, N., and Vasileva-Tonkova, E., Unusually high archaeal diversity in a crystallizer pond, Pomorie salterns, Bulgaria, revealed by phylogenetic analysis, Archaea, 2016a, article ID 7459679.
Kambourova, M., Tomova, I., Boyadzhieva, I., Radchenkova, N., and Vasileva-Tonkova, E., Phylogenetic analysis of the bacterial community in a crystallizer pond, Pomorie salterns, Bulgaria, Biotechnol. Biotechnol. Equip., 2017, vol.31, no. 2, pp. 325–332.
Kumar, A., Chanderman, A., Makolomakwa, M., Perumal, K., and Singh, S., Microbial production of phytases for combating environmental phosphate pollution and other diverse applications, Crit. Rev. Env. Sci. Tec., 2016, vol. 46, pp. 556–591.
Kumar, S., Karan, R., Kapoor, S., Singh, S.P., and Khare, S.K., Screening and isolation of halophilic bacteria producing industrially important enzymes, Braz. J. Microbiol., 2012, vol. 43, no. 4, pp. 1595–1603.
Labrenz, M., Collins, M.D., Lawson, P.A., Tindall, B.J., Schumann, P., and Hirsch, P. Roseovarius tolerans gen. nov., sp. nov., a budding bacterium with variable bacteriochlorophyll a production from hypersaline Ekho Lake, Int. J. Syst. Bacteriol., 1999, vol. 49, no. 1, pp. 137–147.
León, M.J., Fernández, A.B., Ghai, R., Sánchez-Porro, C., Rodriguez-Valera, F., and Ventosa, A., From metagenomics to pure culture: isolation and characterization of the moderately halophilic bacterium Spiribacter salinus gen. nov., sp. nov., Appl. Environ. Microbiol., 2014, vol. 80, no. 13, pp. 3850–3857.
Luque, R., Béjar, V., Quesada, E., and Llamas, I., Diversity of halophilic bacteria isolated from Rambla Salada, Murcia (Spain), Can. J. Microbiol., 2014, vol. 60, no. 12, pp. 839–846.
Maidak, B.L., Larsen, N., McCaughey, M.J., Overbeek, R., Olsen, G.J., Fogel, K., Blandy, J., and Woese, C.R., The Ribosomal Database Project, Nucleic Acids Res., 1994, vol. 22, no. 17, pp. 3485–3487.
Moreno, M.L., Pérez, D., García, M.T., and Mellado, E., Halophilic bacteria as a source of novel hydrolytic enzymes, Life, 2013, vol. 3, no. 1, pp. 38–51.
Ollivier, B., Hatchikian, C.E., Prensier, G., Guezennec, J., and Garcia, J.L., Desulfohalobium retbaense gen. nov., sp. nov., a halophilic sulphate reducing bacterium from sediments of a hypersaline lake in Senegal, Int. J. Syst. Bacteriol., 1991, vol. 41, no. 1, pp. 74–81.
Oren, A., The bioenergetic basis for the decrease in metabolic diversity at increasing salt concentrations: implications for the functioning of salt lake ecosystems, Hydrobiologia, 2001, vol. 466, nos. 1−3, pp. 61–72.
Oren, A., Diversity of halophilic microorganisms: environments, phylogeny, physiology, and applications, J. Ind. Microbiol. Biotechnol., 2002, vol. 28, no. 1, pp. 56–63.
Oren, A., Industrial and environmental applications of halophilic microorganisms, Environ. Technol., 2010, vol. 31, nos. 8−9, pp. 825–834.
Pasić, L., Ulrih, N.P., Crnigoj, M., Grabnar, M., and Velikonja, B.H., Haloarchaeal communities in the crystallizers of two adriatic solar salterns, Can. J. Microbiol., 2007, vol. 53, no. 1, pp. 8–18.
Poli, A., Kazak, H., Gürleyendağ, B., Tommonaro, G., Pieretti, G., Öner, E.T., and Nicolaus, B., High level synthesis of levan by a novel Halomonas species growing on defined media, Carbohydr. Polym., 2009, vol. 78, no. 4, pp. 651–657.
Poli, A., Moriello, V.S., Esposito, E., Lama, L., Gambacorta, A., and Nicolaus, B., Exopolysaccharide production by a new Halomonas strain CRSS isolated from saline lake Cape Russell in Antarctica growing on complex and defined media, Biotechnol. Lett., 2004, vol. 26, no. 21, pp. 1635–1638.
Poli, A., Nicolaus, B., Denizci, A.A., Yavuzturk, B., and Kazan, D., Halomonas smyrnensis sp. nov., a moderately halophilic, exopolysaccharide-producing bacterium, Int. J. Syst. Evol. Microbiol., 2013, vol. 63, no. 1, pp. 10–18.
Rohban, R., Amoozegar, M. A., and Ventosa, A., Screening and isolation of halophilic bacteria producing extracellular hydrolyses from Howz Soltan Lake, Iran, J. Ind. Microbiol. Biotechnol., 2009, vol. 36, no. 3, pp. 333–340.
Romano, I., Gambacorta, A., Lama, L., Nicolaus, B., and Giordano, A., Salinivibrio costicola subsp. alcaliphilus subsp. nov., a haloalkaliphilic aerobe from Campania Region (Italy), Syst. Appl. Microbiol., 2005, vol. 28, no. 7, pp. 34–42.
Rothschild, L.J. and Mancinelli, R.L., Life in extreme environments, Nature, 2001, vol. 409, no. 6823, pp. 1092–1101.
Sabet, S., Diallo, L., Hays, L., Jung, W., and Dillon, J.G., Characterization of halophiles isolated from solar salterns in Baja California, Mexico, Extremophiles, 2009, vol. 13, no. 4, pp. 643–656.
Sánchez-Porro, C.S., Martґın, S., Mellado, E., and Ventosa, A., Diversity of moderately halophilic bacteria producing extracellular hydrolytic enzymes, J. Appl. Microbiol., 2003, vol. 94, no. 2, pp. 295–300.
Sorokin, D.Yu., Tourova, T.P., Lysenko, A.M., and Muyzer, G., Diversity of culturable halophilic sulfur-oxidizing bacteria in hypersaline habitats, Microbiology (UK), 2006, vol. 152, no. 10, pp. 3013–3023.
Swofford, D.L., PAUP*: Phylogenetic Analysis Using Parsimony (* and Other Methods), Version 4, Sunderland, MA: Sinauer Associates, 2002.
Tambekar, D.H. and Dhundale, V.R., Screening of antimicrobial potentials of haloalkaliphilic bacteria isolated from Lonar Lake, Int. J. Pharm. Chem. Biol. Sci., 2013, vol. 3, no. 3, pp. 820–825.
Tamura, K., Dudley, J., Nei, M., and Kumar, S., MEGA4: molecular evolutionary genetics analysis (MEGA) software version 4.0, Mol. Biol. Evol., 2007, vol. 24, no. 8, pp. 1596− 1599.
Tomova, I., Gladka, G., Tashyrev, A., and Vasileva-Tonkova, E., Isolation, identification and hydrolytic enzymes production of aerobic heterotrophic bacteria from two Antarctic islands, Int. J. Environ. Sci., 2014, vol. 4, no. 5, pp. 614–625.
Tomova, I., Lazarkevich, I., Tomova, A., Kambourova,M., and Vasileva-Tonkova, E., Diversity and biosynthetic potential of culturable aerobic heterotrophic bacteria isolated from Magura Cave, Bulgaria, Int. J. Speleol., 2013, vol. 42, no. 1, pp. 65–76.
Venkatachalam, S., Gowdaman, V., and Prabagaran, S.R., Culturable and culture-independent bacterial diversity and the prevalence of cold-adapted enzymes from the Himalayan mountain ranges of India and Nepal, Microb. Ecol., 2015, vol. 69, no. 3, pp. 472–491.
Ventosa, A., De la Haba, R., Sanchez-Porro, C., and Thane Papke, R., Microbial diversity of hypersaline environments: a metagenomic approach. Curr. Opin. Microbiol., 2015, vol. 25, pp. 80–87.
Yeon, S.H., Jeong, W.J., and Park, J.S., The diversity of culturable organotrophic bacteria from local salterns, J. Microbiol., 2005, vol. 43, no. 1, pp. 1–10.
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Boyadzhieva, I., Tomova, I., Radchenkova, N. et al. Diversity of Heterotrophic Halophilic Bacteria Isolated from Coastal Solar Salterns, Bulgaria and Their Ability to Synthesize Bioactive Molecules with Biotechnological Impact. Microbiology 87, 519–528 (2018). https://doi.org/10.1134/S0026261718040033
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0026261718040033