-
PDF
- Split View
-
Views
-
Cite
Cite
S. Porre, S. Heinonen, R. Mäntyjärvi, M. Rytkönen-Nissinen, O. Perola, J. Rautiainen, T. Virtanen, Psoriasin, a calcium-binding protein with chemotactic properties is present in the third trimester amniotic fluid, Molecular Human Reproduction, Volume 11, Issue 2, February 2005, Pages 87–92, https://doi.org/10.1093/molehr/gah141
Close - Share Icon Share
Abstract
Psoriasin is a small calcium-binding protein first found in psoriatic lesions and also up-regulated in other inflammatory skin diseases and cancer tissues. Psoriasin is also present in the fetal epithelial cells. Its biological function is unclear, but there is both in vitro and in vivo evidence for its chemokine-like activity. The aim of the present study was to find whether psoriasin could be found in the amniotic fluid and thus could have long-range immunobiological effects. Two recombinant psoriasins were prepared, one in Escherichia coli, the other one in Pichia pastoris. The former was used to produce a rabbit antiserum against psoriasin. Fractionation of full-term amniotic fluids with polyacrylamide gel electrophoresis (PAGE) and gel filtration associated with immunodetection with the antiserum were used to identify a protein compatible with the size of psoriasin. The identity of psoriasin was further verified by mass spectrometric analysis. Expression of psoriasin in cells of the amniotic membranes was detected with nested RT–PCR. Because of its chemokine-like activity, psoriasin present in the amniotic fluid might have consequential immunobiological effects during the fetal development.
Introduction
Psoriasin is a protein with a molecular mass of 11 kDa. It was first described as one of the proteins with an up-regulated expression in psoriatic skin lesions (Madsen et al., 1991). Sequencing of the corresponding cDNA revealed a potential calcium-binding site of the EF-hand type, and calcium binding was confirmed experimentally (Hoffmann et al., 1994). Sequence analyses further revealed that psoriasin belongs to the S100 family of calcium-binding proteins, and it has been entitled S100A7 (Prosite, http://www.expasy.ch/cgi-bin/get-prodoc-entry?PDOC00275). Psoriasin has a three-dimensional structure similar to other proteins of the same family (Brodersen et al., 1998), and under physiological conditions it forms homodimers. Psoriasin gene has been mapped to the chromosomal band 1q21 (Hardas et al., 1996), within a region containing genes of several calcium-binding proteins (Watson et al., 1998).
Later studies have shown that the expression of psoriasin is not limited to psoriasis. In situ hybridization on skin specimens detected psoriasin mRNA in a number of hyperproliferative and inflammatory skin diseases including atopic dermatitis (Algermissen et al., 1996). Furthermore, expression of psoriasin has been found in carcinomatous tissues from breast and bladder (Moog-Lutz et al., 1995; Celis et al., 1996). The expression of psoriasin in normal tissues is low or absent, but screening of a variety of fetal tissues by two-dimensional gel electrophoresis showed that psoriasin is expressed in the fetal ear, tongue and skin but not detectable in other tissues (Madsen et al., 1991).
The association between psoriasin expression and inflammation raised the question on the biological function of psoriasin in these conditions. The data are limited, but in leukocyte chemotaxis assays psoriasin was found to have chemokine-like properties, and the effect was selective for CD4+ T lymphocytes and neutrophils (Jinquan et al., 1996). Recently, psoriasin has been proposed to play an important role in the innate immunity (Foell et al., 2004).
Immunological phenomena associated with gravidity affect both the fetus and the mother. We have earlier described a bovine dander allergen which has a 63% amino acid identity with psoriasin (Rautiainen et al., 1995). This incited our interest in psoriasin, and the present studies show that psoriasin, in a dimeric form, is present in the amniotic fluid. Since psoriasin has been reported to exert chemokine activity and implicated in the innate immunity it may have important immunobiological effects during the fetal development.
Materials and methods
Amniotic fluid samples
Samples of amniotic fluid were obtained by amniocentesis of caesarean sections performed at term on healthy mothers. In pilot tests, samples were used which had been obtained by amniocentesis at the 10th–12th weeks of pregnancy for diagnostic purposes. The study was approved by the Ethics Committee of Kuopio University Hospital.
Production of recombinant proteins
DNA isolation, agarose gel electrophoresis and cloning procedures were performed using established techniques (Sambrook et al., 1989). All plasmid constructions were verified by DNA sequencing (A.L.F express DNA Sequencer, Amersham Pharmacia Biotech, Uppsala, Sweden).
Cloning vector for the expression of psoriasin in Escherichia coli was pONNI, a modified version of pGEX-2T (Amersham Pharmacia Biotech). pONNI has an additional glycin linker between the thrombin cleavage site and the multiple cloning site. This vector expresses sequences fused to the carboxyl terminus of the glutathione-S-transferase from Schistosoma japonicum (Smith and Johnson, 1988). The recombinant human psoriasin was PCR-amplified from a human psoriasin cDNA clone, which was kindly gifted by Peder Madsen, Institute of Medical Biochemistry, Aarhus Denmark. The generated plasmid DNA was transformed into chemically competent E. coli TG10B cells. Fusion protein expression, purification and thrombin treatment to release psoriasin from the fusion partner, was performed using GST Gene Fusion system according to the manufacturer's instructions (Amersham Pharmacia Biotech). In the following, this recombinant psoriasin is called rEcoPso.
For expression in Pichia pastoris, psoriasin was PCR-amplified from the pONNI-human psoriasin clone. The primers were designed to introduce SnaBI and EcoRI restriction sites at the 5′ and 3′ ends, respectively, to allow subcloning into the P. pastoris pPIC9 vector, which is used for secreted expression (Invitrogen, Groningen, The Netherlands). Moreover, the 3′ primer included a sequence of six histidines (His-tag). The pPIC9 human psoriasin plasmid DNA was then linearized with BglII and integrated into P. pastoris strain GS115 cells by electroporation using a gene pulser apparatus (Gene Pulser® II, Bio-Rad, Hercules, USA) according to the manufacturer's instructions from Invitrogen. The protein production was performed according to the manual of methods for the expression of recombinant proteins in P. pastoris (Invitrogen). The recombinant human psoriasin with His-tag was purified from the culture supernatant using HisTrap™ Kit (Amersham Pharmacia Biotech) followed by gel filtration in a Superdex 75 (Amersham Pharmacia Biotech). In the following, this recombinant psoriasin is called rPicPso.
Dot ELISA
A New Zealand white rabbit was immunized essentially as described previously (Ylönen et al., 1990) with recombinant psoriasin rEcoPso to obtain hyperimmune serum. Recombinant proteins or samples of amniotic fluid fractions were applied in volumes of 50–100 μl in phosphate buffered saline (PBS) to Hybond enhanced chemiluminescence (ECL) nitrocellulose membranes (Amersham Pharmacia Biotech) by using a dot blot apparatus (Schleicher & Schüll, Dessel, Germany). The membranes were blocked for 1 h at room temperature in PBS-Tween (0.1% v/v) containing 1% w/v gelatin. The membranes were then incubated in a shaker for 1 h at room temperature with the hyperimmune serum diluted appropriately in PBS-Tween-gelatin and subsequently washed three times with PBS-Tween. HRP-labelled goat anti-rabbit immunoglobulin (DAKO, Glostrup, Denmark) diluted 1:20 000 was used as the secondary antibody. After incubation for 45 min on a shaker at room temperature and washings, ECL+reagents (Amersham Pharmacia Biotech) were applied on the membranes according to the manufacturer's instructions. After the ECL treatment, the membranes were exposed to Hyperfilm ECL (Amersham Pharmacia Biotech).
For quantification, the film was digitized and the analysis was performed using the public domain NIH Image program (http://rsb.info.nih.gov/nih-image).
PAGE and immunoblotting
Sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) with the disscontinuous buffer system of Laemmli was run at 50 mA using 15% (bis)Tris gels. Mini-Protean II electrophoresis cell and Mini trans-blot cell (Bio-Rad Laboratories, Hercules, CA) were used for electrophoresis and protein transfer, respectively. Samples were heated at 100°C for 5 min. Gels were stained with Coomassie Brilliant Blue R-250 (Bio-Rad, Sundbyberg, Sweden). Electrophoretical transfer of the protein bands to Hybond ECL membranes (Amersham Pharmacia Biotech) was conducted at 100 V for 40 min. After transfer, the membrane was blocked in PBS-Tween-gelatin and immunodetection of the proteins was carried out in the same way as in dot enzyme linked immunosorbent assay (ELISA).
Gel filtration
Samples of amniotic fluids were fractionated by gel filtration in a Superdex 75 10/30 column attached to the Äkta Purifier chromatography system (Amersham Pharmacia Biotech). PBS was used as the eluent, and 0.5 ml fractions were collected. Bovine serum albumin, beta-lactoglobulin and lysozyme were used as molecular weight standards.
Immunoaffinity purification
The immunoglobulin G (IgG) fraction of the hyper-immune serum against rEcoPso was purified in a HiTrap Protein G Hp column (Amersham Biosciences, Uppsala, Sweden) and the purified IgG fraction was then coupled to a HiTrap NHS-activated HP column (Amersham Biosciences) as recommended in the manufacturer's protocols. This immmunoaffinity column was used to purify the fraction of interest from the amniotic fluid samples according to the manufacturer's instructions. After immunoaffinity purification, the collected fractions were first concentrated to 10-fold with Microsep™ microconcentrators (Pall Life Sciences, MI, USA) and then 10-fold with a Nanosep® 3K Centrifugal device (Pall Life Sciences).
Western blot inhibition
For the inhibition experiment, SDS-PAGE and western blot transfer was done as described above. The samples were 5 ng of rPicPso and 20 μl of concentrated immunoaffinity purified amniotic fluid. Equal volumes of the inhibiting proteins at 2 μg/ml and antiserum (diluted to 1:10 000) were mixed, incubated at room temperature for 1 h and applied on western blot membranes. After another incubation for 1 h at room temperature, the membranes were processed as in dot ELISA.
Mass spectrometric analysis
Coomassie Brilliant Blue stained protein bands of interest were cut out of the SDS-PAGE gel and ‘in-gel’ digested essentially as described by Shevchenko et al. (1996). Proteins were reduced with dithiothreitol and alkylated with iodoacetamide before digestion with trypsin (Sequencing Grade Modified Trypsin, V5111, Promega). The recovered peptides were desalted and subjected to matrix-assisted laser desorption/ionization-time of flight (MALDI-TOF) mass spectrometric analysis. MALDI-TOF mass spectra for mass fingerprinting and MALDI-TOF/TOF mass spectra for identification by fragment ion analysis were acquired using an Ultraflex TOF/TOF instrument (Bruker-Daltonik GmbH, Bremen, Germany). Protein identification from the generated data was performed using Mascot® Peptide Mass Fingerprint and MS/MS Ion Search programs (http://www.matrixscience.com).
RT–PCR
Amniotic membranes were obtained from the same-term pregnancies as the late amniotic fluid samples. The membrane was detached from the underlying tissue, and the RNAgents® Total RNA Isolation System (Promega, Madison, WI, USA) was used to prepare total RNA by following the manufacturer's instructions. The cDNA was synthesized using random hexamers and M-MLV reverse transcriptase RNase H Minus (Promega) as described earlier (Rautiainen et al., 1998).
The amplification of the human psoriasin cDNA was performed with two sets of primers. The first set comprised the sense primer GCTGAGAGGTCCATAATAGG (5′U65) and the antisense primer GCTCTGCTTGTGGTAGTCTG (3′L300), and the nested set was the sense primer GCCTGCTGACGATGATGAAGG (5′U141) and the antisense primer GCAAGGACAGAAACTCAGAA (3′L265). In the nested PCR, one tenth of the end product of the first PCR was used for amplification.
The amplification product of the nested PCR was fractionated on a 1.8% agarose gel followed by purification using an Ultrafree-DA Centrifugal Filter Device (Millipore, Bedford, MA, USA). The DNA sequence of the amplified fragment was verified with the A.L.F. express DNA Sequencer using an Auto Read Sequencing Kit (Amersham Pharmacia Biotech).
Results
Characterization of rabbit antiserum against psoriasin
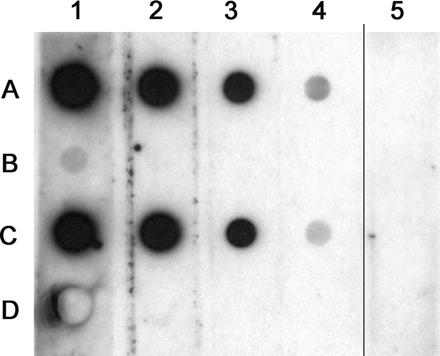
Recombinant psoriasin produced in E. coli (rEcoPso) was used to produce hyperimmune serum against psoriasin. The reactivity of the serum was tested by dot ELISA against different antigens (Figure 1). When 10 ng/spot of each protein was used on the membrane, the serum showed a positive reaction at a dilution of 10−6 both against the psoriasin preparation used for immunization as well as against recombinant psoriasin produced in the Pichia yeast (rPicPso). The specificity of the reaction was verified with the extract of E. coli (host of the plasmid used for production of rEcoPso) and with an irrelevant recombinant protein (Can f 1, the major allergen of dog dander containing a C-terminal His-tag in the same way as rPicPso) produced in Pichia. Pre-immunization serum at the dilution of 10−3 was negative against all antigens tested.
Immunodetection of psoriasin in amniotic fluid
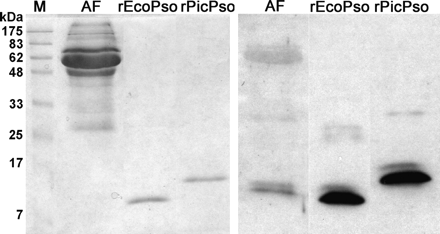
When samples of amniotic fluids were analysed with SDS-PAGE, no visible band corresponding to the expected monomer size of psoriasin was seen in gels stained by Coomassie Brilliant Blue (Figure 2, left panel). However, in immunoblotting, a clear signal in the form of a double band was observed suggesting the presence of psoriasin (Figure 2, right panel). The larger size of rPicPso as compared to rEcoPso and psoriasin band in the amniotic fluid is caused by amino acids of the His-tag in the rPicPso.
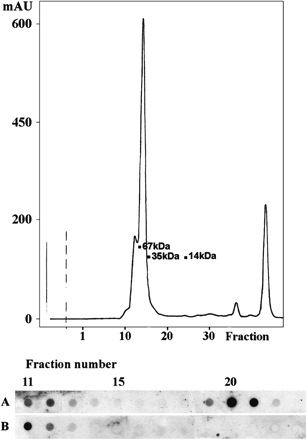
Preliminary experiments showed that samples of amniotic fluids applied on to nitrocellulose membranes gave a strong reaction of non-specific binding in dot ELISA with the pre-immune rabbit serum. Fractionation of the samples by gel filtration was applied in an attempt to separate the material causing non-specific binding and psoriasin. Figure 3 shows a representative result. Non-specific binding was found in association with high molecular weight material, adjacent to the highest peak, probably albumin. The UV280 absorbance curve did not show a peak where dimeric or monomeric forms of psoriasin should have been located (Figure 3, upper panel). However, dot ELISA revealed the presence of material recognized specifically by the immune serum (Figure 3, lower panel). A comparison with the molecular weight standards showed that the positive fractions corresponded to a molecular weight of 20–22 kDa in agreement with the size of psoriasin dimers. Samples of four different amniotic fluids analysed with the same protocol yielded identical results. Similar analyses of samples from early pregnancy remained negative for psoriasin (results not shown).
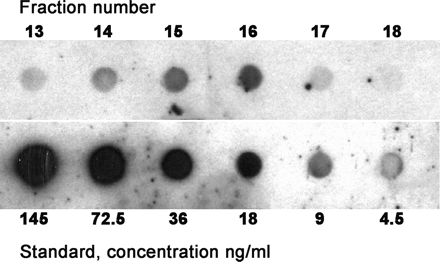
To estimate the concentration of psoriasin in amniotic fluids, dot ELISA reactivity of gel filtration fractions was compared with those of a recombinant psoriasin standard (Figure 4). Densities of the dots were measured as indicated in the Methods, and 2-fold dilutions of the rEcoPso were used to obtain a standard curve. Concentrations in the fractions were calculated and converted to concentrations in the original amniotic fluids. Three separate measurements of each two amniotic fluids resulted in an average concentration of 31±16 ng/ml (SD).
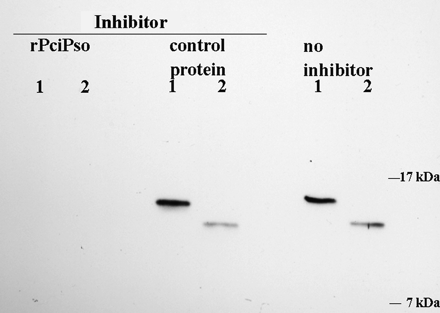
Since the concentration of the psoriasin in full-term amniotic fluid was quite low, immunoaffinity purification was performed to isolate and concentrate the fraction of interest. The western blot inhibition experiment was performed to verify the identity of the fraction as psoriasin. The assay showed that the recombinant psoriasin (rPicPso) prevented the binding of antiserum to the ca. 10 kDa protein at concentrations similar to those inhibiting the binding to rPicPso. The control recombinant protein caused no inhibition (Figure 5).
Mass spectrometric analysis of psoriasin
The immunoaffinity purified sample gave several protein bands in SDS-PAGE; two at the position approximately 10 kDa compatible with the size of psoriasin (data not shown). For identification of psoriasin, the two protein bands stained with Coomassie Brilliant Blue were subjected to ‘in gel’ digestion and the generated peptides analysed by MALDI-TOF/TOF mass spectrometry. The lower of the two bands was identified to be human psoriasin. In peptide mass fingerprint analysis, the masses obtained from the digested lower protein band gave a score of 62 to human psoriasin (2PSR, NCBI database) with a peptide sequence coverage of 61% unambiguously identifying the protein band as human psoriasin. To further confirm this result, one peptide from the mass fingerprint (obtained protonated mass 1569.76, suggested sequence ENFPNFLSACDKK) was subjected to fragment ion analysis by MALDI-TOF/TOF mass spectrometry. MS/MS Ion Search analysis with the obtained fragment masses gave the best score (Ion Score 34) to human psoriasin peptide ENFPNFLSACDKK with a calculated monoisotopic mass of 1568.73, which further confirmed the result from mass fingerprint analysis. The analysis of the upper band by mass spectrometry showed that it contained both human haemoglobin β-chain and the same psoriasin peptides as the lower band (data not shown).
Detection of psoriasin mRNA in the amniotic membranes
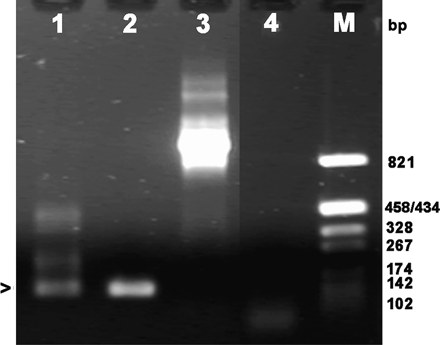
Psoriasin is expressed in the fetal squamous epithelium, especially in the skin (Madsen et al., 1991). Since the amniotic membranes have a large surface area consisting of fetal cells, we wanted to find out whether they could also be a source of psoriasin present in the amniotic fluid. Nested RT–PCR was used to detect the expression and the results are shown in Figure 6. When the amplicon corresponding to the expected size (arrowhead in Figure 6) was isolated and sequenced, it proved to have a sequence identical to that of psoriasin.
Discussion
Our results show that psoriasin is present at low concentrations in the full-term amniotic fluid. Although psoriasin does not contain a signal sequence for secretion (Madsen et al., 1991), the presence of extracellular psoriasin in the amniotic fluid was not surprising since psoriasin has been found to be released in culture fluids of psoriatic fibroblasts (Madsen et al., 1991), in bovine amniotic fluid (Hitomi et al., 1996) and in the urine of bladder cancer patients (Celis et al., 1996).
Because of our early observation of the non-specific binding of IgG on amniotic fluid in immunoblotting, special attention was paid to verify the identity of the protein detected by anti-psoriasin antiserum. The use of recombinant psoriasins produced in two different hosts with appropriate control proteins in the immunoblotting experiments suggested that psoriasin was indeed present in the amniotic fluid. Theoretically, it was possible that our antiserum could recognize, in addition to psoriasin, other members of the S100 family of Ca++ binding proteins. However, as there are several isoforms of psoriasin (Madsen et al., 1991; Celis et al., 1996), it seems reasonable that the two bands from the amniotic fluid in the western blot (Figure 2) represent two variant forms of psoriasin. The mass spectometric analysis of the immunoaffinity-purified sample of amniotic fluid confirmed that the lower band was human psoriasin. Moreover, the upper band contained peptides compatible with the presence of psoriasin. The finding of psoriasin expression in the cells of amniotic membranes, detected by RT–PCR, identified another potential source of secreted psoriasin. The larger amplicons observed after RT–PCR are compatible in size with products resulting from the cross-use of primers in nested PCR.
As predicted from the amino acid sequence, psoriasin has been found to bind Ca++ with the C-terminal EF-hand (Brodersen et al., 1999). Although the function of psoriasin in keratinocytes remains unknown, its expression seems to be associated with proliferation and abnormal differentiation (Algermissen et al., 1996; Watson et al., 1998). As the name indicates, the first association detected between a clinical condition and up-regulation of psoriasin expression was in psoriatic skin lesions (Madsen et al., 1991). Soon after it became apparent that the up-regulation was not disease-specific, but was also observed in other skin lesions characterized by inflammation and/or proliferation of epidermal keratinocytes (Algermissen et al., 1996). Another dimension was added to the significance of psoriasin as a biomarker when the association between psoriasin and neoplastic growth was observed (Celis et al., 1996; Leygue et al., 1996; Al-Haddad et al., 1999). It was suggested by Celis et al. (1996) that psoriasin present in the urine of patients with bladder squamous cell carcinoma might serve as a follow-up marker. In breast cancer, high levels of psoriasin expression were associated with the absence of estrogen and progesterone receptors (Al-Haddad et al., 1999).
The women in our study were undergoing elective caesarean deliveries after normal term pregnancies but were not yet in labour. This is likely to preclude intrauterine infection, pre-term birth and other complications as biasing our results. We did not detect psoriasin in the early samples of amniotic fluid suggesting that it is temporally up-regulated or accumulated during gestation. Since psoriasin has been observed to exhibit antimicrobial activity (Gläser et al., 2001) the temporal up-regulation or accumulation of psoriasin could be related to the role of S100 members of Ca-binding proteins in the innate immune defence (Foell et al., 2004). Psoriasin has been shown to be present in vernix caseosa together with other antimicrobial substances (Yoshio et al., 2003).
The presence of psoriasin in the fetal keratinocytes and in the amniotic fluid probably reflects the proliferative state of keratinocytes. However, the possibility that psoriasin might have immunobiological effects during the fetal development should be considered. In vitro experiments have revealed that psoriasin shows chemokine-like activity, especially for Th (CD4+) cells and neutrophils (Jinquan et al., 1996). Comparisons with the reactivity of known amounts of recombinant psoriasin in dot ELISA experiments led to the estimations of concentrations of about 10−11 mol/ml in the amniotic fluid. This concentration is higher than that producing chemotactic effects in vitro (Jinquan et al., 1996). Observations compatible with the chemotactic activity of psoriasin in vivo have also been made (Di Nuzzo et al., 2000). UVB exposure of skin induced expression of psoriasin in association with the recruitment of CD4+ T cells. Receptors for psoriasin have not been identified. However, it is known that the presence of different chemokine receptors depends, to a certain degree, on the subset of CD4+ cells (Cosmi et al., 2001). Therefore, it is conceivable that the chemoattractive effects of psoriasin might have highly specialized features. These observations warrant further studies on the immunobiological effects of psoriasin during fetal development.
Reactivity of the rabbit antiserum against recombinant psoriasins. Row A: recombinant psoriasin used for immunization (rEcoPso); Row B: crude E. coli extract (control antigen for rEcoPso); Row C: recombinant psoriasin produced in P. pastoris (rPicPso); Row D: control recombinant protein produced in Pichia. All antigens at a concentration of 10 ng of protein per dot. Columns 1–4: series of 10-fold dilutions of the antiserum from 10−3 to 10−6. Column 5: pre-immune serum at 10−3.
PAGE analysis of full-term amniotic fluid. Left panel: Coomassie Brilliant Blue staining; Right panel: immunoblot with anti-psoriasin immune serum (a dilution of 10−4). M: molecular weight marker; AF: amniotic fluid; rEcoPso and rPicPso: recombinant psoriasins produced in E. coli and P. pastoris, respectively.
Sephadex gel filtration of full-term amniotic fluid. Upper panel: UV280 absorbance curve of the eluate is shown with molecular weight markers. Lower panel: dot ELISA test of fractions 11–23 with the anti-psoriasin antiserum (Row A) and with the pre-immunization serum (Row B) at dilutions of 10−4.
Quantification of psoriasin in amniotic fluid. Dot ELISA was performed on fractions from a Sephadex gel filtration together with a series of 2-fold dilutions of recombinant psoriasin (rEcoPso). Density readings of the dots were used to calculate the concentrations. The antiserum was used at 10−4.
Identification of psoriasin in amniotic fluid with western blot inhibition test. Binding of rabbit antiserum (a dilution of 10−4) on immunoaffinity purified fraction of amniotic fluid was inhibited by recombinant psoriasin (rPicPso), but not by a control recombinant protein (Equ c 1, the major allergen of horse dander). Lane 1: 5 ng rPciPso (the size of protein is ca. 13 kDa); Lane 2: 20 μl of concentrated immunoaffinity purified amniotic fluid (the size of immunoreactive band is ca. 10 kDa).
Expression of psoriasin in amniotic membranes. Products of nested RT–PCR resolved by agarose gel electrophoresis. Lane 1: amplicon from an extract of amniotic membranes; the band with the size expected of psoriasin marked with an arrowhead; Lane 2: positive control, rEcoPso plasmid as template; Lane 3: control to show the absence of amplification from genomic DNA; Lane 4: negative control to exclude template contamination of PCR; Lane M: size markers, base pairs (bp).
This work was supported by the Academy of Finland (Contracts 2939, 48657, and 205871), by the Paulo Foundation and by the Kuopio University Hospital (Project 5021605). The skilful technical assistance of Ms Mirja Saarelainen is gratefully acknowledged (Department of Clinical Microbiology, University of Kuopio). Ms Gunilla Rönnholm (Protein Chemistry Research Group and Core Facility, University of Helsinki, Finland) is acknowledged for the ‘in-gel’ digestion and mass spectrometric identification of psoriasin.
References
Algermissen B, Sitzmann J, LeMotte P and Czarnetzki B (
Al-Haddad S, Zhang Z, Leygue E, Snell L, Huang A, Niu Y, Hiller-Hitchcock T, Hole K, Murphy LC and Watson PH (
Brodersen DE, Etzerodt M, Madsen P, Celis JE, Thogersen HC, Nyborg J and Kjeldgaard M (
Brodersen DE, Nyborg J and Kjeldgaard M (
Celis JE, Rasmussen HH, Vorum H, Madsen P, Honore B, Wolf H and Orntoft TF (
Cosmi L, Annunziato F, Maggi E, Romagnani S and Manetti R (
Di Nuzzo S, Sylva-Steenland RM, Koomen CW, de Rie MA, Das PK, Bos JD and Teunissen MB (
Foell D, Frosch M, Sorg C and Roth J (
Gläser R, Harder J, Bartels J, Christophers E and Schröder J-M (
Hardas BD, Zhao X, Zhang J, Longqing X, Stoll S and Elder JT (
Hitomi J, Yamaguchi K, Kikuchi Y, Kimura T, Maruyama K and Nagasaki K (
Hoffmann HJ, Olsen E, Etzerodt M, Madsen P, Thogersen HC, Kruse T and Celis JE (
Jinquan T, Vorum H, Larsen CG, Madsen P, Rasmussen HH, Gesser B, Etzerodt M, Honore B, Celis JE and Thestrup-Pedersen K (
Leygue E, Snell L, Hiller T, Dotzlaw H, Hole K, Murphy LC and Watson PH (
Madsen P, Rasmussen HH, Leffers H, Honore B, Dejgaard K, Olsen E, Kiil J, Walbum E, Andersen AH, Basse B et al. (
Moog-Lutz C, Bouillet P, Regnier CH, Tomasetto C, Mattei MG, Chenard MP, Anglard P, Rio MC and Basset P (
Rautiainen J, Rytkönen M, Parkkinen S, Pentikäinen J, Linnala-Kankkunen A, Virtanen T, Pelkonen J and Mäntyjärvi R (
Rautiainen J, Rytkönen M, Syrjänen K, Pentikäinen J, Zeiler T, Virtanen T and Mäntyjärvi R (
Sambrook J, Fritsch EF and Maniatis T (
Shevchenko A, Wilm M, Vorm O and Mann M (
Smith DB and Johnson KS (
Ylönen J, Nuutinen J, Rautiainen M, Ruoppi P, Mäntyjärvi R and Virtanen T (
Author notes
1Department of Clinical Microbiology, University of Kuopio, FIN-70211, and 2Department of Obstetrics and Gynaecology, Kuopio University Hospital, FIN-70210, Kuopio, Finland