Abstract
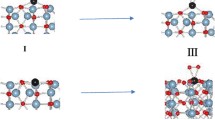
In situ IR spectroscopic studies show that a formate, an aldehyde-like complex, and bridging and linear methoxy groups exist on the alumina surface involved in methanol conversion. In the absence of methanol in the gas phase, the interaction between two bridging methoxy groups yields dimethyl ether in the gas phase. When methanol is present in the gas phase, it interacts with methoxy groups on the surface. This reaction makes the main contribution to the formation of dimethyl ether. The linear methoxy group undergoes conversion via several routes. The main route is desorption with methanol formation in the gas phase, and no more than 10% of the linear methoxy groups are converted into formate and aldehyde, which are CO2 sources in the gas phase. In the absence of methanol in the gas phase, the conversion rate of the methoxy groups is independent of the presence of water and oxygen. A scheme of the surface reactions is suggested to explain the conversion of the methoxy groups.
Similar content being viewed by others
References
Vishwanathan, M., Roh, H.-S., and Kim, J.-N., Catal. Lett., 2004, vol. 96, p. 23.
Tleimat-Manzalji, R., Bianchi, D., and Pajonk, G.M., React. Kinet. Catal. Lett., 1993, vol. 51, p. 29.
Bianchi, D., Chafik, T., Khalfallah, M., and Teichner, S.J., Appl. Catal., A, 1995, vol. 123, p. 89.
Sambeth, J.E., Juan, A., and Gambaro, L., J. Mol. Catal. A: Chem., 1997, vol. 118, p. 283.
Sambeth, J.E., Centeno, M.A., and Paul, A., J. Mol. Catal. A: Chem., 2000, vol. 161, p. 89.
Lindström, B., Pettersson, L., and Menon, P.G., Appl. Catal., A, 2002, vol. 234, p. 111.
Zhang, X. and Shi, P., J. Mol. Catal. A: Chem., 2003, vol. 194, p. 99.
Iwasa, N., Mayanagi, T., and Ogawa, N., Catal. Lett., 1998, vol. 54, p. 119.
Kapoor, M.P., Raj, A., and Matsumura, Y., Microporous Mesoporous Mater, 2001, vol. 44, no. 5, p. 565.
Schiffino, R.S. and Merril, R.P., J. Phys. Chem., 1993, vol. 97, p. 6425.
Krylov, O.V. and Matyshak, V.A., Promezhutochnye soedineniya v geterogennom katalize (Intermediate Compounds in Heterogeneous Compounds), Moscow: Nauka, 1996.
Matyshak, V.A., Khomenko, T.I., Lin, G.I., Zavalishin, I.N., and Rozovskii, A.Ya., Kinet. Katal., 1999, vol. 40, no. 2, p. 295 [Kinet. Catal. (Engl. Transl.), vol. 40, no. 2, p. 269].
Neophytides, S.G., Marchi, A.J., and Froment, G.F., Appl. Catal., A, 1992, vol. 86, p. 45.
Martin, K.A. and Zabransky, R.F., Appl. Spectrosc., 1991, vol. 45, p. 68.
Davydov, A.A., IK-spektroskopiya v khimii poverkhnosti okislov (IR Spectroscopy Applied to the Chemistry of Oxide Surfaces), Novosibirsk: Nauka, 1984.
Daturi, M., Binet, C., Lavalley, J-C., Galtayries, A., and Spoken, R., Phys. Chem. Chem. Phys., 1999, vol. 1, p. 5717.
Sidkon, A. and Nix, R.M., J. Phys. Chem. B, 1999, vol. 103, p. 6984.
Novakova, J., Kubelkova, L., and Dolysek, Z., J. Catal., 1987, vol. 108, p. 208.
Lamotte, J., Moravek, V., Bensitel, M., and Lavalley, J.C., React. Kinet. Catal. Lett., 1988, vol. 36, p. 113.
Knop-Gericke, A., Havecker, M., Schedel-Niedrig, Th., and Schlogl, R., Top. Catal., 2000, vol. 10, p. 187.
Carley, A.P., Owens, A.W., Rajumon, M.K., Roberts, M.W., and Jackson, S.D., Catal. Lett., 1996, vol. 37, p. 79.
Ueno, A., Onishi, T., and Tamaru, K., Trans. Faraday Soc., 1971, vol. 67, p. 3585.
Matyshak, V.A., Khomenko, T.I., Bondareva, N.K., Panchishnyi, V.I., and Korchak, V.N., Kinet. Katal., 1998, vol. 39, no. 1, p. 100 [Kinet. Catal. (Engl. Transl.), vol. 39, no. 1, p. 93].
Matyshak, V.A. and Krylov, O.V., Catal. Today, 1996, vol. 25, p. 1.
Millar, G.J., Rochester, C.H., and Waugh, K.C., J. Chem. Soc., Faraday Trans., 1991, vol. 87, no. 17, p. 2795.
Millar, G.J., Rochester, C.H., and Waugh, K.C., J. Chem. Soc., Faraday Trans., 1991, vol. 87, no. 17, p. 2785.
Burcham, L.J. and Wachs, I.E., Catal. Today, 1999, vol. 49, p. 467.
Busca, G., Rossi, P.F., Lorenzelli, V., Benaissa, M., Travert, J., and Lavalley, J.-C., J. Phys. Chem., 1985, vol. 89, p. 5433.
Malakhova, I.V., Ermolaev, V.K., Danilova, I.G., Paukshtis, E.A., and Zolotarskii, I.A., Kinet. Katal., 2003, vol. 44, no. 4, p. 587 [Kinet. Catal. (Engl. Transl.), vol. 44, no. 4, p. 536].
Busca, G., Lamotte, J., Lavalley, J.-C., and Lorenzelli, V., J. Am. Chem. Soc., 1987, vol. 109, no. 11, p. 5197.
Clarke, D.B., Lee, D.-K., Sandoval, M.J., and Bell, A.T., J. Catal., 1994, vol. 150, p. 81.
Kharlanov, A.A., Cand. Sci. (Chem.) Dissertation, Moscow: Moscow State Univ., 1995.
Ortelli, E.E., Weigel, J.M., and Wokaun, A., Catal. Lett., 1998, vol. 54, p. 41.
Tsyganenko, A.A., Doctoral (Phys.-Math.) Dissertation, St. Petersburg: St. Petersburg State Univ., 2000.
Chen, B. and Falconer, J.L., J. Catal., 1993, vol. 144, p. 214.
Clayborne, P.A., Nelson, T.C., and DeVore, T.C., Appl. Catal., A, 2004, vol. 257, p. 225.
Matyshak, V.A. and Krylov, O.V., Kinet. Katal., 2002, vol. 43, no. 3, p. 422 [Kinet. Catal. (Engl. Transl.), vol. 43, no. 3, p. 391].
DeCanio, E.C., Nero, V.P., and Bruno, J.W., J. Catal., 1992, vol. 135, p. 444.
Matsushima, T. and White, J., J. Catal., 1976, vol. 44, p. 183.
Jain, J.R. and Pillai, C.N., J. Catal., 1967, vol. 9, p. 322.
Padmanabhan, V.R. and Eastburn, F.J., J. Catal., 1972, vol. 24, p. 88.
Knozinger, H., Kochloefl, K., and Meye, W., J. Catal., 1973, vol. 28, p. 69.
Ivanov, S.I. and Makhlin, V.A., Kinet. Katal., 1996, vol. 37, no. 6, p. 873 [Kinet. Catal. (Engl. Transl.), vol. 37, no. 6, p. 812].
Rozovskii, A.Ya., Kinet. Katal., 2003, vol. 44, no. 3, p. 391 [Kinet. Catal. (Engl. Transl.), vol. 44, no. 3, p. 360].
Busca, G., Catal. Today, 1996, vol. 27, p. 457.
Spinicci, R., Thermochim. Acta, 1997, vol. 296, p. 87.
Herd, A.C., Onishi, T., and Tamaru, K., Bull. Chem. Soc. Jpn., 1974, vol. 47, no. 3, p. 575.
Groff, R.P. and Marogue, W.H., J. Catal., 1984, vol. 87, p. 461.
Yamashita, K., Naito, S., and Tamaru, K., J. Catal., 1985, vol. 94, p. 535.
Amenomiya, Y., J. Catal., 1979, vol. 57, p. 64.
Solymosi, F., Kiss, J., and Kovacs, I., Surf. Sci., 1987, vol. 192, p. 47.
Dowden, D.A., J.Chem. Soc, 1950, nos. 1–2, p. 242.
Tsyganenko, A.A., Smirnov, K.S., Rzhevskiy, A.M., and Mardilovich, P.P., Mater. Chem. Phys., 1990, vol. 26, no. 1, p. 35.
Vishnetskaya, M.V., Takhtarova, G.N., and Topchieva, K.V., Kinet. Katal., 1985, vol. 26, no. 5, p. 1271.
Vishnetskaya, M.V., Takhtarova, G.N., and Topchieva, K.V., Kinet. Katal., 1985, vol. 26, no. 5, p. 1272.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © V.A. Matyshak, L.A. Berezina, O.N. Sil’chenkova, V.F. Tret’yakov, G.I. Lin, A.Ya. Rozovskii, 2009, published in Kinetika i Kataliz, 2009, Vol. 50, No. 1, pp. 120–131.
Rights and permissions
About this article
Cite this article
Matyshak, V.A., Berezina, L.A., Sil’chenkova, O.N. et al. Properties of surface compounds in methanol conversion on γ-Al2O3: Data of in situ IR spectroscopy. Kinet Catal 50, 111–121 (2009). https://doi.org/10.1134/S0023158409010157
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0023158409010157