Abstract
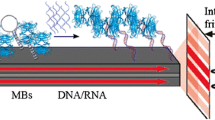
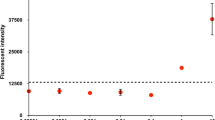
Externally sensitized electron-transfer fragmentation in dithiane PPG (photoremovable protecting group)-protected carbonyls is adopted for detection and amplification of molecular recognition events. The new methodology allows for detection of as low as 50 attomoles of avidin utilizing an imager based on a low sensitivity mass-produced consumer CCD camera. Numeric modelling is carried out to demonstrate the intrinsic limitations of 2D amplification on surfaces and the advantages of unconstrained amplification in a compartmentalized volume of spatially addressable 3D solutions.
Similar content being viewed by others
Notes and references
For a historical overview, see: R. M. Lequin, Enzyme Immunoassay (EIA)/Enzyme-Linked Immunosorbent Assay (ELISA), Clin. Chem., 2005, 51, 2415.
R. M. Eglen, T. Reisine, P. Roby, N. Rouleau, C. Illy, R. Bossé and M. Bielefeld, The use of AlphaScreen technology in HTS: current status, Curr. Chem. Genomics, 2008, 1, 2.
M. Bielefeld-Sevigny, AlphaLISA Immunoassay Platform–The “No-Wash” High-Throughput Alternative to ELISA, Assay Drug Dev. Technol., 2009, 7, 90.
N. C. Gianneschi, S. T. Nguyen and C. A. Mirkin, Signal Amplification and Detection via a Supramolecular Allosteric Catalyst, J. Am. Chem. Soc., 2005, 127, 1644.
M. S. Masar III, N. C. Gianneschi, C. G. Oliveri, C. L. Stern, S. T. Nguyen and C. A. Mirkin, Allosterically Regulated Supramolecular Catalysis of Acyl Transfer Reactions for Signal Amplification and Detection of Small Molecules, J. Am. Chem. Soc., 2007, 129, 10149.
H. D. Sikes, R. R. Hansen, L. M. Johnson, R. Jenison, J. W. Birks, K. L. Rowlen, et al., Using polymeric materials to generate an amplified response to molecular recognition events, Nat. Mater., 2007, 7, 52.
H. J. Avens and C. N. Bowman, Development of fluorescent polymerization-based signal amplification for sensitive and non-enzymatic biodetection in antibody microarrays, Acta Biomater., 2010, 6, 83.
For a hybrid enzymatic methodology augmented with polymerization-based amplification, see
B. J. Berron, L. M. Johnson, X. Aa, J. D. McCall, N. J. Alvey, K. S. Anseth and C. N. Bowman, Glucose Oxidase-Mediated Polymerization as a Platform for Dual-Mode Signal Amplification and Biodetection, Biotechnol. Bioeng., 2011, 108, 1521.
E. J. Corey and D. Seebach, Carbanions of 1,3-dithianesReagents for C–C bond formation by nucleophilic displacement and carbonyl addition, Angew. Chem., Int. Ed. Engl., 1965, 4, 1075;. For a review, see
B.-T. Gröbel and D. Seebach, Umpolung of the reactivity of carbonyl compounds through sulfur-containing reagents, Synthesis, 1977, 357.
W. A. McHale and A. G. Kutateladze, An Efficient Photo-SET-Induced Cleavage of Dithiane-Carbonyl Adducts and Its Relevance to the Development of Photoremovable Protecting Groups for Ketones and Aldehydes, J. Org. Chem., 1998, 63, 9924.
R. Kottani, R. A. Valiulin and A. G. Kutateladze, Direct Screening of Solution Phase Combinatorial Libraries Encoded with Externally Sensitized Photolabile Tags, Proc. Natl. Acad. Sci. U. S. A., 2006, 103, 13917.
R. R. Kottani, J. R. R. Majjigapu, A. N. Kurchan, K. Majjigapu, T. P. Gustafson and A. G. Kutateladze, Photoinduced Signal Amplification Through Controlled Externally Sensitized Fragmentation in Masked Sensitizers, J. Am. Chem. Soc., 2006, 128, 14794.
K. Majjigapu, J. R. R. Majjigapu and A. G. Kutateladze, Photoamplification and Multiple Tag Release in a Linear Peptide-Based Array of Dithiane Adducts, Angew. Chem., Int. Ed., 2007, 46, 6137.
For the initial communication, see: T. P. Gustafson, G. A. Metzel and A. G. Kutateladze, Photochemically Amplified Detection of Molecular Recognition Events: An Ultra-Sensitive Fluorescence Turn-Off Binding Assay, Org. Biomol. Chem., 2011, 9, 4752.
M. W. Wolf, K. D. Legg, R. E. Brown, L. A. Singer and J. H. Parks, Photophysical studies on the benzophenonesPrompt and delayed fluorescences and self-quenching, J. Am. Chem. Soc., 1975, 97, 4490.
P. Vath, D. E. Falvey, L. A. Barnhurst and A. G. Kutateladze, Photoinduced C-C Bond Cleavage in Dithiane-Carbonyl Adducts: A Laser Flash Photolysis Study, J. Org. Chem., 2001, 66, 2887.
The term Markov chain is used here to indicate that we limited our numerical model to a random walk process where the next step is defined by the current state of the system (not by the history of events leading to the current state of the system). For an overview of numerical modelling, see: W. J. Stewart, Introduction to the Numerical Solution of Markov Chains, Princeton Univ. Press., 1994.
I. B. Berlman, Handbook of Fluorescence Spectra of Aromatic Molecules, Academic Press, 1971.
H. Bücher and W. Chow, A 1 MWP-terphenyl dye laser, Appl. Phys., 1977, 13, 267.
N. I. Nijegorodov, W. S. Downey and M. B. Danailov, Systematic investigation of absorption, fluorescence and laser properties of some p- and m-oligophenylenes, Spectrochim. Acta, Part A, 2000, 56, 783.
A reviewer suggested that the mistriggering of the amplification cascade in the absence of benzophenone could also be due to a complex with oxygen. Most of our initial bulk experiments were carried out after 4–5 freeze-thaw degassing cycles; yet, after some induction period samples not seeded with benzophenone exhibited amplified fluorescence quenching. It is believed that the freeze-thaw degassing lowers the concentration of oxygen to the ppb levels. Since benzophenone itself is capable of initiating the amplification cascade at ppb levels, it is difficult to assess the feasibility of oxygen triggering the fragmentation at these low levels.
For manufacturer’s specifications, see: http://www.nichia.com/specification/en/product/led/NCSU033B-E.pdf.
G. Jones, S. F. Griffin, C. Y. Choi and W. R. Bergmark, Electron donor–acceptor quenching and photoinduced electron transfer for coumarin dyes, J. Org. Chem., 1984, 49, 2705.
G. Jones and W. R. Bergmark, Photodegradation of coumarin laser dyes: an unexpected singlet self-quenching mechanism, J. Photochem., 1984, 26, 179.
P. Gray and A. Williams, Chem. Rev., 1959, 59, 239.
A. Pal and G. Dass, Excess molar volumes and viscosities of binary mixtures of tetraethylene glycol dimethyl ether (tetraglyme) with chloroalkanes at 298.15 K, J. Mol. Liq., 2000, 84, 327.
C. Lafuente, H. Artigas, J. Pardo, F. M. Royo and J. S. Urieta, Viscosities of 1-chlorobutane and 1,4-dichlorobutane with isomeric butanols at 25 and 40 °C, J. Solution Chem., 1996, 25, 303.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gustafson, T.P., Metzel, G.A. & Kutateladze, A.G. Externally sensitized deprotection of PPG-masked carbonyls as a spatial proximity probe in photoamplified detection of binding events. Photochem Photobiol Sci 11, 564–577 (2012). https://doi.org/10.1039/c2pp05326h
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1039/c2pp05326h