Abstract
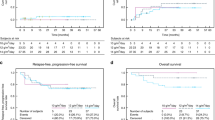
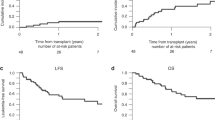
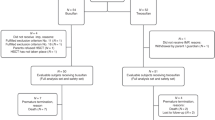
Although infants with stage 4 neuroblastoma (NB) usually have a good prognosis, metastatic relapses after 1 year of age and amplification of the N-myc oncogene are established poor prognostic factors. In order to improve the survival of patients with such high-risk factors, we performed consolidation with a busulfan (600 mg/m2)-melphalan (140 mg/m2)-containing regimen followed by autologous stem cell transplantation (SCT). From 1986 to 1998, 12 patients were treated according to this strategy. Their median age at diagnosis was 9 months (1–11). Consolidation was performed after a metastatic relapse in five children, because of persistent bone metastases in one and as first-line consolidation in six patients whose tumor exhibited N-myc amplification. The 5-year EFS rate is 64.5% (36–85%) with a median follow-up of 92 months (20–126). One toxicity-related death occurred in a very heavily pretreated patient. Hepatic veno-occlusive disease was the major side-effect that occurred in nine of 12 children. This busulfan-melphalan combination appears to dramatically improve the prognosis of these high-risk infants with metastatic NB. Given its high toxicity, indications for this consolidation must be restricted to high-risk infants and a lower dose of busulfan (480 mg/m2) is recommended in children weighing less than 10 kg. Bone Marrow Transplantation (2000) 25, 937–942.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Evans AE, D’Angio GJ, Randolp J . A proposed staging for children with neuroblastoma. Children’s Cancer Study Group A Cancer 1971 27: 374–378
Brodeur GM, Pritchard J, Berthold F et al. Revisions of the International Criteria for Neuroblastoma Diagnosis, Staging and Response to Treatment J Clin Oncol 1993 11: 1466–1477
Castel Sanchez V, Melero Moreno C, Garcia-Miguel Garcia-Rosados A et al. Stage 4 neuroblastoma en ninos menores de 1 ano Ann Esp Pediatr 1997 47: 584–590
Evans AE, Chatten GJ, D’Angio GJ et al. A review of 17 IV-S neuroblastoma patients at the Children’s hospital of Philadelphia Cancer 1980 45: 833–839
Labreveux de Cervens C, Hartmann O, Bonnin F et al. What is the prognostic value of osteomedullary uptake on MIBG scan in neuroblastoma patients under 1 year of age? Med Ped Oncol 1994 22: 107–114
Bowman LC, Castelberry RP, Cantor A et al. Genetic staging of unresectable or metastatic neuroblastoma in infants: a Pediatric Oncology Group study J Natl Cancer Inst 1997 89: 373–380
Lampert F, Christiansen H, Terpe HJ et al. Disseminated neuroblastomas under 1 year of age: cell biology and prognosis J Neuro-Oncol 1997 31: 181–184
Hartmann O, Rubie H, Coze C et al. Stage 4 neuroblastoma in infants: N-myc amplification (NMA) correlates with adverse outcome. SIOP XXX meeting Med Ped Oncol 1998 31: 267 (Abstr. P-41)
Matthay KK . Stage 4s neuroblastoma: what makes it special? J Clin Oncol 1998 16: 2003–2006
Katzenstein HM, Bowman L, Brodeur GM et al. Prognostic significance of age, MYCN oncogene amplification, tumor cell ploidy, and histology in 110 infants with stage D(S) neuroblastoma: the Pediatric Oncology Group experience. A Pediatric Oncology Group study J Clin Oncol 1998 16: 2007–2017
Coze C, Hartmann O, Michon J et al. NB 87 induction protocol for Stage 4 neuroblastoma in children over 1 year of age: a report from the French Society of Pediatrics Oncology J Clin Oncol 1997 12: 3433–3440
Frappaz D, Michon J, Hartmann O et al. Etoposide and carboplatin in neuroblastoma: a French Society of Pediatric Oncology phase II study J Clin Oncol 1992 10: 1592–1601
Avril M, Hartmann O, Valteau-Couanet D et al. Anti-infective prophylaxis with ceftazidime and teicoplanin in children undergoing high-dose chemotherapy and bone-marrow transplantation Pediatr Hematol Oncol 1994 11: 63–73
Vassal G, Deroussent A, Hartmann O et al. Dose-dependent neurotoxicity of high-dose busulfan in children: a clinical and pharmacological study Cancer Res 1990 50: 6203–6207
Bearman SI, Appelbaum FR, Buckner CD et al. Regimen-related toxicity in patients undergoing bone marrow transplantation J Clin Oncol 1988 6: 1562–1568
McDonald GB, Sharma P, Matthews DE et al. Veno-occlusive disease of the liver after bone marrow transplantation: diagnosis, incidence and predisposing factors Hepatology 1984 4: 116–122
Valteau-Couanet D, Rubie H, Meresse V et al. Phase I–II study of interleukin-2 after high-dose chemotherapy and autologous bone marrow transplantation in poorly responding neuroblastoma Bone Marrow Transplant 1995 16: 515–520
Peto R, Pike MC, Armitage P et al. Design and analysis of randomised clinical trials requiring prolonged observations of each patient. II Analysis and examples Br J Cancer 1977 35: 1–39
Rothman KJ . Estimation of confidence limits for the cumulative probability of survival in life table analysis J Chron Dis 1978 31: 557–560
Peto R, Peto J . Asymptotically efficient rank invariant test procedures JR S Stat Soc A 1972 135: 185–207
Matthay KK, Harris R, Reynolds CP et al. Improved event-free survival (EFS) for autologous bone marrow transplantation (ABMT) vs chemotherapy in neuroblastoma: a phase III randomized Children’s Cancer Group (CCG) study Proc ASCO 1998 17: 525a (Abstr. 2018)
Paul SR, Tarbell NJ, Korf B et al. Stage IV neuroblastoma in infants Cancer 1991 67: 1493–1497
Seeger RC, Brodeur GM, Sather H et al. Association of multiple copies of N-myc oncogene with rapid progression of neuroblastomas New Engl J Med 1985 318: 111–116
Look AT, Hayes FA, Nitschke R et al. Clinical relevance of tumor cell ploidy and N-myc gene amplification in childhood neuroblastoma: a Pediatric Oncology Group study J Clin Oncol 1991 9: 581–591
Brodeur GM, Azar C, Brother M et al. Neuroblastoma. Effect of genetic factors on prognosis and treatment Cancer 1999 70: 1685–1694
Rubie H, Hartmann O, Michon J et al. N-myc gene amplification is a major prognostic factor in localized neuroblastoma: results of the French NBL 90 study J Clin Oncol 1997 15: 1171–1182
Hartmann O, Valteau-Couanet D, Vassal G et al. Prognostic factors in metastatic neuroblastoma in patients over 1 year of age treated with high-dose chemotherapy and stem cell transplantation: a multivariate analysis of 218 patients treated in a single institution Bone Marrow Transplant 1999 23: 789–795
Boland I, Vassal G, Morizet J et al. Busulfan is active against neuroblastoma and medulloblastoma xenografts in athymic mice at clinically achievable plasma drug concentrations Br J Cancer 1999 79: 787–792
Hartmann O, Valteau-Couanet D, Capelli C et al. High-dose chemotherapy with autologous bone-marrow support in neuroblastoma Sangre 1994 39: 168–171
Hartmann O, Valteau-Couanet D, Benhamou E et al. Stage IV neuroblastoma in patients over 1 year at diagnosis: consolidation of poor responders with combined busulfan, cyclophosphamide and melphalan followed by in vitro mafosfamide purged autologous bone marrow transplantation Eur J Cancer 1997 33: 2126–2129
Ladenstein R, Philip T, Lasset C et al. Multivariate analysis of risk factors in stage 4 neuroblastoma patients over the age of 1 year treated with megatherapy and stem-cell transplantation: a report from the European Bone Marrow Transplantation Solid Tumor Registry J Clin Oncol 1998 16: 953–965
Stram DO, Matthay K, Leary M et al. Consolidation chemotherapy and autologous bone marrow transplantation versus continued chemotherapy for metastatic neuroblastoma: a report of two concurrent children’s cancer group studies J Clin Oncol 1996 14: 2417–2426
Rosti G, Philip T, Chauvin F et al. European Group for Blood and Marrow Transplantation (EBMT) registry in solid tumors: 12 years of experience Bone Marrow Transplant 1996 17: (Suppl 1) 106 (Abstr. 469)
Hartmann O, Le Coroller AG, Blaise D et al. Peripheral blood stem cell and bone marrow transplantation for solid tumors and lymphomas: hematologic recovery and costs Ann Intern Med 1997 126: 600–607
Meresse V, Hartmann O, Vassal G et al. Risk factors for hepatic veno-occlusive disease after high-dose busulfan containing regimen followed by autologous bone and marrow transplantation: a study of 136 children Bone Marrow Transplant 1992 10: 135–141
Essel JH, Schroeder MT, Harman GS et al. Ursodiol prophylaxis against hepatic complications of allogeneic bone marrow transplantation Ann Intern Med 1998 128: 975–981
Slattery JT, Sanders JE, Buckner CD et al. Graft rejection and toxicity following bone marrow transplantation in relation to busulfan pharmacokinetics. (published erratum appears in Bone Marrow Transplant 1996; 18: 829) Bone Marrow Transplant 1995 16: 31–42
Vassal G, Koscielny S, Challine D et al. Busulfan disposition and hepatic veno-occlusive disease in children undergoing bone marrow transplantation Cancer Chemother Pharmacol 1996 37: 247–253
Vassal G . Pharmacologically guided dose adjustment of busulfan in high-dose chemotherapy regimens: rationale and pitfalls (review) Anticancer Res 1994 14: 2363–2370
Schuler US, Ehrsam M, Schneider A et al. Pharmacokinetics of intravenous busulfan and evaluation of the bioavailability of the oral formulation in conditioning for haematopoietic stem cell transplantation Bone Marrow Transplant 1998 22: 241–244
Bhagwatwar HP, Phadungpojna S, Chow DS et al. Formulation and stability of busulfan for intravenous administration in high-dose chemotherapy Cancer Chemother Pharmacol 1996 37: 401–408
Valteau-Couanet D, Kalifa C, Benhamou E et al. Repeated high-dose chemotherapy (HDC) followed by stem cell transplantation (SCT) in young children with poor prognosis medulloblastoma or PNET: tolerance and efficacy Bone Marrow Transplant 1998 21: (Suppl. 1) 228 (Abstr. 798)
Bown N, Cotteril S, Lastowska M et al. Gain of chromosome arm 17q and adverse outcome in patients with neuroblastoma New Engl J Med 1999 340: 1954–1961
Acknowledgements
We are grateful to L Saint Ange for editing the manuscript, and to Severine Faure for her secretarial assistance.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Valteau-Couanet, D., Benhamou, E., Vassal, G. et al. Consolidation with a busulfan-containing regimen followed by stem cell transplantation in infants with poor prognosis stage 4 neuroblastoma. Bone Marrow Transplant 25, 937–942 (2000). https://doi.org/10.1038/sj.bmt.1702376
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1702376
Keywords
This article is cited by
-
Busulfan–melphalan in high-risk neuroblastoma: the 30-year experience of a single institution
Bone Marrow Transplantation (2016)
-
Toxicities of busulfan/melphalan versus carboplatin/etoposide/melphalan for high-dose chemotherapy with stem cell rescue for high-risk neuroblastoma
Bone Marrow Transplantation (2016)
-
High-dose chemotherapy for high-risk retinoblastoma: clinical course and outcome of 14 cases in the National Cancer Center, Japan
Bone Marrow Transplantation (2015)
-
Physico-Chemical Stability of Busulfan in Injectable Solutions in Various Administration Packages
Drugs in R&D (2013)
-
Hepatic veno-occlusive disease in children after hematopoietic stem cell transplantation: incidence, risk factors, and outcome
Bone Marrow Transplantation (2010)