Summary
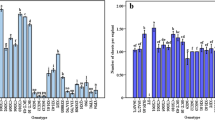
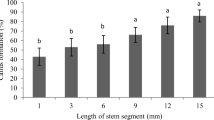
Improved in vitro tissue culture systems are needed to facilitate the application of transgene technology to the improvement of sugar beet germplasms. Several commercially important sugar beet breeding lines (SDM, 3, 5, 8, 9, 10, 11, HB 526, and CMS 22003) and commercial varieties (Roberta and Gala) were tested for their regeneration capacity through adventitious shoot organogenesis from cotyledons, hypocotyls, root/hypocotyl/shoot transition zone tissues, and leaf lamina and petiole via an intervening callus phase. Callus induction and adventitious shoot regeneration was dependent on genotype and combinations of plant growth regulators. With cotyledon or hypocotyl explants, SDM 3 and 10 showed a better response on adventitious shoot regeneration in medium containing benzyladenine (BA) and 2,3,5-triiodobenzoic acid or 1-naphthaleneacetic acid (NAA) than SDM 11, 5, and 9. Shoot regeneration was obtained from hypocytyl-root or hypocotyl-shoot transition zone tissue in SDM 9, 10, and HB 526 grown on PGo medium supplemented with BA to induce callus, and the regeneration frequency was 25%. Adventitious shoots were also regenerated from leaf explants of SDM 3 and 9 cultured on medium containing NAA for callus induction and BA and NAA to induce shoot regeneration, and in SDM 10 and CSM 22003 cultured on medium containing BA for callus induction and to induce shoot regeneration.
Similar content being viewed by others
References
Atanassov, A. I. Sugar beet. In: Evans, D. A.; Sharpe, W. R.; Ammirato, P. V., eds. Handbook of plant cell culture, vol. 4, New York: Macmillan Publishing Co.; 1986:652–689.
Brandstatter, J.; Kieber, J. J. Two genes with similarity to bacterial response regulators are rapidly and specifically induced by cytokinins in Arabidopsis. Plant Cell 10:1009–1019; 1998.
Catlin, D. W. The effect of antibiotics on the inhibition of callus induction and regeneration from cotyledons of sugarbeet. Plant Cell Rep. 9:285–288; 1990.
De Greef, W.; Jacobs, M. In vitro culture of the sugarbeet: description of a cell line with high regeneration capacity. Plant Sci. Lett., 17:55–61; 1979.
Detrez, C.; Tetu, T.; Sangwan, R. S.; Sangwan-Norreel, B. S. Direct organogenesis from petiole and thin cell layer explants in sugar beet cultured in vitro. J. Exp. Bot. 39:917–926; 1988.
D'Halluin, K.; Bossut, M.; Bonne, E.; Mazur, B.; Leemans, J.; Betterman, J. Transformation of sugarbeet (Beta vulgaris L.) and evaluation of herbicide resistance in transgenic plants. Bio/Technology 10:309–313; 1992.
Doley, W. P.; Saunders, J. W. Hormone-free medium will support callus production and subsequent shoot regeneration from whole plant leaf explants in some sugarbeet (Beta vulgaris L.) populations. Plant Cell Rep. 8:222–225; 1989.
Elliott, M. C.; Chen, D.-F.; Fowler, M. R.; Kirby, M. J.; Kubalakova, M.; Scott, N. W.; Slater, A. Transgenesis—a scheme for improving sugar beet productivity. Russian J. Plant Physiol. 43:544–551; 1996.
Frank, M.; Rupp, H. M.; Prinsen, E.; Motyka, V.; Van Onckelen, H.; Schmulling, T. Hormone autotrophic growth and differentiation identifies mutant lines of Arabidopsis thaliana with altered cytokinin and auxin content or signaling. Plant Physiol. 122:721–730; 2000.
Freytag, A. H.; Anand, S. C.; Rao-Arelli, A. P.; Owens, L. D. An improved medium for adventitious shoot formation and callus induction in Beta vulgaris L. in vitro. Plant Cell Rep. 7:30–34; 1988.
Grieve, T. M.; Gartland, K. M. A.; Elliott, M. C. Micropropagation of commercially important sugar beet cultivars. Plant Growth Regul. 21:15–18; 1997.
Hosemans, D.; Bossoutrot, D. Induction of haploid plants from in vitro culture of unpollinated beet ovules (Beta vulgaris L.). Z. Pflanzenzucht 91:74–77; 1983.
Jacq, B.; Tetu, T.; Sangwan, R. S.; De Laat, A.; Sangwan-Norreel, B. S. Plant regeneration from sugarbeet (Beta vulgaris L.) hypocotyls cultured in vitro and flow cytometric nuclear DNA analysis of regenerants. Plant Cell Rep. 11:329–333; 1992.
Jacq, B.; Tetu, T.; Sangwan, R. S.; Sangwan-Norreel, B. S. Efficient production of uniform plants from cotyledon explants of sugarbeet (Beta vulgaris L.). Plant Breed. 110:185–191; 1993.
Kakimoto, T. CKI1, a histidine kinase homolog implicated in cytokinin signal transduction. Science 274:982–985; 1996.
Kubalakova, M. Somatic embryogenesis and cytoplasmic sterility in Beta vulgaris L. var. Saccharifera. Biol. Plant. 32:414–419; 1990.
Levall, M. W.; Bornman, J. F. Selection in vitro for UV-tolerant sugarbeet (Beta vulgaris) somaclones. Physiol. Plant. 88:37–43; 1993.
Liu, C.-M.; Xu, Z.-H.; Chua, N.-H. Auxin polar transport is essential for the establishment of bilateral symmetry during early plant embryogenesis. Plant Cell 5:621–630; 1963.
Murashige, T.; Skoog, F. A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol. Plant. 15:473–497; 1962.
Murthy, B. N. S.; Murch, S. J.; Saxena, P. K. Thidiazuron induced somatic embryogenesis in intact seedling of peanut (Arachis hypogaea): endogenous growth regulator levels and significance of cotyledons. Physiol. Plant. 94:268–276; 1995.
Pedersen, M. G.; Andersen, J. M. Callus morphology and endogenous cytokinin in regenerating sugar beet callus cultures. Proc. Beta Eucarpia Meeting, Helsingor; 1988.
Pua, E. C.; Thorpe, T. A. Differential response of non-selected and Na2SO4-selected callus cultures of Beta vulgaris L. to salt stress. J. Plant Physiol. 123:241–248; 1986.
Ritchie, G. A.; Short, K. C.; Davey, M. R. In vitro shoot regeneration from callus, leaf axils and petioles of sugar beet (Beta vulgaris L.). J. Exp. Bot. 40:277–283; 1989.
Roussy, I.; Dubois, F.; Sangwan, R. S.; Sangwan-Norreel, B. S. In planta 2,3,5-triiodobenzoic acid treatment promotes high frequency and routine in vitro regeneration of sugarbeet (Beta vulgaris L.) plants. Plant Cell Rep. 16:142–146; 1996.
Rupp, H. M.; Frank, M.; Werner, T.; Strnad, M.; Schmulling, T. Increased steady mRNA levels of the STM and KNAT1 homeobox genes in cytokinin overproducing Arabidopsis thaliana indicate a role for cytokinins in the shoot apical meristem. Plant J. 18:557–563; 1999.
Saunders, J. W.; Acquaah, G.; Renner, K. A.; Doley, W. P. Monogenic dominant sulfonylurea resistance in sugarbeet from somatic cell selection. Crop Sci. 32:1357–1360; 1992.
Saunders, J. W.; Doley, W. P. One step shoot regeneration from callus of whole plant leaf explants of sugarbeet lines and a somaclonal variation for in vitro culture behaviour. J. Plant Physiol. 124:473–479; 1986.
Steel, R. G. D.; Torrie, J. H. Principles and procedures of statistics, a biometrical approach, 2nd edn. New York: McGraw Hill; 1980.
Tenning, P.; Weich, E. W.; Kjarsgaard, U.-B.; Lelu, M.-A.; Nihlgard, M. Somatic embryogenesis from zygotic embryos of sugarbeet (Beta vulgaris L.). Plant Sci. 81:103–109; 1992.
Tetu, T.; Sangwan, R. S.; Sangwan-Norreel, B. S. Hormonal control of organogenesis and somatic embryogenesis in Beta vulgaris callus. J. Exp. Bot. 38:506–517; 1987.
Van Geyt, J.; Speckmann, G. J. Jr.; D'Halluin, K.; Jacobs, M. In vitro induction of haploid plants from unpollinated ovules and ovaries of the sugarbeet (Beta vulgaris L.). Theor. Appl. Genet. 73:920–925; 1987.
Yu, M. H. Growth and reproduction performance of ovule-induced sugarbeet plants. SABRAO J. 24:47–55; 1992.
Zhang, C.-L.; Chen, D.-F.; Elliott, M. C.; Slater, A. TDZ-induced organogenesis and somatic embryogenesis in sugar beet (Beta vulgaris L.). In Vitro Cell. Dev. Biol. Plant 37:305–310; 2001a.
Zhang, C.-L.; Chen, D.-F.; McCormac, A. C.; Scott, N. W.; Elliott, M. C.; Slater, A. Use of the GFP reporter as a vital marker for Agrobacterium-mediated transformation of sugar beet (Beta vulgaris L.). Mol. Biotech. 17:109–117; 2001b.
Zhong, Z.; Smith, H. G.; Thomas, T. H. In vitro culture of petioles and intact leaves of sugar beet (Beta vulgaris). Plant Growth Regul. 12:59–66; 1993.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, CL., Chen, DF., Elliott, M.C. et al. Efficient procedures for callus induction and adventitious shoot organogenesis in sugar beet (Beta vulgaris L.) breeding lines. In Vitro Cell.Dev.Biol.-Plant 40, 475–481 (2004). https://doi.org/10.1079/IVP2004563
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1079/IVP2004563