Abstract
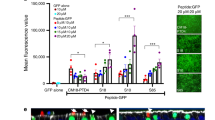
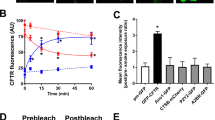
Delivery of a normal copy of CFTR cDNA to airway epithelia may provide a novel treatment for cystic fibrosis lung disease. Unfortunately, current vectors are inefficient because of limited binding to the apical surface of airway epithelia. We recently reported that incorporation of adenovirus in a calcium phosphate coprecipitate (Ad:CaPi) improves adenovirus-mediated gene transfer to airway epithelia in vitro and in vivo. To understand better how coprecipitation improves gene transfer, we tested the hypothesis that incorporation in a CaPi coprecipitate increases the binding of adenovirus to the apical surface of differentiated human airway epithelia. When a Cy3-labelled adenovirus was delivered in a coprecipitate, binding increased 54-fold as compared with adenovirus alone. Moreover, infection by Ad:CaPi was independent of fiber knob-CAR and penton base–integrin interactions. After binding to the cell surface, the virus must enter the cell in order to infect. We hypothesized that Ad:CaPi may stimulate fluid phase endocytosis, thereby facilitating entry. However, we found that neither adenovirus nor Ad:CaPi coprecipitates altered fluid phase endocytosis. Nevertheless, Ad:CaPi preferentially infected cells showing endocytosis. Thus, CaPi coprecipitation improves adenovirus-mediated gene transfer by coating the epithelial surface with a layer of virus which enters cells during the normal process of endocytosis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
O’Neal WK, Beaudet AL . Somatic gene therapy for cystic fibrosis Hum Mol Genet 1994 3: 1497–1502
Crystal RG . Transfer of genes to humans: early lessons and obstacles to success Science 1995 270: 404–410
Welsh MJ, Tsui L-C, Boat TF, Beaudet AL . Cystic fibrosis. In: Scriver CR, Beaudet AL, Sly WS, Valle D (eds) The Metabolic and Molecular Basis of Inherited Disease McGraw-Hill: New York 1995 pp 3799–3876
Grubb BR et al. Inefficient gene transfer by adenovirus vector to cystic fibrosis airway epithelia of mice and humans Nature 1994 371: 802–806
Zabner J, Zeiher BG, Friedman E, Welsh MJ . Adenovirus-mediated gene transfer to ciliated airway epithelia requires prolonged incubation time J Virol 1996 70: 6994–7003
Zabner J et al. Lack of high affinity fiber receptor activity explains the resistance of ciliated airway epithelia to adenovirus infection J Clin Invest 1997 100: 1144–1149
Pickles RJ et al. Limited entry of adenovirus vectors into well-differentiated airway epithelium is responsible for inefficient gene transfer J Virol 1998 72: 6014–6023
Walters RW et al. Basolateral localization of fiber receptors limits adenovirus infection from the apical surface of airway epithelia J Biol Chem 1999 274: 10219–10226
Bergelson JM et al. Isolation of a common receptor for Coxsackie B viruses and adenoviruses 2 and 5 Science 1997 275: 1320–1323
Wickham TJ, Mathias P, Cheresh DA, Nemerow GR . Integrins alpha beta 3 and alpha beta 5 promote adenovirus internalization but not virus attachment Cell 1993 73: 309–319
Greber UF, Willetts M, Webster P, Helenius A . Stepwise dismantling of adenovirus 2 during entry into cells Cell 1993 75: 477–486
Mathias P, Wickham T, Moore M, Nemerow G . Multiple adenovirus serotypes use alpha integrins for infection J Virol 1994 68: 6811–6814
Fasbender A et al. Incorporation of adenovirus in calcium phosphate precipitates enhances gene transfer to airway epithelia in vitro and in vivo J Clin Invest 1998 102: 184–193
Leopold PL et al. Fluorescent virions: dynamic tracking of the pathway of adenoviral gene transfer vectors in living cells Hum Gene Ther 1998 9: 367–378
Lee JH, Zabner J, Welsh MJ . Delivery of an adenovirus vector in a calcium phosphate coprecipitate enhances the therapeutic index of gene transfer to airway epithelia Hum Gene Ther 1999 10: 603–613
Bai M, Harfe B, Freimuth P . Mutations that alter an Arg-Gly-Asp (RGD) sequence in the adenovirus type 2 penton base protein abolish its cell-rounding activity and delay virus reproduction in flat cells J Virol 1993 67: 5198–5205
Persson R, Svensson U, Everitt E . Virus receptor interaction in the adenovirus system. ii. capping and cooperative binding of virions on hela cells J Virol 1983 46: 956–963
Greber UF, Singh I, Helenius A . Mechanisms of virus uncoating Trends Microbiol 1994 2: 52–56
Wickham TJ, Filardo EJ, Cheresh DA, Nemerow GR . Integrin alpha beta 5 selectively promotes adenovirus mediated cell membrane permeabilization J Cell Biol 1994 127: 257–264
Seth P, Willingham MC, Pastan I . Adenovirus-dependent release of 51cr from kb cells at an acidic ph J Biol Chem 1984 259: 14350–14353
Svensson U . Role of vesicles during adenovirus 2 internalization into hela cells J Virol 1985 55: 442–449
Greber UF et al. The role of the nuclear pore complex in adenovirus DNA entry EMBO J 1997 16: 5998–6007
Kondo M, Finkbeiner WE, Widdicombe JH . Simple technique for culture of highly differentiated cells from dog tracheal epithelium Am J Physiol 1991 261: L106–L117
Yamaya M, Finkbeiner WE, Chun SY, Widdicombe JH . Differentiated structure and function of cultures from human tracheal epithelium Am J Physiol 1992 262: L713–L724
Welsh MJ et al. Adenovirus-mediated gene transfer for cystic fibrosis: Part A. Safety of dose and repeat administration in the nasal epithelium. Part B. Clinical efficacy in the maxillary sinus Hum Gene Ther 1995 6: 205–218
Flotte TR et al. A fluorescence video-endoscopy technique for detection of gene transfer and expression Gene Therapy 1998 5: 166–173
Zolotukhin S et al. A ‘humanized’ green fluorescent protein cDNA adapted for high-level expression in mammalian cells J Virol 1996 70: 4646–4654
Zabner J et al. A chimeric type 2 adenovirus vector with a type 17 fiber enhances gene transfer to human airway epithelia J Virol (in press)
Acknowledgements
We thank Pary Weber, Phil Karp, Tom Moninger, Mike Seiler and Theresa Mayhew for excellent assistance. We especially appreciated the generous help of the late Dr Al Fasbender. We thank Dr Sam Wadsworth, Genzyme (Framingham, MA, USA) for the gift of Ad2/GFP and Ad2/βGal. We thank Dr Paul Freimuth for the gift of wild-type adenovirus, and the RAE mutant adenovirus. We thank Dr Jeff Bergelson for the gift of pcDNA1hCAR. We thank the University of Iowa Gene Transfer Vector Core (supported in part by the Roy J Carver Charitable Trust, the National Heart, Lung and Blood Institute, and the Cystic Fibrosis Foundation) for support. This work was supported by the National Heart, Lung and Blood Institute and the Cystic Fibrosis Foundation. MJW is an Investigator of the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Walters, R., Welsh, M. Mechanism by which calcium phosphate coprecipitation enhances adenovirus-mediated gene transfer. Gene Ther 6, 1845–1850 (1999). https://doi.org/10.1038/sj.gt.3301020
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3301020
Keywords
This article is cited by
-
A Self-Biomineralized Novel Adenovirus Vectored COVID-19 Vaccine for Boosting Immunization of Mice
Virologica Sinica (2021)
-
Toll-like Receptor 9 Triggers an Innate Immune Response to Helper-dependent Adenoviral Vectors
Molecular Therapy (2007)
-
Calcium phosphate precipitates augment adenovirus-mediated gene transfer to blood vessels in vitro and in vivo
Gene Therapy (2000)