Abstract
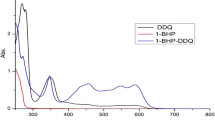
Electron donor acceptor complexes (EDA) of the phenytoin sodium (Pyn) as a rich donor with different π-acceptors like p-chloranil (p-CHL), 2,3-dichloro-5,6-dicyano-1,4-benzoquinone (DDQ), tetracyanoquinodimethane (TCNQ), chloranilic acid (CLA), N-bromosuccinimide (NBS) and picric acid (PA) have been synthesized. Spectroscopic and physical data such as formation constant (K CT), molar extinction coefficient (ɛCT), standard free energy (ΔG 0), oscillator strength (f), transition dipole moment (μ), resonance energy (R N) and ionization potential (I p) have been determined. Based on the elemental analysis and photometric titrations the CT-complexes were found to have 1 : 1 stoichiometry.
Similar content being viewed by others
References
Rogawski, M.A. and Löscher, W., Nat. Rev. Neurosci., 2004, vol. 5. p. 553.
AHFS Drug Information 2004, McEvoy, G.K., Ed., American Society of Health-System Pharmacists, 2004, pp. 2117–2120.
Kosower, E.M., Prog. Phys. Org. Chem., 1965, vol. 3, p. 81.
Fla, F.P., Palou, J., Valero, R., Hall, C.D., and Speers, P., JCS Perkin Trans. 2, 1991, p. 1925.
Yakuphanoglu, F. and Arslan, M., Opt. Mater., 2004, vol. 27, p. 29.
Yakuphanoglu, F. and Arslan, M., Solid State Commun., 2004, vol. 132, p. 229.
Yakuphanoglu, F., Arslan, M., Kucukislamoglu, M., and Zengin, M., Sol. Energy, 2005, vol. 79, p. 96.
Chakraborty, B., Mukherjee, A.S., and Seal, B.K., Spectrochim. Acta A, 2001, vol. 57, p. 223.
Korolkovas, A., Essentials of Medical Chem., 2 ed., New York: Wiley, ch. 3, 1998.
Takahasi, K., Horino, K., Komura, T., and Murata, K., Bull. Chem. Soc. Jpn. 1993, vol. 66, p. 733.
Andrade, S.M., Costa, S.M.B., and Pansu, R., J. Colloid. Interf. Sci., 2000, vol. 226, p. 260.
Slifkin, A.M., Charge-Transfer Interaction of Biomolecules, New York: Academic Press, 1971.
Abou Attia, F.M., Farmaco, 2000, vol. 55, p. 659.
Basavaiah, K., Farmaco, 2004, vol. 59, p. 315.
Das, S.K., Krishnamoorthy, G., and Dofra, S.K., Can. J. Chem., 2000, vol. 78, p. 191.
Jones, G., and Jimenez, J.A.C., Tetrahedron Lett., 1999, vol. 40, p. 8551.
Smith, G., Bott, R.C., Rae, A.D., and Willis, A.C., Aust. J. Chem., 2000, vol. 53, p. 531.
Smith, G., Lynch, D.E., and Bott, R.C., Aust. J. Chem., 1998, vol. 51, p. 159.
Smith, G., Lynch, D.E., Byriel, K.A., and Kennard, C.H.L., J. Chem. Crystallogr., 1997, vol. 27, p. 307.
Refat, M.S., Ibrahim, O.B., Saad, H.A., and Adam, A.M.A., J. Mol. Struct., 2014, vol. 1064, p. 58.
Eldaroti, H.H., Gadir, S.A., Refat, M.S., and Adam, A.M.A., J. Pharm. Anal., 2014, vol. 4, p. 81.
El-Habeeb, A.A., Al-Saif, F.A., and Refat, M.S., Spectrochim. Acta Part A, 2014, vol. 123, p. 455.
Elqudaby, H.M., Mohamed, G.G., and El-Din, G.M.G., Spectrochim. Acta Part A., 2014, vol. 129, p. 84.
Eldaroti, H.H., Gadir, S.A., Refat, M.S., and Adam, A.M.A., Spectrochim. Acta Part A, 2013, vol. 115, p. 309.
El-Habeeb, A.A., Al-Saif, F.A., and Refat, M.S., J. Mol. Struct., 2013, vol. 1036, p. 464.
El-Habeeb, A.A., Al-Saif, F.A., and Refat, M.S., J. Mol. Struct., 2013, vol. 1034, p. 1.
Skoog, D.A., Principle of Instrumental Analysis, 3 ed., New York: Saunders College Publishing, 1985, ch. 7.
Abu-Eittah, R. and Al-Sugeir, F., Can. J. Chem., 1976, vol. 54, p. 3705.
Tsubomura, H. and Lang, R.P., J. Am. Chem. Soc., 1964, vol. 86, p. 3930.
Teleb, S.M. and Refat, M.S., Spectrochimica Acta Part A, 2004, vol. 60, p. 1579.
Nour, E.M., Teleb, S.M., El-Mosallamy, M.A.F., and Refat, M.S., South Afr. J. Chem., 2003, vol. 56, p. 10.
Rathone, R., Lindeman, S.V., and Kochi, J.K., J. Am. Chem. Soc., 1997, vol. 119, p. 9393.
Aloisi, G, and Pignataro, S., J. Chem. Soc. Faraday Trans., 1972, vol. 69, p. 534.
Briegleb, G., Z. Angew. Chem., 1960, vol. 72, p. 401.
Briegleb, G., Z. Angew. Chem., 1964, vol. 76, p. 326.
Briegleb, G., and Czekalla, J., Z. Physikchem. (Frankfurt), 1960, vol. 24, p. 237.
Martin, A.N., Swarbrick, J., and Cammarata, A., Physical Pharmacy, 3 ed., Philadelphia: Lee and Febiger, 1969, p. 344.
Bellamy, L.J., The Infrared Spectra of Complex Molecules, London: Chapman & Hall, 1975.
Kross, R.D. and Fassel, V.A., J. Am. Chem. Soc., 1957, vol. 79, p. 38.
Foster, R., Organic Charge-Transfer Complexes, London: Academic Press, 1969, pp. 51, 387.
Fukuzumi, S. and Kochi, J.K., J. Am. Chem. Soc., 1982, vol. 104, p. 7599.
Melby, L.R., Harder, R.J., Hertler, W.R., Benson, R.L., and Mochel, W.E., J. Am. Chem. Soc., 1962, vol. 84, p. 3374.
Garito, A.F. and Heeger, A.J., J. Acc. Chem. Res., 1974, vol. 7, p. 232.
Wheland, R.C. and Gillson, J.L., J. Am. Chem. Soc., 1976, vol. 98, p. 3916.
Bell, S.E.J. and Sirimuthu, N.M.S., Chem. Soc. Rev., 2008, vol. 37, p. 1012.
Kagan, M.R. and McCreery, R.L., Anal. Chem., 1994, vol. 66, p. 4159.
Shamsipur, M. and Mashhadizadeh, M., J. Incl. Phenom. & Macrocyclic Chem., 2000, vol. 38, p. 277.
Author information
Authors and Affiliations
Corresponding author
Additional information
The text was submitted by the authors in English.
Rights and permissions
About this article
Cite this article
El-Habeeb, A.A., Refat, M.S. Spectroscopic studies on the utility of charge transfer interactions for determination of phenytoin sodium. Russ J Gen Chem 85, 939–950 (2015). https://doi.org/10.1134/S1070363215040313
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363215040313