Abstract
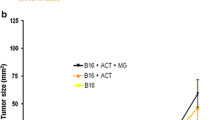
Melanoma is one of the most aggressive tumors and is accompanied by the induction of local and systemic inflammatory responses. Combinations of chemotherapeutic agents with immunotherapy are therefore commonly used for melanoma treatment. A B16 melanoma model was used to study the tumor suppressive, immunostimulating, and hepatotoxic effects of a combination of a small double-stranded immunostimulatory RNA (isRNA) with 3'-trinucleotide overhangs and the cytotoxic drug dacarbazine compared with respective monotherapies. The drugs efficiently suppressed the tumor growth and acted synergistically. Histological and immunohistochemical examinations of tumor nodes showed that the combination of isRNA and dacarbazine significantly decreased mitotic activity and more efficiently increased apoptosis in tumor tissue as compared with either monotherapy. Regardless of the treatment regimen, signs of immune activation were observed in the spleen, including an increase in the number and diameter of lymphoid follicles and the volume density of the white pulp. Destructive changes were detected in the livers of nontreated animals with B16 melanoma. Administration of isRNA in combination with dacarbazine did not cause any additional damage to liver parenchyma, while stimulating regenerative processes in hepatic tissue of tumor-bearing animals.


Similar content being viewed by others
REFERENCES
Miller K.D., Nogueira L., Mariotto A.B., Rowland J.H., Yabroff K.R., Alfano C.M., Jemal A., Kramer J.L., Siegel R.L. 2019. Cancer treatment and survivorship statistics, 2019. CA Cancer J. Clin.69 (5), 363–385. https://doi.org/10.3322/caac.21565
Tromme I., Legrand C., Devleesschauwer B., Leiter U., Suciu S., Eggermont A., Francart J., Calay F., Haagsma J.A., Baurain J.F., Thomas L., Beutels P., Speybroeck N. 2016. Melanoma burden by melanoma stage: Assessment through a disease transition model. Eur. J. Cancer. 53, 33–41.
Wu Y.P., Aspinwall L.G., Conn B.M., Stump T., Grahmann B., Leachman S.A. 2016. A systematic review of interventions to improve adherence to melanoma preventive behaviors for individuals at elevated risk. Prev. Med.788, 153–167.
Eggermont A.M., Spatz A., Robert C. 2014. Cutaneous melanoma. Lancet.383 (9919), 816–827.
Wilson M.A., Schuchter L.M. 2016. Chemotherapy for melanoma. Cancer Treat. Res.167, 209–229.
Luo C., Shen J. 2017. Research progress in advanced melanoma. Cancer Lett.397, 120–126.
Al-Badr A.A., Alodhaib M.M. 2016. Dacarbazine. Profiles Drug Subst. Excip. Relat. Methodol.41, 323–377.
Hölzel M., Tüting T. 2016. Inflammation-induced plasticity in melanoma therapy and metastasis. Trends Immunol.37 (6), 364–374.
Neagu M., Constantin C., Caruntu C., Dumitru C., Surcel M., Zurac S. 2019. Inflammation: A key process in skin tumorigenesis. Oncol. Lett.17 (5), 4068–4084.
Eggert T., Medina-Echeverz J., Kapanadze T., Kruhlak M.J., Korangy F., Greten T.F. 2014. Tumor induced hepatic myeloid derived suppressor cells can cause moderate liver damage. PLoS One.29 (11), e112717. https://doi.org/10.1371/journal.pone.0112717
Rodríguez-Cerdeira C., Carnero Gregorio M., López-Barcenas A., Sánchez-Blanco E., Sánchez-Blanco B., Fabbrocini G., Bardhi B., Sinani A., Guzman R.A. 2017. Advances in immunotherapy for melanoma: A comprehensive review. Mediat. Inflamm., 2017, Art. ID 3264217.https://doi.org/10.1155/2017/3264217
Franklin C., Livingstone E., Roesch A., Schilling B., Schadendorf D. 2017. Immunotherapy in melanoma: Recent advances and future directions. Eur. J. Surg. Oncol.43 (3), 604–611.
Kabilova T.O., Meschaninova M.I., Venyaminova A.G., Nikolin V.P., Zenkova M.A., Vlassov V.V., Chernolovskaya E.L. 2012. Short double-stranded RNA with immunostimulatory activity: Sequence dependence. Nucl. Acids Ther.22 (3), 196–204.
Kabilova T.O., Kovtonyuk L.V., Zonov E.V., Ryabchikova E.I., Popova N.A., Nikolin V.P., Kaledin V.I., Zenkova M.A., Vlassov V.V., Chernolovskaya E.L. 2014. Immunotherapy of hepatocellular carcinoma with small double-stranded RNA. BMC Cancer.14, 338. https://doi.org/10.1186/1471-2407-14-338
Kabilova T.O., Sen’kova A.V., Nikolin V.P., Popova N.A., Zenkova M.A., Vlassov V.V., Chernolovskaya E.L. 2016. Antitumor and antimetastatic effect of small immunostimulatory RNA against B16 melanoma in mice. PLoS One.11 (3), e0150751. https://doi.org/10.1371/journal.pone.0150751
Maslov M.A., Kabilova T.O., Petukhov I.A., Morozova N.G., Serebrennikova G.A., Vlassov V.V., Zenkova M.A. 2012. Novel cholesterol spermine conjugates provide efficient cellular delivery of plasmid DNA and small interfering RNA. J. Control. Release.160, 182–193.
Patutina O.A., Mironova N.L., Ryabchikova E.I., Popova N.A., Nikolin V.P., Kaledin V.I., Vlassov V.V., Zenkova M.A. 2011. Inhibition of metastasis development by daily administration of ultralow doses of RNase A and DNase I. Biochimie.93 (4), 689–696.
Chou T.C. 2006. Theoretical basis, experimental design, and computerized simulation of synergism and antagonism in drug combination studies. Pharmacol. Rev.58 (3), 621–681.
Sen’kova A.V., Mironova N.L., Patutina O.A., Ageeva T.A., Zenkova M.A. 2012. The toxic effects of polychemotherapy onto the liver are accelerated by the upregulated MDR of lymphosarcoma. ISRN Oncol.2012, Art. ID 721612. https://doi.org/10.5402/2012/721612
Sen’kova A.V., Mironova N.L., Patutina O.A., Mitkevich V.A., Markov O.V., Petrushanko I.Y., Burnysheva K.M., Zenkova M.A., Makarov A.A. 2014. Ribonuclease binase decreases destructive changes of the liver and restores its regeneration potential in mouse lung carcinoma model. Biochimie.101, 256–259.
Teicher B.A. 2006. Tumor models for efficacy determination. Mol. Cancer Ther.5 (10), 2435–2443.
Garbe C., Eigentler T.K., Keilholz U., Hauschild A., Kirkwood J.M. 2011. Systematic review of medical treatment in melanoma: Current status and future prospects. Oncologist.16 (1), 5–24.
Mocellin S., Pasquali S., Rossi C.R., Nitti D. 2010. Interferon alpha adjuvant therapy in patients with high-risk melanoma: A systematic review and meta-analysis. J. Natl. Cancer Inst.102 (7), 493–501.
Yan Y., Leontovich A.A., Gerdes M.J., Desai K., Dong J., Sood A., Santamaria-Pang A., Mansfield A.S., Chadwick C., Zhang R., Nevala W.K., Flotte T.J., Ginty F., Markovic S.N. 2019. Understanding heterogeneous tumor microenvironment in metastatic melanoma. PLoS One. 14 (6), e0216485. https://doi.org/10.1371/journal.pone.0216485
Domingues B., Lopes J.M., Soares P., Pópulo H. 2018. Melanoma treatment in review. Immunotargets Ther.7, 35–49.
Baharara J., Amini E., Nikdel N., Salek-Abdollahi F. 2016. The cytotoxicity of dacarbazine potentiated by sea cucumber saponin in resistant B16F10 melanoma cells through apoptosis induction. Avicenna J. Med. Biotechnol.8 (3), 112–129.
Jin J.L., Gong J., Yin T.J., Lu Y.J., Xia J.J., Xie Y.Y., Di Y., He L., Guo J.L., Sun J., Noteborn M.H., Qu S. 2011. PTD4-apoptin protein and dacarbazine show a synergistic antitumor effect on B16-F1 melanoma in vitro and in vivo.Eur. J. Pharmacol.654 (1), 17–25.
Urosevic-Maiwald M., Barysch M.J., Cheng P.F., Karpova M.B., Steinert H., Okoniewski M.J., Dummer R. 2015. In vivo profiling reveals immunomodulatory effects of sorafenib and dacarbazine on melanoma. Oncoimmunology.4 (2), e988458.
Al-Badr A.A., Alodhaib M.M. 2016. Dacarbazine. Profiles Drug Subst. Excip. Relat. Methodol.41, 323–377.
Mishra H., Mishra P.K., Ekielski A., Jaggi M., Iqbal Z., Talegaonkar S. 2018. Melanoma treatment: From conventional to nanotechnology. J. Cancer. Res. Clin. Oncol.144 (12), 2283–2302.
George D.D., Armenio V.A., Katz S.C. 2017. Combinatorial immunotherapy for melanoma. Cancer Gene Ther.24 (3), 141–147.
Rodríguez-Cerdeira C., Carnero Gregorio M., López-Barcenas A., Sánchez-Blanco E., Sánchez-Blanco B., Fabbrocini G., Bardhi B., Sinani A., Guzman R.A. 2017. Advances in immunotherapy for melanoma: A comprehensive review. Mediat. Inflamm.2017, Art. ID 3264217.https://doi.org/10.1155/2017/3264217
Jiang G., Li R.H., Sun C., Liu Y.Q., Zheng J.N. 2014. Dacarbazine combined targeted therapy versus dacarbazine alone in patients with malignant melanoma: A meta-analysis. PLoS One.9 (12), e111920.https://doi.org/10.1371/journal.pone.0111920
Sasse A.D., Sasse E.C., Clark L.G., Ulloa L., Clark O.A. 2007. Chemoimmunotherapy versus chemotherapy for metastatic malignant melanoma. Cochrane Database Syst. Rev.1, CD005413.
Yang C., Cao H., Liu N., Xu K., Ding M., Mao L.J. 2016. Oncolytic adenovirus expressing interleukin-18 improves antitumor activity of dacarbazine for malignant melanoma. Drug Des. Devel. Ther.10, 3755–3761.
Zhang X.H., Qiao E.Q., Gao Z., Yuan H.Q., Cai P.F., Li X.M., Gu Y.H. 2013. Efficacy of combined axitinib with dacarbazine in a B16F1 melanoma xenograft model. Oncol. Lett. 6 (1), 69–74.
Urosevic-Maiwald M., Barysch M.J., Cheng P.F., Karpova M.B., Steinert H., Okoniewski M.J., Dummer R. 2015. In vivo profiling reveals immunomodulatory effects of sorafenib and dacarbazine on melanoma. Oncoimmunology. 4 (2), e988458.
ACKNOWLEDGMENTS
We are grateful to M.I. Meshchaninova and A.G. Ven’ya-minova (Institute of Chemical Biology and Fundamental Medicine) for isRNA synthesis.
Funding
This work was supported by the Russian Science Foundation (project no. 19-74-30011) and by the Russian State Funded Budget Project of ICBFM SB RAS (project no. AAAA-A17-117020210024-8).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interests. The authors declare that they have no conflict of interest.
Statement on the welfare of animals. Experiments with animals followed the guidelines established by The Directive 2010/63/EU of the European Parliament and the Council of 22 September 2010 and were approved by the Ethics Committee at the Institute of Cytology and Genetics (Novosibirsk, Russia; Minutes no. 22.11 dated May 30, 2014).
Additional information
Translated by T. Tkacheva
Abbreviations: isRNA, immunostimulating RNA; TGS, tumor growth suppression; CI, combination index; DT, doubling time.
Rights and permissions
About this article
Cite this article
Sen’kova, A.V., Savin, I.A., Kabilova, T.O. et al. Tumor-Suppressing, Immunostimulating, and Hepatotoxic Effects of Immunostimulatory RNA in Combination with Dacarbazine in a Murine Melanoma Model. Mol Biol 54, 233–242 (2020). https://doi.org/10.1134/S0026893320020144
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0026893320020144