Abstract
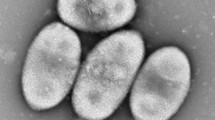
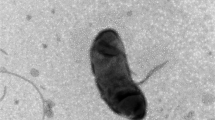
Two strains, NF4 and NF5, of a yellow-colored gram-negative bacterium were isolated from sediments of Lake Baikal and from old oil sludge of the Nizhnekamsk oil-processing plant. The cells of the strains are ultrasmall coccoids or short rods, measuring 0.2–0.4 × 0.2–0.5 μm; the average cell volume ranges from 0.004 to 0.04 μm3. A considerable proportion (30–60%) of cells have nanometer dimensions (180–300 nm in diameter and 0.004–0.02 μm3 in volume). The new isolates are thus among the smallest representatives of presently known free-living ultramicrobacteria. The two studied isolates are gram-negative nonmotile cells possessing a pronounced outer membrane. The cells do not have flagella and are not capable of gliding motility. They divide by constriction, budding, and multiple septation. The multiplicity of reproduction mechanisms results in a high degree of cell polymorphism. The isolates are chemoorganotrophic, aerobic, psychrotolerant, oxidase- and catalase-positive. Their characteristic trait is the absence of extracellular hydrolytic enzymes, such as proteases, lipases, pectinases, and cellulases. Menaquinone MK-6 is the main respiratory quinone; the flexirubin pigment was not detected. The G + C contents of the DNA of strains NF4 and NF5 are 40.8 and 40.5 mol %, respectively. The DNA-DNA hybridization level of strains NF4 and NF5 was close to 100%. Analysis of the 16S rRNA gene sequences and the fatty acid compositions showed that the isolates are most closely related to certain representatives of the genus Chryseobacterium (C. solincola, C. antarcticum, and C. jeonii). However, the differences in the 16S rRNA gene sequences, as well as in the phenotypic properties, such as formation of ultrasmall cells, the absence of extracellular hydrolases, oligotrophy, and the capacity for epibiosis on bacterial cells, suggest that the studied strains belong to a new species of the genus Chryseobacterium. The capacity for epibiosis, i.e., the ability to exist in a tightly adhered state on the surfaces of host Bacillus subtilis cells, is a peculiar trait of the studied isolates. It is assumed that adhesion of the cells of strains NF4 and NF5 (members of the phylum Bacteroidetes) occurs via by the same unique mechanism as the mechanism that we previously described for representatives of Alphaproteobacteria (Kaistia sp., NF1, and NF3), which use polysaccharide chains equipped with sticky granules as trapping and constricting cords.
Similar content being viewed by others
References
Kmpfer, P., Lodders, N., Vaneechoutte, M., and Wauters, G., Transfer of Sejongia antarctica, Sejongia jeonii, and Sejongia marina to the Genus Chryseobacterium as Chryseobacterium antarcticum comb. nov., Chryseobacterium jeonii comb. nov. and Chryseobacterium marinum comb. nov, Int. J. Syst. Evol. Microbiol, 2009, vol. 59, pp. 2238–2240.
Vandamme, P., Bernardet, J.F., Segers, P., Kersters, K., and Holmes, B., New Perspectives in the Classification of the Flavobacteria: Description of Chryseobacterium gen. nov., Bergeyella gen. nov., and Empedobacter nom. rev, Int. J. Syst. Bacteriol., 1994, vol. 44, pp. 827–831.
Euzby, J.P., List of Prokaryotic Names with Standing in Nomenclature — Genus Chryseobacterium, 2009. URL: www.bacterio.net pp. 1–19
Yi, H., Yoon, Ho., and Chun, J., Sejongia antarctica gen. nov., sp. nov. and Sejongia jeonii sp. nov., Isolated from the Antarctic, Int. J. Syst. Evol. Microbiol, 2005, vol. 55, pp. 409–416.
Kmpfer, P., Vaneechoutte, M., Lodders, N., De Baere, T., Avesani, V., Janssens, M., Busse, H.-J., and Wauters, G., Description of Chryseobacterium antropy sp. nov. To Accommodate Clinical Isolates Biochemically Similar to Kaistella koreensis and Chryseobacterium haifense, Proposal To Reclassify Kaistella koreensis as Chryseobacterium koreense comb. nov. and Emended Description of the Genus Chryseobacterium, Int. J. Syst. Evol. Microbiol, 2009, vol. 59, pp. 2421–2428.
Benmalek, Y., Cayol, J.-L., Bouanane, N.A., Hacene, H., Fauque, G., and Fardeau, M.-L., Chryseobacterium solincola sp. nov. Isolated from Soil, Int. J. Syst. Evol. Microbiol, 2010, vol. 60, pp. 1876–1880.
Cone, L.A., Cronin, T.J., Curti, M., and Friscia, D., Osteomyelitis Due to Chryseobacterium (Flavobacterium) meningosepticum, Antimicrob. Infect. Dis. News, 1998, vol. 17, pp. 60–62.
Miteva, V.I. and Brenchley, J.E., Detection and Isolation of Ultrasmall Microorganisms from a 120,000-Year-Old Greenland Glacier Ice Core, J. Appl. Environ. Microbiol., 2005, vol. 71, pp. 7806–7818.
Smibert, R.M. and Krieg, N.R., Phenotypic Characterization, Methods for General and Molecular Bacteriology, Gerhardt, P., Murray, R.G.E., Wood, W.A., and Krieg, N.R., Eds., Washington, DC: Am. Soc. Microbiol., 1994.
Bernardet, J.F., Nakagawa, Y., and Holmes, B., Proposed Minimal Standards for Describing New Taxa of the Family Flavobacteriaceae and Emended Description of the Family, Int. J. Syst. Evol. Microbiol, 2002, vol. 52, pp. 1049–1070.
McCammon, S.A. and Bowman, J.P., Taxonomy of Antarctic Flavobacterium Species: Description of Flavobacterium gillisiae sp. nov., Flavobacterium tegetincola sp. nov., and Flavobacterium xanthum sp. nov., nom. rev. and Reclassification of [Flavobacterium] salegens as Salegentibacter salegens gen. nov., comb. Nov., Int. J. Syst. Evol. Microbiol, 2000, vol. 50, pp. 1055–1063.
Suzina, N.E., Esikova, T.Z., Akimov, V.N, Abashina, T.N., Dmitriev, V.V., Polivtseva, V.N., Duda, V.I., and Boronin, A.M., Study of Ectoparasitism of Ultramicrobacteria of the Genus Kaistia, strains NF1 and NF3 by Electron and Fluorescence Microscopy,, Mikrobiologiya, 2008, vol. 77, pp. 55–62 [Microbiology (Engl. Transl.), vol. 77, no. 1, pp. 47–54].
Hector, J.S.R. and Johnson, A.R., Determination of Genome Size of Pseudomonas aeruginosa by PFGE: Analysis of Restriction Fragments, Nucleic Acids Res., 1990, vol. 18, pp. 3171–3174.
Duda, V.I., Suzina, N.E., Esikova, T.Z., Akimov, V.N., Oleinikov, R.R., Polivtseva, V.N., Abashina, T.N., Shorokhova, A.P., and Boronin, A.M., The Cytological Characterization of Parasitic Action of Ultramicrobacteria NF1 and NF3 from the Genus Kaistia on Chemoorganotrophic and Phototrophic Bacteria, FEMS Microbiol. Ecol., 2009, vol. 69, pp. 180–199.
Luft, J.H., Electron Microscopy of Cell Extraneous Coats as Revealed by Ruthenium Red Staining, J. Cell Biol., 1964, p. 54.
Reynolds, E., The Use of Lead Citrate at High pH as an Electron Opaque Stain in Electron Microscopy, J. Cell Biol., 1963, vol. 17, pp. 208–212.
Janssen, P.H., Schuhmann, A.M., Mrrschel, E., and Rainey, F.A., Novel Anaerobic Ultramicrobacteria Belonging to the Verrucomicrobiales Lineage of Bacterial Descent Isolated by Dilution Culture from Anoxic Rice Paddy Soil, Appl. Environ. Microbiol., 1997, vol. 63, pp. 1382–1388.
Zwillenberg, L.O., Electron Microscopic Features of Gram-Negative and Gram-Positive Bacteria Embedded in Phosphotungstate, Antonie van Leeuwenhoek, 1964, vol. 30, pp. 154–162.
Duda, V.I., Suzina, N.E., Esikova, T.Z., and Polivtseva, V.N., and Boronin, A.M., A New Mechanism of Cell Cohesion during Interaction between Predatory Bacteria and Their Preys, Dokl. Akad. Nauk, 2008, vol. 422, pp. 841–844 [Dokl. Biol. Sci. (Engl. Transl.), 2008,, vol. 422, p. 369].
Vasil’eva, L.V. and Zavarzin, G.A., Dissipotrophs in the Microbial Community, Mikrobiologiya, 1995, vol. 64, pp. 239–244.
Stolp, H., The Bdellovibrios: Bacterial Parasites of Bacteria, Annu. Rev. Phytopathol., 1973, vol. 11, pp. 53–76.
Davidov, Y. and Jurkevitch, E., Diversity and Evolution of Bdellovibrio-and-Like Organisms (BALOS), Reclassification of Bacteriovorax atarrii as Peredibacter starrii gen. nov. comb. nov. and Description of the Bacteriovorax-Peredibacter Clade as Bacteriovoracaceae fam. nov, Int. J. Syst. Evol. Microbiol, 2004, vol. 54, pp. 1439–1452.
Jurkevitch, E., Predatory Behaviors in Bacteria—Diversity and Transitions, Microbe, 2007, vol. 2, pp. 67–73.
Martin, M.O., Predatory Prokaryotes: An Emerging Research Opportunity, J. Mol. Microbiol., 2002, vol. 4, pp. 467–477.
Gromov, B.V. and Mamkaeva, K.A., Proposal of the New Genus Vampirovibrio for the Chlorellavorus Bacteria, Formerly Assigned to Bdellovibrio, Mikrobiologiya, 1980, vol. 49, pp. 15–167.
Waters, E., Hohn, M.J., Ahel, I., Graham, D.E., Adams, M.D., Barnstead, M., Beeson, K.Y., Bibbs, L., Bolanos, R., Keller, M., Kretz, K., Lin, X., Mathur, E., Ni, J., Podar, M., Richardson, T., Sutton, G.G., Simon, M., Sll, D., Stetter, K.O., Short, J.M., and Noordewier, M., The Genome of Nanoarchaeum equitans: Insights into Early Archaeal Evolution and Derived Parasitism, Proc. Natl. Acad. Sci. USA, 2003, vol. 100, pp. 12984–12988.
Huber, H., Hohn, M.J., Rachel, R., Fuchs, T., Wimmer, V.C., and Stetter, K.O., A New Phylum of Archaea Represented by a Nanosized Hyperthermophilic Symbiont, Nature, 2002, vol. 417, pp. 63–67.
Gromov, B.V. and Pavlenko, G.V., Ekologiya bakterii (Ecology of Bacteria), Leningrad: Izdatel’stvo Leningradskogo universiteta, 1989.
Parasitism. Biologicheskii entsiklopedicheskii slovar’ (Biological Encyclopedia), Gilyarov, M.S., Ed., Moscow: Sovetskaya Entsiklopediya, 1986.
Kadouri, D. and O’Toole, G.A., Susceptibility of Biofilms to Bdellovibrio bacteriovorus Attack, Appl. Environ. Microbiol., 2005, vol. 71, pp. 4044–4051.
Kadouri, D., Venzon, N.C., and O’Toole, G.A., Vulnerability of Pathogenic Biofilms to Micavibrio aeruginosavorus, Appl. Environ. Microbiol., 2007, vol. 73, pp. 605–614.
Duda, V.I., Suzina, N.E., Akimov, V.N., Vainshtein, M.B., Dmitriev, V.V., Barinova, E.S., Abashina, T.N., Oleynikov, R.R., Esikova, T.Z., and Boronin, A.M., Ultrastructural Organization and Development Cycle of Soil Ultramicrobacteria Belonging to the Class Alphaproteobacteria, Mikrobiologiya, 2007, vol. 76, no. 5, pp. 652–661 [Microbiology (Engl. Transl.), vol. 76, no. 5, pp. 575–584].
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © N.E. Suzina, V.I. Duda, T. Z. Esikova, A.P. Shorokhova, A.B. Gafarov, R.R. Oleinikov, V.N. Akimov, T.N. Abashina, V.N. Polivtseva, A.M. Boronin, 2011, published in Mikrobiologiya, 2011, Vol. 80, No. 4, pp. 529–542.
Rights and permissions
About this article
Cite this article
Suzina, N.E., Duda, V.I., Esikova, T.Z. et al. Novel ultramicrobacteria, strains NF4 and NF5, of the genus Chryseobacterium: Facultative epibionts of Bacillus subtilis . Microbiology 80, 535–548 (2011). https://doi.org/10.1134/S0026261711040187
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0026261711040187