Abstract
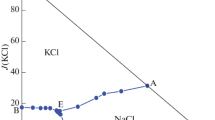
The solubility in the 2Na+,Mg2+‖2Cl−, 2ClO −3 -H2O system was studied at 20 and 100°C and the solubility diagrams were plotted. New compounds were not found to form in the title quaternary reciprocal system. The sodium chloride field was observed to expand with rising temperature.
Similar content being viewed by others
References
A. Kh. Kul’maksimov, Extended Abstract of Candidate’s Dissertation in Chemistry (Ashkhabad, 1989).
A. G. Bergman and N. P. Luzhnaya, Physicochemical Foudnations of Exploration of and Use of Chloride-Sulfate Salt Deposits (Akad. Nauk SSSR, Moscow, 1951) [in Russian].
Yu. M. Martynov, M. A. Matveev, L. M. Yakimenko, and A. A. Furman, Khim. Prom-st., No. 7, 420 (1958).
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.B. Azlarov, Kh.M. Polvonov, M.K. Askarova, S. Tukhtaev, 2011, published in Zhurnal Neorganicheskoi Khimii, 2011, Vol. 56, No. 1, pp. 128–132.
Rights and permissions
About this article
Cite this article
Azlarov, A.B., Polvonov, K.M., Askarova, M.K. et al. Isothermal solubility diagram of the 2Na+,Mg2+‖2Cl−, 2ClO ‒3 -H2O quaternary system at 20 and 100°C. Russ. J. Inorg. Chem. 56, 124–127 (2011). https://doi.org/10.1134/S0036023611010025
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036023611010025