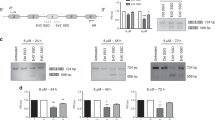
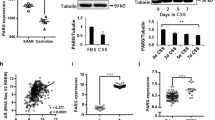
Abstract
Claims about molecular mechanisms underlying the resistance to anti-hormones of prostate cancer cells find support in biological experiments, which involve hormone-independent activation of the androgen receptor's (AR) transcriptional activity. In order to test this hypothesis, we attempted to shut down the expression of AR by the means of target-directed antisense oligonucleotides. A set of 49 oligonucleotides matching sequences of the AR mRNA either in the coding sequence or in the 3′ and 5′ untranslated regions were synthesized and examined in a cellular AR-dependent reporter system. Five antisense oligonucleotides were identified as highly potent inhibitors of AR-driven gene expression in a cellular reporter assay. These five were further profiled using point-mutated control sequences for the assessment of AR inhibition. In addition the expression of another AR-driven gene, the modulator of PSA expression (gene for inhibition of prostate specific antigen, an endogenous, AR-driven gene) was examined. Finally, we observed that the hormone-independent but AR-mediated transactivation by IGF-1 could also be specifically shut-down by these antisense oligonucleotides. The selection of highly target-restricted antisense oligonucleotides in the prostate cancer cell line LNCaP provided tools to study a central role of the androgen receptor in growth regulation of prostatic cancer cell lines and could be of utility in cancer situations in vivo.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 4 print issues and online access
$259.00 per year
only $64.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Trapman J, Brinkmann AO . The androgen receptor in prostate cancer. Path Res Pract 1996; 192: 752–760.
Yeung F, et al. Regions of prostate-specific antigen (PSA) promoter confer androgen-independent expression of PSA in prostate cancer cell line. J Biol Chem 2000; 275: 40846–40855.
Visakorpi T, et al. In vivo amplification of the androgen receptor gene and progression of human prostate cancer. Nature Genet 1995; 9: 401–404.
Gottlieb B, et al. Update of the androgen receptor gene mutation database. Hum Mutat 1999; 14: 103–114.
Suzuki H, et al. Androgen receptor gene mutations in human prostate cancer. Steroid Biochem Mol Biol 1993; 46: 759–765.
Nessler-Menardi C, et al. Expression of androgen receptor co-regulatory proteins in prostate cancer ans stromal-cell culture models. Prostate 2000; 45: 124–131.
Culig Z, et al. Androgen receptor activation in prostatic tumor cell lines by insulin-like growth factor-I, Keratinocyte growth factor and epidermal growth factor. Eur Urol 1995; 27: 45–47.
Horoszewicz JS, et al. LNCaP model of human prostatic carcinoma. Cancer Res 1983; 43: 1809–1818.
Dodd JG, Sheppard PC, Matusik RJ . Characterization and cloning of rat dorsal prostate mRNAs. Androgen regulation of two closely related abundant mRNAs. J Biol Chem 1983; 258: 177–180.
Matusik RJ, et al. Regulation of prostatic genes: role of androgens and zinc in gene expression. Biochem Cell Biol 1986; 64: 601–607.
Sweetland R, Sheppard PC, Dodd JG, Matusik RJ . Post-castration rebound of an androgen regulated prostatic gene. Mol Cell Biochem 1988; 84: 3–15.
Rennie PS, et al. Characterization of two cis-acting DNA elements involved in the androgen regulation of the probasin gene. Mol Endocrinol 1993; 7: 23–26.
Laudet V . Evolution of the nuclear receptor superfamily: early diversification from an ancestral orphan receptor. J Mol Endocrinol 1997; 19: 207–226.
Stamey TA, et al. Prostate-specific antigen as a serum marker for adenocarcinoma of the prostate. N Engl J Med 1987; 317: 909–916.
Riegman PHJ, et al. The promoter of the prostate-specific antigen gene contains an androgen-responsive element. Mol Endocrinol 1991; 5: 1921–1930.
Helene C, Toulme JJ . Specific gene regulation of gene expression by antisense, sense and antigene nucleic acids. Biochim Biophys Acta 1990; 1049: 99–125.
Wyatt JR, et al. Combinatorially selected guanosine-quartet structure is a potent inhibitor of human immunodeficiency virus envelope-mediated cell fusion. Proc Natl Acad Sci USA 1994; 91: 1356–1360.
Weiler J, Hoheisel JD . Combining the preparation of oligonucleotide arrays and synthesis of high-quality primers. Anal Biochem 1996; 243: 218–227.
Eder IE, et al. Inhibition of LNCaP prostate cancer cells by means of androgen receptor antisense oligonucleotides. Cancer Gene Ther 2000; 7: 997–1007.
Eder IE, et al. Inhibition of LNCaP prostate tumor growth in-vivo by an antisense oligonucleotide directed against the human androgen receptor. Cancer Gene Ther 2002; 9: 117–125.
Acknowledgements
We would like to express our thanks to Bernard Cuenoud and Jan Weiler for their technological expertise and for stimulating discussion, and Geneviève Albrecht for excellent technical assistance!
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hamy, F., Brondani, V., Spoerri, R. et al. Specific block of androgen receptor activity by antisense oligonucleotides. Prostate Cancer Prostatic Dis 6, 27–33 (2003). https://doi.org/10.1038/sj.pcan.4500603
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.pcan.4500603
Keywords
This article is cited by
-
ARe we there yet? Understanding androgen receptor signaling in breast cancer
npj Breast Cancer (2020)