Abstract
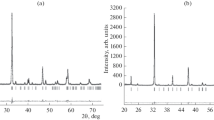
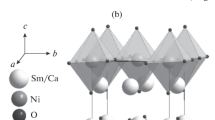
Complex oxides in the Sm2O3–CaO–MO and Ln2O3–SrO–MO systems (Ln = Sm, Gd; M = Fe, Co) were prepared via the glycerol-nitrate technique at 1100°С in air. The homogeneity ranges for the solid solutions were determined as follows: Sr1 – хGdхCoO3 – δ (0.1 ≤ х ≤ 0.4); Sr2 – yGdyCoO4 – δ (0.8 ≤ y ≤ 1.2); Sr1 – хGdхFeO3 – δ (0.05 ≤ х ≤ 0.30 and 0.80 ≤ х ≤ 1.0); Ca1 – xSmxFeO3 – δ (0.70 ≤ x ≤ 1.0); Sr2 – ySmyFeO4 – δ (0.7 ≤ y ≤ 0.8); Sr2 – yGdyFeO4 – δ (0.75 ≤ y ≤ 0.8); Ca2 – уSmуFeO4 – δ (у = 0.90); Sr3 – zSmzFe2O7 – δ (0 ≤ z ≤ 0.3 and z = 1.8); and Sr3 – zGdzFe2O7 – δ (0 ≤ z ≤ 0.3 and z = 1.9). For all single phase oxides, the crystal structure was determined and the unit cell parameters, the unit cell volume, and the structural parameters were refined.







Similar content being viewed by others
REFERENCES
Tu, H.Y., Takeda, Y., Imanishi, N., and Yamamoto, O., Ln1 – xSrxCoO3 (Ln = Sm, Dy) for the electrode of solid oxide fuel cells, Solid Stare Ionics, 1997, vol. 100, pp. 283–288.
Park, I., Im, J., Choi, J., Ahn, J., and Shin, D., Enhanced performance of the Sm0.5Sr0.5CoO3 – δ double layer porous cathode for solid oxide fuel cells, Solid State Ionics, 2011, vol. 184, pp. 35–38.
Dong, F., Chen, D., Ran, R., Park, H., Kwak, C., and Shao, Z., A comparative study of Sm0.5Sr0.5MO3 – δ (M = Co and Mn) as oxygen reduction electrodes for solid oxide fuel cells, Int. J. Hydrogen Energy, 2012, vol. 37, no. 4, pp. 4377–4387.
Fukunaga, H., Koyama, M., Takahashi, N., Wen, C., and Yamada, K., Reaction model of dense Sm0.5Sr0.5CoO3 as SOFC cathode, Solid State Ionics, 2000, vol. 132, nos. 3–4, pp. 279–285.
Tsipis, E.V. and Kharton, V.V., Electrode materials and reaction mechanisms in solid oxide fuel cells: a brief review. III. Recent trends and selected methodological aspects, J. Solid State Electrochem., 2011, vol. 15, no. 5, pp. 1007–1040.
Petric, A., Huang, P., and Tietz, F., Evaluation of La–Sr–Co–Fe–O perovskites for solid oxide fuel cells and gas separation membranes, Solid State Ionics, 2000, vol. 135, nos. 1–4, pp. 719–725.
Michel, C.R., Delgado, E., Santillan, G., Martínez, A.H., and Chavez-Chavez, A., An alternative gas sensor material: synthesis and electrical characterization of SmCoO3, Mater. Res. Bull., 2007, vol. 42, pp. 84–93.
Kovalevsky, A.V., Kharton, V.V., Tikhonovich, V.N., Naumovich, E.N., Tonoyan, A.A., Reut, O.P., and Boginsky, L.S., Oxygen permeation through Sr(Ln)CoO3 – δ (Ln = La, Nd, Sm, Gd) ceramic membranes, Mater. Sci. Eng., B., 1998, vol. 52, nos. 2–3, pp. 105–116.
Nitadori, T., Ichiki, T., and Misono, M., Catalytic properties of perovskite-type mixed oxides (ABO3) consisting of rare earth and 3d transition metals. The roles of the A- and B-site ions, Bull. Chem. Soc. Jpn., 1988, vol. 61, pp. 621–626.
Arakawa, T., Yoshida, A., and Shiokawa, J., Catalytic properties and activity of rare-earth orthoferrites in oxidation of methanol, Mater. Res. Bull., 1980, vol. 15, no. 2, pp. 347–352.
Skinner, S.J., Characterisation of La2NiO4 + δ using in-situ high temperature neutron powder diffraction, Solid State Sci., 2003, vol. 5, pp. 419–426.
Galayda, A.P., Volkova, N.E., Gavrilova, L.Ya., Balymov, K.G., and Cherepanov, V.A., Phase equilibria, structure and properties of intermediate phases in the Sm2O3–Fe2O3–CoO and Sm2O3–CaO–CoO systems, J. Alloys Compd., 2017, vol. 718, pp. 288–297.
Volkova, N.E., Maklakova, A.V., Gavrilova, L.Ya., and Cherepanov, V.A., Phase equilibria, crystal structure, and properties of intermediate oxides in the Sm2O3–SrO–CoO system, Eur. J. Inorg. Chem., 2017, pp. 3285–3292.
Shannon, R.D., Revised effective ionic radii and systematic studies of interatomic distances in halides and chalcogenides, Acta. Crystallogr., Sect. A: Cryst. Phys., Diffr., Theor. Gen. Crystallogr., 1976, vol. 32, no. 5, pp. 751–767.
James, M., Avdeev, M., Barnes, P., Morales, L., Wallwork, K., and Withers, R., Orthorhombic superstructures within the rare earth strontium-doped cobaltate perovskites: Ln1 – xSrxCoO3 – δ (Ln = Y3+, Dy3+–Yb3+; 0.750 ≤ x ≤ 0.875), J. Solid State Chem., 2007, vol. 180, pp. 2233–2247.
Istomin, S.Y., Drozhzhin, O.A., and Svensson, G., Synthesis and characterization of Sr1 – xLnxCoO3 – δ, Ln = Y, Sm–Tm, 0.1 ≤ x ≤ 0.5, J. Solid State Sci., 2004, vol. 6, pp. 539–546.
Volkova, N.E., Khvostova, L.V., Gavrilova, L.Ya., and Cherepanov, V.A., Role of Sm content to the crystal structure and properties of Sr1 – xSmxFeO3 – δ, J. Solid State Chem., 2018, vol. 267, pp. 113–118.
Khvostova, L.V., Volkova, N.E., Gavrilova, L.Ya., and Cherepanov, V.A., Crystal structure, oxygen nonstoichiometry and properties of novel Ruddlesden–Popper phase Sm1.8Sr1.2Fe2O7 – δ, Mater. Lett., 2018, vol. 213, pp. 158–161.
Gurusinghe, N., de la Figuera, J., Marco, J., Thomas, M.F., Berry, F.J., and Greaves, C., Synthesis and characterisation of the n = 2 Ruddlesden–Popper phases Ln2Sr(Ba)Fe2O7 (Ln = La, Nd, Eu), Mater. Res. Bull., 2013, vol. 48, pp. 3537–3544.
Funding
This work was supported by the Russian Federation Ministry of Science and Higher Education (state research target, project no. 4.2288.2017/PCh).
Author information
Authors and Affiliations
Corresponding author
Additional information
The work was presented at 16th International IUPAC Conference on High Temperature Materials Chemistry (HTMC-XVI), July 2–6, 2018, Yekaterinburg, Russia.
Rights and permissions
About this article
Cite this article
Khvostova, L.V., Galayda, A.P., Maklakova, A.V. et al. Crystal Structure of Solid Solutions in the Sm 2 O 3 –CaO–MO and Ln 2 O 3 –SrO–MO Systems (Ln = Sm, Gd; M = Fe, Co) . Inorg Mater 55, 1001–1006 (2019). https://doi.org/10.1134/S0020168519080041
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0020168519080041