Abstract
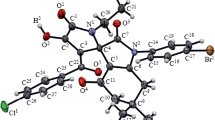
The reactions of 3-arylamino-1,4-naphthoquinone-4-oximes with 2,2-dihydroxyindane-1,3-dione proceed without involving the oxime group and lead to (6E)-5-arylamino-4b,11b-dihydroxy-6-hydroxyimino-4b,5-dihydrobenzo[f]indeno[1,2-b]indole-11,12(6H,11bH)-diones. The reactions of 2-arylamino(alkylamino)-1,4-naphthoquinone-4-oximes with 2,2-dihydroxy-1,3-indandione involve the oxime group and lead to 6-[arylamino(alkylamino)]-6b,11b-dihydroxy-5,7-dioxo-5,6b,7,11b-tetrahydrobenzo[g]indeno[1,2-b]indole 12-oxides.





Similar content being viewed by others
REFERENCES
Tandon, V.K., Maurya, H.K., Mishra, N.N., and Shukla, P.K., Eur. J. Med. Chem., 2009, vol. 44, p. 3130. https://doi.org/10.1016/j.ejmech.2009.03.006
Zhang, J., Redman, N., Litke, A.P., Zhan, J., Chan, K.Y., and Chang, C.-W.T., Bioorg. Med. Chem., 2011, vol. 19, p. 498. https://doi.org/10.1016/j.bmc.2010.11.001
Chan, K.Y., Zhang, J., and Chang, C.-W.T., Bioorg. Med. Chem. Lett., 2011, vol. 21, p. 6353. https://doi.org/10.1016/j.bmcl.2011.08.107
Fosso, M.Y., Chan, K.Y., Gregory, R., and Chang, C.W., ACS Combinat. Sci., 2012, vol. 14, p. 231. https://doi.org/10.1021/co2002075
Beachy, Ph.A., Chen, J.K., and Mann, R.K., WO Patent no. WO 200533048; Chem. Abstr., 2005, vol. 142, no. 386022.
Gornostaev, L.M., Tsvetkov, V.B., Markova, A.A., Lavrikova, T.I., Khalyavina, Y.G., Kuznetsova, A.S., Kaluzhny, D.N., Shunayev, A.V., Tsvetkova, M.V., Glazunova, V.A., Chernyshev, V.V., and Shtil, A.A., Anti-Cancer Agents Med. Chem., 2017, vol. 17, p. 1814. https://doi.org/10.2174/1871520617666170327112216
Gornostaev, L.M., Fominykh, O.I., Rukovets, T.A., Lavrikova, T.I., Khalyavina, J.G., Shtil, A.A., Dunaev, S.F., Murashova, E.V., and Chernyshev, V.V., Chem. Heterocycl. Compd., 2020, vol. 56, p. 47. https://doi.org/10.1007/s10593-020-02621-8
Goldstein, H. and Grandjean, P., Helv. Chim. Acta, 1943, vol. 43, p. 468.
Gornostaev, T.A., Rukovets, E.V., Arnold, Yu.G., Khalyavina, Yu.V., and Gatilov, U.V., Russ. J. Org. Chem., 2018, vol. 54, p. 78. https://doi.org/10.1134/S1070428018010062
Gornostaev, L.M., Fominyh, O.I., Lavrikova, T.I., Khalyavina, Yu.G., Gatilov, Yu.V., and Stashina, G.A., Russ. Chem. Bull., 2019, vol. 68, p. 86. https://doi.org/10.1007/s11172-019-2420-8
Cambridge Crystallographic Data Centre (CCDC), https://www.ccdc.cam.ac.uk/strucrures
Funding
The work was financially supported by the Russian Foundation for Basic Research (project no. 18-33-00663).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare no conflict of interest.
Additional information
Translated from Zhurnal Organicheskoi Khimii, 2021, Vol. 57, No. 2, pp. 194–200 https://doi.org/10.31857/S0514749221020051.
Rights and permissions
About this article
Cite this article
Gornostaev, L.M., Rudenko, D.S., Rukovets, T.A. et al. Reactions of (4E)-3-Arylamino-4-(hydroxyimino)naphthalen-1(4H)-ones and (4E)-2-[Arylamino(alkylamino)]-4-(hydroxyimino)naphthalen-1(4H)-ones with 2,2-Dihydroxyindane-1,3-dione. Russ J Org Chem 57, 165–169 (2021). https://doi.org/10.1134/S1070428021020056
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070428021020056