Abstract
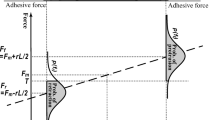
Under many circumstances, cell migration speed is limited by the rate of cell-substratum detachment at the cell rear. We have constructed a mathematical model to integrate how the biophysical and biochemical interactions between integrins, the cytoskeleton, and the matrix affect rear retraction and linkage dissociation mechanisms. Our model also examines how applied forces and integrin clustering affect retraction kinetics. The model predicts two distinct detachment phenotypes. In the first, detachment is extremely rapid, dominated by integrin extracellular-matrix dissociation, and it occurs at high forces or low adhesiveness. In the second, detachment is much slower, dominated by integrin-cytoskeleton dissociation, and it occurs at low forces or high adhesiveness. The amount of integrin extracted from the rear of the cell is an assay for the detachment phenotype. During rapid detachment cells leave little integrin on the substratum whereas during slow detachment a large fraction of integrin rips from the membrane. This model delineates parameters which can be exploited to regulate cell speed in each detachment regime. The model also offers an explanation as to why some cell types, such as leukocytes or keratocytes, are able to detach easily and move very quickly while other cell types, such as fibroblasts, tend to migrate more slowly and release many more integrins during detachment. © 1999 Biomedical Engineering Society.
PAC99: 8717Jj, 8717Aa, 8715Rn, 8716Dg
Similar content being viewed by others
REFERENCES
Akiyama, S. K., and K. M. Yamada. Interaction of plasma fibronectin with fibroblastic cells in suspension. J. Biol. Chem. 260:4492–4500, 1985.
Alon, R., S. Chen, K. D. Puri, E. B. Finger, and T. A. Springer. The kinetics of L-selection tethers and mechanics of selection-mediated rolling. J. Cell Biol. 138:1169–1180, 1997.
Bard, J. B. L., and E. D. Hay. The behavior of fibroblasts from the developing avian cornea. J. Cell Biol. 67:400–418, 1975.
Beckerle, M. C., K. Burridge, G. N. DeMartino, and D. E. Croall. Colocalization of calcium-dependent protease II and one of its substrates at sites of cell adhesion. Cell 51:569–577, 1987.
Bell, G. I. Models for the specific adhesion of cells to cells. Science 200:618–627, 1978.
Chen, W.-T. Mechanism of retraction of the trailing edge during fibroblast movement. J. Cell Biol. 90:187–200, 1981.
Crowley, E., and A. F. Horwitz. Tyrosine phosphorylation and cytoskeletal tension regulate the relase of fibroblast adhesions. J. Cell Biol. 131:525–537, 1995.
de Beus, E., A. de Beus, and K. Jacobson. The role of β1-integrin mediated adhseions in Xenopus laevis keratocyte locomotion. Mol. Biol. Cell Suppl. 8:264a, 1997.
DiMilla, P. A., K. Barbee, and D. A. Lauffenburger. Mathematical model for the effects of adhesion and mechanics on cell migration speed. Biophys. J. 60:15–37, 1991.
Evans, E. A., D. Berk, and A. Leung. Detachment of agglutinin-bonded red blood cells. I. Forces to rupture molecular-point attachments. Biophys. J. 59:838–848, 1991.
Evans, E., and K. Ritchie. Dynamic strength of molecular adhesion bonds. Biophys. J. 72:1541–1555, 1997.
Fath, K. R., C. J. Edgell, and K. Burridge. The distribution of integrins in focal contacts is determined by the substratum composition. J. Cell. Sci. 92:67–75, 1989.
Feltkamp, C. A., M. A. Pijnenburg, and E. Roos. Organization of talin and vinculin in adhesion plaques of wet-cleaved chicken embryo fibroblasts. J. Cell. Sci. 100:579–587, 1991.
Galbraith, C. G., and M. P. Sheetz. A micromachined device provides a new bend on fibroblast traction forces. Proc. Natl. Acad. Sci. USA 94:9114–9118, 1997.
Goodman, S. L., G. Risse, and K. von der Mark. The E8 subfragment of laminin promotes locomotion of myoblasts over extracellular matrix. J. Cell Biol. 109:799–809, 1989.
Hay, E. D. Interaction of migrating embryonic cells with extracellular matrix. Exp. Biol. Bed. 10:174–193, 1985.
Hendey, B., C. B. Klee, and F. R. Maxfield. Inhibition of neutrophil chemotaxis on vitronectin by inhibitors of calcineurin. Science 258:296–299, 1992.
Hughes, P. E., F. Diaz-Gonzalez, L. Leong, C. Wu, J. A. McDonald, S. Shattil, and M. H. Ginsberg. Breaking the integrin hinge: A defined structural constraint regulates integrin signaling. J. Biol. Chem. 271:6571–6574, 1996.
Huttenlocher, A., S. P. Palecek, Q. Lu, W. Zhang, R. L. Mellgren, D. A. Lauffenburger, M. H. Ginsberg, and A. F. Horwitz. Regulation of cell migration by the calciumdependent protease calpain. J. Biol. Chem. 272:32719–32722, 1997.
Hynes, R. O. Integrins: Versatility, modulation, and signaling in cell adhesion. Cell 69:11–25, 1992.
Inomata, M., M. Hayashi, Y. Ohno-Iwashita, S. Tsubuki, T. C. Saido, and S. Kawahima. Involvement of calpain in integrin-mediated signal transduction. Arch. Biochem. Biophys. 328:129–134, 1996.
Jacobson, K., A. Ishihara, and R. Inman. Lateral diffusion of proteins in membranes. Annu. Rev. Physiol. 49:163–175, 1987.
Jenkins, A. L., L. Nannizzi-Alaimo, D. Silver, J. R. Sellers, M. H. Ginsberg, D. A. Law, and D. R. Phillips. Tyrosine phosphorylation of the β3 cytoplasmic domain mediates integrin-cytoskeletal interactions. J. Biol. Chem. 273:13878–13885, 1998.
Kuntz, R. M., and W. M. Saltzman. Neutrophil motility in extracellular matrix gels: Mesh size and adhesion affect speed of migration. Biophys. J. 72:1472–1480, 1997.
Lane, R. D., D. M. Allan, and R. L. Mellgren. A comparison of the intracellular distribution of μ-calpain, m-calpain, and calpistatin in proliferating human A431 cells. Exp. Cell Res. 203:5–16, 1992.
Lauffenburger, D. A., and A. F. Horwitz. Cell migration: A physically integrated molecular process. Cell 84:359–369, 1996.
Lawson, M. A., and F. R. Maxfield. Ca2+-and calcineurin-dependent recycling of an integrin to the front of migrating neutrophils. Nature (London) 377:75–79, 1995.
Leckband, D., W. Mueller, F. J. Schmitt, and H. Ringsdorf. Molecular mechanisms determining the strength of receptor-mediated intermembrane adhesion. Biophys. J. 69:1162–1169, 1995.
Lee, J., M. Leonard, T. Oliver, A. Ishihara, and K. Jacobson. Traction forces generated by locomoting keratocytes. J. Cell Biol. 127:1957–1964, 1994.
Mandeville, J. T., and F. R. Maxfield. Effects of buffering intracellular free calcium on neutrophil migration through three-dimensional matrices. J. Cell Physiol. 171:168–178, 1997.
Marks, P. W., B. Hendey, and F. R. Maxfield. Attachment to fibronectin or vitronectin makes human neutrophil migration sensitive to alterations in cytosolic free calcium concentration. J. Cell Biol. 112:149–158, 1991.
Niggimann, B., K. Maaser, H. Lu, R. Kroczek, K. S. Zanker, and P. Friedl. Locomotory phenotypes of human tumor cell lines and T lymphocytes in a three-dimensional collagen lattice. Cancer Lett. (Shannon, Ireland) 118:173–180, 1997.
Palecek, S. P., A. Huttenlocher, A. F. Horwitz, and D. A. Lauffenburger. Physical and biochemical regulation of integrin release during rear detachment of migrating cells. J. Cell. Sci. 111:929–940, 1998.
Palecek, S. P., J. C. Loftus, M. H. Ginsberg, D. A. Lauffenburger, and A. F. Horwitz. Integrin-ligand binding properties govern cell migration speed through cell-substratum adhesiveness. Nature (London) 385:537–540, 1997.
Palecek, S. P., C. E. Schmidt, D. A. Lauffenburger, and A. F. Horwitz. Integrin dynamics on the tail region of migrating fibroblasts. J. Cell. Sci. 109:941–952, 1996.
Saterbak, A., and D. A. Lauffenburger. Adhesion mediated by bonds in series. Biotechnol. Prog. 12:682–699, 1996.
Schmidt, C., A. F. Horwitz, D. A. Lauffenburger, and M. P. Sheetz. Integrin/cytoskeleton interactions in migrating fibroblasts are dynamic, asymmetric, and regulated. J. Cell Biol. 123:977–991, 1993.
Schmidt, C., H. Pommerenke, F. Durr, B. Nebe, and J. Rychly. Mechanical stressing of integrin receptors induces enhanced tyrosine phosphorylation of cytoskeletally anchored proteins. J. Biol. Chem. 273:5081–5085, 1998.
Sczekan, M. M., and R. L. Juliano. Internalization of the fibronectin receptor is a constitutive process. J. Cell. Physiol. 142:575–580, 1990.
Spudich, J. A. S. Motor molecules in motion. Nature (London) 348:284–285, 1990.
Suzuki, K., T. C. Saido, and S. Hirai. Modulation of cellular signals by calpain. Ann. (N.Y.) Acad. Sci. 674:218–227, 1992.
Tranqui, L., and M. R. Block. Intracellular processing of talin occurs within focal adhesions. Exp. Cell Res. 217:149–156, 1995.
Wang, N., and D. E. Ingber. Control of cytoskeletal mechanics by extracellular matrix, cell shape, and mechanical tension. Biophys. J. 66:2181–2189, 1994.
Ward, M. D., and D. A. Hammer. A theoretical analysis for the effect of focal contact formation on cell-substrate attachment strength. Biophys. J. 64:936–959, 1993.
Ward, M. D., and D. A. Hammer. Focal contact assembly through cytoskeletal polymerization: Steady state analysis. J. Math. Biol. 32:677–704, 1994.
Wessels, D., H. Vawter-Hugart, J. Murray, and D. R. Soll. Three-dimensional dynamics of pseudopod formation and the regulation of turning during the motility cycle of Dictyostelium. Cell Motil. Cytoskeleton 27:1–12, 1994.
Wilson, A. K., G. Gorgas, W. D. Claypool, and P. de Lanerolle. An increase or decrease in myosin II phosphorylation inhibits macrophage motility. J. Cell Biol. 114:277–283, 1991.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Palecek, S.P., Horwitz, A.F. & Lauffenburger, D.A. Kinetic Model for Integrin-mediated Adhesion Release During Cell Migration. Annals of Biomedical Engineering 27, 219–235 (1999). https://doi.org/10.1114/1.176
Issue Date:
DOI: https://doi.org/10.1114/1.176