Abstract
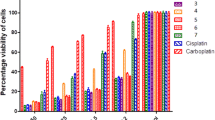
The discovery of the antitumor activity of cisplatin led several research groups to investigate the possible therapeutic applications of other metal based compounds. In an attempt to develop novel metal based drugs with a different therapeutic profile to cisplatin, we have synthesized a new N,N-chelated organotin(IV) trifluoroacetate by the reaction of Ph3SnOCOCF3 with equimolar amounts of 2,9-dimethyl-1,10-phenanthroline (Neocuproine). The complex is characterized by FT-IR and multinuclear NMR (1H, 13C, 19F and 119Sn). FT-IR results authenticate the ligand coordination to the organotin moiety via nitrogen atoms. Furthermore, the cytotoxic activity of the free ligand (Neocuproine) and triorganotin(IV) complex towards human cervix carcinoma HeLa, human myelogenous leukemia K562 and normal immunocompetent cells, peripheral blood mononuclear cells PBMC is evaluated by the MTT (3-[4,5-dimetylthiazol-2-yl]-2,5-diphenyltetrazolium bromide) method. The complex exhibits higher activities than antitumor drug cisplatin in all the tested cell lines. These results indicate that the studied triorganotin(IV) complex can be a potential anticancer agent for further stages of screening in vitro and/or in vivo.
Similar content being viewed by others
References
N. Muhammad, A. Shah, Z. Rehman, S. Shuja, S. Ali, R. Qureshi, A. Meetsma, and M. N. Tahir, J. Organomet. Chem., 694, 3431 (2009).
B. Rosenberg, L. Van Camp, J. E. Trosko, and V. H. Mansour, Nature, 222, 385 (1969).
M. Letizia Falcioni, M. Pellei, and R. Gabbianellic, Mutation Research/Genetic Toxicology and Environmental Mutagenesis, 653, 57 (2008).
N. Muhammad, Z. Rehman, S. Ali, A. Meetsma, and F. Shaheen, Inorg. Chim. Acta, 362, 2842 (2009).
P. Svec, Z. Padelkova, A. Ruzicka, T. Weidlich, L. Dusek, and L. Plasseraud, J. Organomet. Chem., 696, 676 (2011).
M. Kemmer, H. Dalil, M. Biesemans, J. C. Martins, B. Mahieu, E. Horn, D. de Vos, E. R. T. Tiekink, R. Willem, and M. J. Gielen, Organomet. Chem., 608, 63 (2000).
C. Deegan, M. McCann, M. Devereux, B. Coyle, and D. A. Egan, J. Cancer Lett., 247, 224 (2007).
N. Shahabadi, S. Kashanian, and M. Purfoulad, J. Spectrochim. Acta Part A, 72, 757 (2009).
T. Mosmann and J. Immunol, Methods, 65, 55 (1983).
M. Ohno, T. Abe, and J. Immunol, Methods, 145, 199 (1991).
X. Wu, W. Kang, D. Zhu, C. Zhu, and S. Liu, J. Organomet. Chem., 694, 2981 (2009).
W. Rehman, M. K. Baloch, and A. Badshah, Eur. J. Med. Chem., 43, 238 (2008).
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © 2014 M. Yousefi, M. Safari, M. B. Torbati, A. Amanzadeh.
The text was submitted by the authors in English. Zhurnal Strukturnoi Khimii, Vol. 55, No. 1, pp. 106–111, January–February, 2014.
Rights and permissions
About this article
Cite this article
Yousefi, M., Safari, M., Torbati, M.B. et al. In vitro anti-proliferative activity of novel hexacoordinated triphenyltin(IV) trifluoroacetate containing a bidentate N-donor ligand. J Struct Chem 55, 101–106 (2014). https://doi.org/10.1134/S0022476614010168
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0022476614010168