Abstract
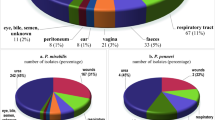
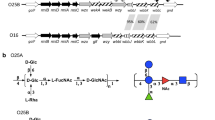
The aim of this work was to serotype Proteus mirabilis urinary tract infection (UTI) strains based on chemically defined O-antigens with the use of two clinical collections from Sweden and Poland consisting of 99 and 24 UTI strains, respectively. A simple two-step serotyping scheme was proposed using enzyme immunoassay with heat-stable surface antigens of Proteus cells and immunoblotting with isolated lipopolysaccharides (LPSs). Using polyclonal anti-P. mirabilis rabbit antisera, 50 Swedish and 8 Polish strains were classified into serogroups O10, O38, O36, O30, O17, O23, O9, O40, O49, O27, O5, O13, O24, O14, and O33. From the Swedish strains, 10 belonged to serogroup O10 and five to each of serogroups O38, O36, and O9. Therefore, none of the O-serogroups was predominant. The majority of the serotyped clinical strains possess acidic O-antigens containing uronic acids and various acidic non-carbohydrate substituents. In immunoblotting, antisera cross-reacted with both O-antigen and core of LPSs. The core region of 19 LPSs bound a single serum, and that of 12 LPSs bound more than two sera. Following bioinformatic analysis of the available sequences, a molecular approach to the prediction of Proteus core oligosaccharide structures was proposed. The identification of the core type of P. mirabilis R110, derived from a serogroup O3 wild strain, using restriction fragments length polymorphism analysis of galacturonic acid transferase is shown as an example. In summary, the most frequent O-serogroups among P. mirabilis UTI stains were identified. The diversity of serological reactions of LPSs is useful for serotyping of P. mirabilis clinical isolates. A possible role of the acidic components of O-antigens in UTI is discussed.
Similar content being viewed by others
Abbreviations
- EIA:
-
enzyme immunoassay
- LPS:
-
lipopolysaccharide
- OPS:
-
O-polysaccharide
- RFLP:
-
restriction fragment length polymorphism
- UTI:
-
urinary tract infections
References
Wilson, C., Thakore, A., Isenberg, D., and Ebringer, A. (1997) Rheumatol. Int., 16, 187–189.
Rozalski, A., Sidorczyk, Z., and Kotelko, K. (1997) Microbiol. Mol. Rev., 61, 65–89.
Rozalski, A., Kwil, I., Torzewska, A., Baranowska, M., and Staczek, P. (2007) Postepy Hig. Med. Dosw., 61, 204–219.
Knirel, Y. A., Perepelov, A. V., Kondakova, A. N., Senchenkova, S. N., Sidorczyk, Z., Rozalski, A., and Kaca, W. (2011) Innate Immun., 17, 70–96.
Zablotni, A., Zych, K., Kondakova, A. N., Siwinska, M., Knirel, Y. A., and Sidorczyk, Z. (2007) Arch. Immunol. Ther. Exp., 55, 347–352.
Knirel, Y. A., Kaca, W., Rozalski, A., and Sidorczyk, Z. (1999) Polish J. Chem., 73, 895–907.
Westphal, O., and Jann, K. (1965) Meth. Carbohydr. Chem., 5, 83–91.
Fudala, R., Kondakova, A. N., Bednarska, K., Senchenkova, S. N., Shashkov, A. S., Knirel, Y. A., Zahringer, U., and Kaca, W. (2003) Carbohydr. Res., 338, 1835–1842.
Laemmli, U. K. (1970) Nature, 227, 680–685.
Tsai, C. M., and Frasch, C. E. (1982) Anal. Biochem., 119, 115–119.
Kondakova, A. N., Fudala, R., Senchenkova, S. N., Shashkov, A. S., Knirel, Y. A., and Kaca, W. (2003) Carbohydr. Res., 338, 1191–1196.
Fresno, S., Jimenez, N., Canals, R., Merino, S., Corsaro, M. M., Lanzetta, R., Parrilli, M., Pieretti, G., Regue, M., and Tomas, J. M. (2007) J. Bacteriol., 189, 1128–1137.
Kondakova, A. N., Lindner, B., Fudala, R., Senchenkova, S. N., Moll, H., Shashkov, A. S., Kaca, W., Zahringer, U., and Knirel, Y. A. (2004) Biochemistry (Moscow), 69, 1034–1043.
Kondakova, A. N., Fudala, R., Senchenkova, S. N., Shashkov, A. S., Knirel, Y. A., and Kaca, W. (2003) Carbohydr. Res., 338, 1191–1196.
Kondakova, A. N., Fudala, R., Bednarska, K., Senchenkova, S. N., Knirel, Y. A., and Kaca, W. (2004) Carbohydr. Res., 339, 623–628.
Fudala, R., Kondakova, A. N., Bednarska, K., Senchenkova, S. N., Shashkov, A. S., Knirel, Y. A., Zahringer, U., and Kaca, W. (2003) Carbohydr. Res., 338, 1835–1842.
Kondakova, A. N., Senchenkova, S. N., Gremyakov, A. I., Shashkov, A. S., Knirel, Y. A., Fudala, R., and Kaca, W. (2003) Carbohydr. Res., 338, 2387–2392.
Kondakova, A. N., Fudala, R., Senchenkova, S. N., Shashkov, A. S., Knirel, Y. A., and Kaca, W. (2005) Carbohydr. Res., 340, 1612–1617.
Larsson, P. (1984) Methods Microbiol., 14, 187–214.
Penner, J. L., and Hennessy, J. N. (1980) J. Clin. Microbiol., 12, 304–309.
Sedlak, J., Tomasoffova, A., and Hatala, M. (1959) J. Hyg. Epidemiol. Microbiol. Immunol., 3, 422–430.
Palusiak, A., Dzieciatkowska, M., and Sidorczyk, Z. (2008) Arch. Immunol. Therap. Exp., 56, 135–140.
Aquilini, E., Azevedo, J., Jimenez, N., Bouamama, L., Tomas, J. M., and Regue, M. (2010) J. Bacteriol., 192, 4413–4424.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Russian in Biokhimiya, 2011, Vol. 76, No. 7, pp. 1039–1050.
This paper is based on a presentation made at the 4th Baltic Conference on Microbial Carbohydrates, Hyytiälä Forestry Field Station, Finland, September 19–22, 2010.
Rights and permissions
About this article
Cite this article
Kaca, W., Glenska, J., Lechowicz, L. et al. Serotyping of Proteus mirabilis clinical strains based on lipopolysaccharide O-polysaccharide and core oligosaccharide structures. Biochemistry Moscow 76, 851–861 (2011). https://doi.org/10.1134/S0006297911070169
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0006297911070169