-
PDF
- Split View
-
Views
-
Cite
Cite
Anat Bahat, Michael Eisenbach, Ilan Tur-Kaspa, Periovulatory increase in temperature difference within the rabbit oviduct, Human Reproduction, Volume 20, Issue 8, August 2005, Pages 2118–2121, https://doi.org/10.1093/humrep/dei006
Close - Share Icon Share
Abstract
BACKGROUND: Earlier studies demonstrated a small temperature difference between the sperm storage and fertilization sites within the oviducts of rabbits and pigs. Our aim was to reveal the time dependence of this temperature difference relative to ovulation, and to determine how this difference is generated—by temperature elevation at one of these sites or by temperature decrease at the other site. METHODS: The temperature at the sperm storage site (at the isthmus near the uterotubal junction) and at the fertilization site (the isthmic–ampullary junction) of rabbit oviducts were measured before, during, and after ovulation by two probes, connected to digital thermometers. Rectal temperature was constantly measured and served as a control for body temperature. RESULTS: The temperature difference between the fertilization site and the storage site was 0.8±0.2°C before ovulation. This difference increased at ovulation, reaching 1.6±0.1°C after ovulation (P<0.03). This increased difference was mainly due to temperature decrease in the sperm storage site. CONCLUSION: The temperature-difference increase within the rabbit oviduct is generated at ovulation by a reduced temperature at the sperm storage site. This temperature gradient may play a role in mammalian reproduction via sperm thermotaxis.
Introduction
Sperm transport in the female genital tract, well studied in a number of mammals, is a highly controlled and synchronized process [for a review, see Barratt and Cooke, (1991)]. After passing the uterus, the spermatozoa reach the uterotubal junction and continue into the isthmic part of the oviduct, where they are arrested by tight association with the epithelial surface (Suarez, 2002). The isthmus is, therefore, considered as the sperm storage site. The spermatozoa are released from the storage site when they become capacitated (Suarez, 2002) [for a review on capacitation, see Jaiswal and Eisenbach, (2002)]. In the rabbit, where ovulation is induced by mating (Ramirez and Soufi, 1994), the capacitation process is synchronized with ovulation (Giojalas et al., 2004). Thereafter, the spermatozoa reach the oocyte at the fertilization site [the isthmic–ampullary junction in the rabbit (Harper, 1982)] within minutes (Overstreet, 1983). It has been argued that, without some guidance mechanism, the prospects of the released spermatozoa meeting the egg at the fertilization site by coincidence is negligible (Eisenbach and Tur-Kaspa, 1999 and references cited therein).
Guidance in the form of sperm chemotaxis to substances secreted from the egg or its surrounding cells, a process demonstrated to occur in mammals (Eisenbach, 2004), is likely restricted to the immediate surroundings of the egg. The reason is peristaltic movements of the oviduct (Battalia and Yanagimachi, 1979), which probably prevent the formation of a chemical gradient in the oviduct over a long distance from the egg (Eisenbach, 1999). On the basis of findings that, at least in rabbits (David et al., 1972) and pigs (Hunter and Nichol, 1986), there is a temperature difference of 2°C and 0.7°C, respectively, within the oviduct, Bahat et al., (2003) recently demonstrated that rabbit and human spermatozoa have the capacity to sense such a temperature difference and respond to it by thermotaxis, i.e. by swimming from the cooler to the warmer temperature.
These findings raise two major questions, which are highly relevant for evaluating the function of thermotaxis in reproduction: Is the temperature difference time-dependent and synchronized with ovulation? If so, how is the temperature difference established—by temperature rise at the fertilization site or by temperature decrease at the storage site? Here we address these questions.
Materials and methods
Experimental animals
Thirteen sexually mature female rabbits (strain New Zealand white) were investigated in this study. The animals were individually housed in standard cages in a room maintained at 20±1°C and 60±10% relative humidity with a 12 hours light/dark cycle. Ten of the animals were mated once with a fertile male at different times before surgery. The Weizmann Institute's ethical committee for animal experiments approved the study protocol.
Pre-operative procedures
Primary analgesia of the animals was induced with 1 ml of Diazepam (5 mg/ml), given intravenously. Anesthesia was achieved by an intramuscularly injection of ketamine hydrochloride (35 mg/kg) and xylazine 2% (5 mg/kg). The animals were placed on a wrapped operating table and an abdominal laparotomy was performed by an experienced reproductive surgeon. The reproductive tract was exposed and the ovaries were examined carefully to determine whether ovulation occurred.
Temperature measurements
Temperatures within the lumen of the oviduct and within the rectum were measured by using two thermistor probes (Technomad Ltd., Israel). Each probe (stainless steel; 0.5 mm in diameter and 30 cm in length) was connected to a separate digital thermometer with a temperature accuracy of ±0.2°C. Before each set of surgeries, the thermometers were adjusted by putting both probes together in the same 37°C water bath. While performing the experiment, one probe, placed constantly within the animal rectum (2–3 cm deep), was used as a reference for the temperature loss due to anesthesia and the open abdomen. [The normal rectal temperature in healthy rabbits is 38.5–40.0°C (Harkness and Wagner, 1989).] The second probe served for alternating temperature measurements within the isthmus (near the uterotubal junction) and within the isthmic-ampullary junction. The probe sited at the isthmus was inserted via a section in the vagina wall, through the uterus, until its tip reached the beginning of the oviduct. The probe sited at the isthmic-ampullary junction was inserted via the fimbria to a depth of 3–4 cm. The temperatures in each region were recorded 2–4 times. The temperature measurements in each animal took no more than 15 min in total. At the end of the experiments the animals were sacrificed by an intravenous injection of sodium pentobarbital (133 mg/kg).
Statistics
The significance of the differences between different time points was determined by the ANOVA test or nonparametric (Mann–Whitney) test, as indicated, using InStat 3 software package (Graph Pad Software, San Diego, CA).
Results
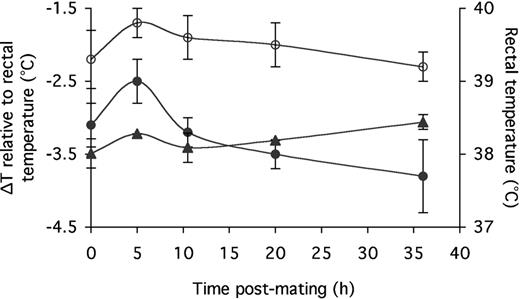
To find out whether the temperatures at the storage and fertilization sites are time- and ovulation-dependent, we measured, as described in Materials and Methods, the temperature at these sites in 13 female rabbits (26 oviducts) as follows: 3 non-mated rabbits; 2 mated, pre-ovulatory rabbits (5 h post-mating); 4 mated, peri-ovulatory rabbits (10.5–11.0 h post-mating); 2 mated, post-ovulatory rabbits close to the time of ovulation (20 h post-mating); and 2 mated, post-ovulatory rabbits far from the time of ovulation (36 h post-mating). Table I depicts the timetable of the temperature measurements performed before, during, and after ovulation. The temperatures measured at the sperm storage and fertilization sites (closed and open circles in Figure 1, respectively) are presented relative to the rectal temperatures (triangles in Figure 1). In spite of the temperature variations between different oviducts, it appears that, following the temperature rise measured 5 h post-mating, the rectal temperature was essentially constant for the next 15 h [the differences between the rectal temperatures at the various time points were not statistically significant (P=0.41 according to ANOVA test)], the temperature at the fertilization site gradually and slightly decreased, and the temperature at the storage site decreased more steeply (Figure 1). Thus, the difference between the temperature at the storage site before and after ovulation (measured at the last time point before ovulation and the first time point after ovulation, 5 and 20 h post-mating, respectively) was significant (P=0.029 according to Mann–Whitney test), whereas the temperature values at the fertilization site at these time points were not significantly different (P=0.68). The consequence of these temperature changes was an increased difference between the temperatures at the fertilization and storage sites, starting at the time of ovulation (Figure 2). The difference between these sites between the last time point before ovulation and the first time point after ovulation was significant (P=0.029 according to Mann–Whitney test). All the experimental points could well be fitted into a sigmoidal curve (R2=0.99; Figure 2), suggesting that the difference between the fertilization and storage sites increased from 0.8±0.2°C before ovulation to 1.6±0.1°C after ovulation. This temperature difference was maintained over a distance of 9.8±0.7 cm (mean±SEM; four oviducts)—the distance that we measured between the uterotubal and isthmic-ampullary junctions in the rabbit oviduct.
To determine whether the temperature difference is maintained for a long period after ovulation, we measured the temperatures 36 h post-mating (about 25 h after ovulation). The results were not conclusive because two oviducts of one rabbit maintained a temperature difference, whereas the temperature difference decreased in the oviducts of another rabbit (Figures 1 and 2).
Discussion
Earlier studies demonstrated the occurrence of a temperature difference within the oviducts of ovulated rabbits [2°C (David et al., 1972)] and pigs [0.7°C (Hunter and Nichol, 1986)]. Our current study endorses the finding that, in the rabbit, the fertilization site is warmer than the sperm storage site. It furthers shows that this temperature difference is time-dependent and increases at ovulation due to a temperature decrease at the storage site. The temperature difference increases significantly from 0.8±0.2°C before ovulation to 1.6±0.1°C after ovulation. Such a temperature difference may have an important role in sperm guidance to the egg, because capacitated rabbit and human spermatozoa have the ability to sense a small temperature difference and to respond to it by thermotaxis toward the warmer temperature (Bahat et al., 2003).
We cannot deduce from our experiments the duration of the temperature difference. In the rabbit it lasts for at least 10 h (Figure 2), more than enough for attracting the capacitated spermatozoa by thermotaxis. This is because, in the rabbit—an induced ovulator in which the timing of the capacitated state is synchronized with egg availability in the female genital tract—spermatozoa are capacitated during a time window of ∼8 h, ranging from ∼12 h to ∼20 h post-mating (Giojalas et al., 2004).
Our findings indicate that although the temperature difference between the storage and fertilization sites increases around the time of ovulation, a small temperature difference already exists prior to ovulation. The spermatozoa probably do not utilize this small temperature difference prior to ovulation for thermotaxis because, at this pre-ovulatory time, the spermatozoa are not yet capacitated (Giojalas et al., 2004) and, consequently, are not yet thermotactically responsive (Bahat et al., 2003).
Although human spermatozoa are thermotactically responsive too (Bahat et al., 2003), it is not known whether or not an ovulation-dependent temperature gradient exists in humans. Assuming—on the basis of the two mammals in which the temperature was measured and on the basis of the study of Cicinelli et al. (2004) demonstrating temperature differences in the female genital tract of humans before and after ovulation—that it does, the time window in humans may be much longer. This is because in humans, where no linkage between ovulation and sperm capacitation exists, capacitated spermatozoa are available for extended time periods (Giojalas et al., 2004).
An intriguing question is how the time-dependent temperature difference is established between two sites, ∼10 cm apart, within the same abdominal organ. On the basis of current knowledge, at least three mechanisms appear plausible.
One potential mechanism for the generation of a temperature difference within the oviduct is a biochemically generated endothermic process such as hydration. Such a mechanism was originally proposed by Luck et al. (2001) to explain the lower temperature in human (Grinsted et al., 1985), pig (Hunter et al., 1997), and rabbit (Grinsted et al., 1980) follicles relative to the adjacent tissue. Luck et al. (2001) proposed that the follicle growth involves continuous secretion of a large molecule(s) [e.g. mucopolysaccharide (Zachariae, 1959)], whose hydration results in the endothermic cooling of the fluid. We propose that such a mechanism may also apply to the cooling of the storage site relative to the fertilization site. The basis for this possibility is the finding of Jansen and Bajpai (1982) that in the rabbit, acid mucus glycoprotein—a highly hydrated macromolecule—is secreted predominantly from the isthmus, to a lesser extent from the isthmic–ampullary junction, and not at all from the ampulla, and the finding of Gandolfi (1995) that this glycoprotein secretion is controlled by the level of circulating steroid hormones. Thus, it is possible that acid mucus glycoprotein or a similar hydratable molecule, the secretion of which is hormone-dependent and which is mainly confined to the sperm storage site in the isthmus, causes the oviductal fluid in this specific region to undergo hydration with a resultant temperature decrease.
Another potential mechanism is counter-current heat exchange, proposed by David et al. (1972) and Hunter and Nichol (1986) to explain their findings of temperature differences within the oviduct. The vessel that supplies blood to the storage site is different from the blood vessel to the fertilization site, and both of them are aligned close to the ovarian vein, flowing blood in the opposite direction (Leese, 1988). According to this possibility, cold blood from the ovary cools the blood that enters the storage site by counter-current heat exchange, in a manner similar to the heat transfer known to occur between the testicular veins and artery (Glad Sorensen et al., 1991).
The recent finding that, in women, the source of blood supply to the storage site appears to change at ovulation (Cicinelli et al., 2004) may provide another mechanism. Thus, prior to ovulation, the blood supply to the storage site appears to be mainly carried out by the ovarian artery. However, subsequent to ovulation, the supply is mainly by the uterine artery. It is, therefore, possible that the blood in this artery, thought to be cooled by counter-current heat exchange with blood flowing from the vagina (Cicinelli et al., 2004), is responsible for the temperature drop found at ovulation. It could also account for our observation that the basal temperature at the storage site is lower than that at the fertilization site.
The temperature difference within the rabbit oviduct, demonstrated in this study to be generated at ovulation by lowering the temperature at the sperm storage site, may play an important role in mammalian reproduction. Sperm thermotaxis from the cooler storage site to the warmer fertilization site should be further explored as a natural mechanism for sperm selection in both in vivo and in vitro fertilization.
The temperatures at the isthmus near the uterotubal junction (the storage site; closed circles) and at the isthmic-ampullary junction (the fertilization site; open circles), relative to the rectal temperature (triangles). The values shown at t=0 were obtained with non-mated rabbits.
Time-dependent temperature differences between the storage and fertilization sites within the rabbit oviduct. The squares stand for the experimental points (mean±SEM; 4–8 oviducts for each point). The connecting line is a theoretical sigmoidal-curve fitting (R2=0.99), obtained according to the Boltzmann equation (Origin 6.1 software, OriginLab):
Temperatures measured in oviducts of non-mated (t=0), pre-ovulatory (5 h post-mating), peri-ovulatory (10.5 h post-mating), and post-ovulatory (20 and 36 h post-mating) rabbits
| Time post-mating (h) . | No. of oviducts tested . | Ovulation . | Temperature (°C) measured in the . | . | . | ||
|---|---|---|---|---|---|---|---|
| . | . | . | Rectum . | Sperm storage site . | Fertilization site . | ||
| 0 | 6 | No | 38.0±0.04 | 35.1±0.2 | 35.9±0.2 | ||
| 5 | 4 | No | 38.3±0.03 | 35.8±0.2 | 36.6±0.1 | ||
| 10.5 | 8 | Yes | 38.2±0.05 | 34.9±0.2 | 36.2±0.3 | ||
| 20 | 4 | Yes | 38.2±0.02 | 34.7±0.1 | 36.3±0.2 | ||
| 36 | 4 | Yes | 38.4±0.02 | 34.8±0.3 | 36.1±0.1 | ||
| Time post-mating (h) . | No. of oviducts tested . | Ovulation . | Temperature (°C) measured in the . | . | . | ||
|---|---|---|---|---|---|---|---|
| . | . | . | Rectum . | Sperm storage site . | Fertilization site . | ||
| 0 | 6 | No | 38.0±0.04 | 35.1±0.2 | 35.9±0.2 | ||
| 5 | 4 | No | 38.3±0.03 | 35.8±0.2 | 36.6±0.1 | ||
| 10.5 | 8 | Yes | 38.2±0.05 | 34.9±0.2 | 36.2±0.3 | ||
| 20 | 4 | Yes | 38.2±0.02 | 34.7±0.1 | 36.3±0.2 | ||
| 36 | 4 | Yes | 38.4±0.02 | 34.8±0.3 | 36.1±0.1 | ||
The results shown are the mean ± SEM.
Temperatures measured in oviducts of non-mated (t=0), pre-ovulatory (5 h post-mating), peri-ovulatory (10.5 h post-mating), and post-ovulatory (20 and 36 h post-mating) rabbits
| Time post-mating (h) . | No. of oviducts tested . | Ovulation . | Temperature (°C) measured in the . | . | . | ||
|---|---|---|---|---|---|---|---|
| . | . | . | Rectum . | Sperm storage site . | Fertilization site . | ||
| 0 | 6 | No | 38.0±0.04 | 35.1±0.2 | 35.9±0.2 | ||
| 5 | 4 | No | 38.3±0.03 | 35.8±0.2 | 36.6±0.1 | ||
| 10.5 | 8 | Yes | 38.2±0.05 | 34.9±0.2 | 36.2±0.3 | ||
| 20 | 4 | Yes | 38.2±0.02 | 34.7±0.1 | 36.3±0.2 | ||
| 36 | 4 | Yes | 38.4±0.02 | 34.8±0.3 | 36.1±0.1 | ||
| Time post-mating (h) . | No. of oviducts tested . | Ovulation . | Temperature (°C) measured in the . | . | . | ||
|---|---|---|---|---|---|---|---|
| . | . | . | Rectum . | Sperm storage site . | Fertilization site . | ||
| 0 | 6 | No | 38.0±0.04 | 35.1±0.2 | 35.9±0.2 | ||
| 5 | 4 | No | 38.3±0.03 | 35.8±0.2 | 36.6±0.1 | ||
| 10.5 | 8 | Yes | 38.2±0.05 | 34.9±0.2 | 36.2±0.3 | ||
| 20 | 4 | Yes | 38.2±0.02 | 34.7±0.1 | 36.3±0.2 | ||
| 36 | 4 | Yes | 38.4±0.02 | 34.8±0.3 | 36.1±0.1 | ||
The results shown are the mean ± SEM.
We thank Prof. Edna Schechtman, a professional statistician, for helping us with the statistical analysis, and Dr. Bella Finarov for helping us with the animals. M.E. is an incumbent of Jack and Simon Djanogly Professorial Chair in Biochemistry. This study was supported by the Horowitz Foundation, the Philip M. Klutznick Research Fund, the Woman's Health Research Center, and the Gerald Bamberger research grant.
References
Bahat A, Tur-Kaspa I, Gakamsky A, Giojalas LC, Breitbart H and Eisenbach M (
Barratt CLR and Cooke ID (
Battalia DE and Yanagimachi R (
Cicinelli E, Einer-Jensen N, Barba B, Luisi D, Alfonso R and Tartagni M (
David A, Vilensky A and Nathan H (
Eisenbach M (
Gandolfi F (
Giojalas LC, Rovasio RA, Fabro G, Gakamsky A and Eisenbach M (
Glad Sorensen H, Lambrechtsen J and Einer-Jensen N (
Grinsted J, Blendstrup K, Andreasen MP and Byskov AG (
Grinsted J, Kjer JJ, Blendstrup K and Pedersen JF (
Harkness JE and Wagner JE (
Harper MJK (
Hunter RHF and Nichol R (
Hunter RHF, Gronadahl C, Greve T and Schmidt M (
Jaiswal BS and Eisenbach M (
Jansen RP and Bajpai VK (
Luck MR, Griffiths S, Gregson K, Watson E, Nutley M and Cooper A (
Overstreet JW (
Ramirez VD and Soufi WL (
Suarez SS (
Author notes
1Department of Biological Chemistry, The Weizmann Institute of Science, 76100 Rehovot and 2The Institute for Human Reproduction, and the Reproductive Genetics Institute, Chicago, IL 60657, USA



![Time-dependent temperature differences between the storage and fertilization sites within the rabbit oviduct. The squares stand for the experimental points (mean±SEM; 4–8 oviducts for each point). The connecting line is a theoretical sigmoidal-curve fitting (R2=0.99), obtained according to the Boltzmann equation (Origin 6.1 software, OriginLab): \batchmode \documentclass[fleqn,10pt,legalpaper]{article} \usepackage{amssymb} \usepackage{amsfonts} \usepackage{amsmath} \pagestyle{empty} \begin{document} \(\mathit{Y}=(\mathit{A}_{1}{-}\ \mathit{A}_{2})/[1+\mathit{e}^{(\mathit{X}{-}\mathit{X}_{0})/\mathit{dX}}]+\mathit{A}_{2}\) \end{document} where A1 and A2 stand for the initial and final Y-axis values, respectively, dX for the distance on the X-axis between these points, and X0 for the center of dX. The weighting of the experimental points was according to 1/SEM2. Note that the theoretical sigmoidal curve is restricted to the time window between several hours prior to ovulation and several hours subsequent to it. It should not be extrapolated beyond this time window, where ΔT probably drops.](https://oup.silverchair-cdn.com/oup/backfile/Content_public/Journal/humrep/20/8/10.1093_humrep_dei006/3/m_141586f2.jpeg?Expires=1716321116&Signature=OzvUqOXBEA0G80brly09GBRPe1~1m5U7tBjrBuG4D--yd63OYgHY6QmewYM3-Kun1m5MgeB2y5fV~Z7KCS~Gq8K~LziSZvjo5bMc5L1Y3ouHYSu-KD0txjKcoXBIZUoGp-1WteDeN44i3ZihEBkNAr1ANicSZKRqmAC298ywpWSJjn5MTJzeqCrupjqx9wzb3VEMUh-Np4b2c8UFTVhEF1fN0-pPcKtnYrxO31VnUi4HbiZsAfk86O0cHeaujPKhKsr~AJSvZkK~b981HWnzxU6greLe9RJQVw0Rtn082xn0QXWgL-2ZqhxGAcG3eOcIqQL3iBEuG~UY9dqUUcEWng__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)
