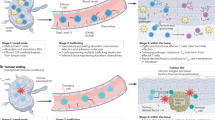
Abstract
γδ T cells are important tissue-resident, innate T cells that are critical for tissue homeostasis. γδ cells are associated with positive prognosis in most tumors; however, little is known about their heterogeneity in human cancers. Here, we phenotyped innate and adaptive cells in human colorectal (CRC) and endometrial cancer. We found striking differences in γδ subsets and function in tumors compared to normal tissue, and in the γδ subsets present in tumor types. In CRC, an amphiregulin (AREG)-producing subset emerges, while endometrial cancer is infiltrated by cytotoxic cells. In humanized CRC models, tumors induced this AREG phenotype in Vδ1 cells after adoptive transfer. To exploit the beneficial roles of γδ cells for cell therapy, we developed an expansion method that enhanced cytotoxic function and boosted metabolic flexibility, while eliminating AREG production, achieving greater tumor infiltration and tumor clearance. This method has broad applications in cellular therapy as an ‘off-the-shelf’ treatment option.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
Sequencing data from this study have been deposited in the NCB Gene Expression Omnibus under accession no. GSE210040. Furthermore, data from this study have been provided in the supplementary information. Publicly available data that were used to generate the survival curves are available from the NCI Genetic Data Commons (https://portal.gdc.cancer.gov/). Further scRNA-seq datasets were used from published sources, including GSE178341 and GSE216534. Source data are provided with this paper. All other data supporting the findings of this study are available from the corresponding author on reasonable request.
Code availability
The code used in this paper was modified from publicly available vignettes for the Seurat package (https://satijalab.org/seurat/index.html) and Scanpy package (https://scanpy.readthedocs.io/en/stable/). Survival analysis code can be found at https://brucemoran.github.io/charmon_tcga.
References
Siegel, R. L. et al. Colorectal cancer statistics, 2020. CA Cancer J. Clin. 70, 145–164 (2020).
Guinney, J. et al. The consensus molecular subtypes of colorectal cancer. Nat. Med. 21, 1350–1356 (2015).
Galon, J. et al. Towards the introduction of the ‘Immunoscore’ in the classification of malignant tumours. J. Pathol. 232, 199–209 (2014).
Li, X. et al. The immunological and metabolic landscape in primary and metastatic liver cancer. Nat. Rev. Cancer 21, 541–557 (2021).
Fontana, E., Eason, K., Cervantes, A., Salazar, R. & Sadanandam, A. Context matters—consensus molecular subtypes of colorectal cancer as biomarkers for clinical trials. Ann. Oncol. 30, 520–527 (2019).
Thorsson, V. et al. The immune landscape of cancer. Immunity 48, 812–830 (2018).
Singh, P. P., Sharma, P. K., Krishnan, G. & Lockhart, A. C. Immune checkpoints and immunotherapy for colorectal cancer. Gastroenterol. Rep. 3, gov053 (2015).
Nielsen, M. M., Witherden, D. A. & Havran, W. L. γδ T cells in homeostasis and host defence of epithelial barrier tissues. Nat. Rev. Immunol. 17, 733–745 (2017).
Melsen, J. E., Lugthart, G., Lankester, A. C. & Schilham, M. W. Human circulating and tissue-resident CD56bright natural killer cell populations. Front. Immunol. 7, 262 (2016).
Schenkel, J. M. & Masopust, D. Tissue-resident memory T cells. Immunity 41, 886–897 (2014).
Vivier, E. et al. Innate lymphoid cells: 10 years on. Cell 174, 1054–1066 (2018).
Suzuki, T., Hayman, L., Kilbey, A., Edwards, J. & Coffelt, S. B. Gut γδ T cells as guardians, disruptors, and instigators of cancer. Immunol. Rev. 298, 198–217 (2020).
Raverdeau, M., Cunningham, S. P., Harmon, C. & Lynch, L. γδ T cells in cancer: a small population of lymphocytes with big implications. Clin. Transl. Immunology 8, e01080 (2019).
Gentles, A. J. et al. The prognostic landscape of genes and infiltrating immune cells across human cancers. Nat. Med. 21, 938–945 (2015).
Wu, D., Wu, P., Qiu, F., Wei, Q. & Huang, J. Human γδT-cell subsets and their involvement in tumor immunity. Cell. Mol. Immunol. 14, 245–253 (2017).
Wu, P. et al. γδT17 cells promote the accumulation and expansion of myeloid-derived suppressor cells in human colorectal cancer. Immunity 40, 785–800 (2014).
Holderness, J., Hedges, J. F., Ramstead, A. & Jutila, M. A. Comparative biology of γδ T cell function in humans, mice, and domestic animals. Annu. Rev. Anim. Biosci. 1, 99–124 (2013).
Hoeres, T., Smetak, M., Pretscher, D. & Wilhelm, M. Improving the efficiency of Vγ9Vδ2 T-cell immunotherapy in cancer. Front. Immunol. 9, 800 (2018).
Meraviglia, S. et al. Distinctive features of tumor-infiltrating γδ T lymphocytes in human colorectal cancer. Oncoimmunology 6, e1347742 (2017).
Dvorak, H. F. Tumors: wounds that do not heal. N. Engl. J. Med. 315, 1650–1659 (1986).
Dvorak, H. F. Tumors: wounds that do not heal-redux. Cancer Immunol. Res. 3, 1–11 (2015).
Pelka, K. et al. Spatially organized multicellular immune hubs in human colorectal cancer. Cell 184, 4734–4752 (2021).
Chan, C. J. et al. The receptors CD96 and CD226 oppose each other in the regulation of natural killer cell functions. Nat. Immunol. 15, 431–438 (2014).
Llosa, N. J. et al. The vigorous immune microenvironment of microsatellite instable colon cancer is balanced by multiple counter-inhibitory checkpoints. Cancer Discov. 5, 43–51 (2015).
Boland, R. C. & Goel, A. Microsatellite instability in colorectal cancer. Gastroenterology 138, 2073–2087 (2010).
Anitei, M.-G. et al. Prognostic and predictive values of the immunoscore in patients with rectal cancer. Clin. Cancer Res. 20, 1891–1899 (2014).
de Vries, N. L. et al. γδ T cells are effectors of immunotherapy in cancers with HLA class I defects. Nature 613, 743–750 (2023).
Almeida, A. R. et al. Delta one T cells for immunotherapy of chronic lymphocytic leukemia: clinical-grade expansion/differentiation and preclinical proof of concept. Clin. Cancer Res. 22, 5795–5804 (2016).
Poznanski, S. M. et al. Metabolic flexibility determines human NK cell functional fate in the tumor microenvironment. Cell Metab. 33, 1205–1220 (2021).
Lunt, S. Y. & Vander Heiden, M. G. Aerobic glycolysis: meeting the metabolic requirements of cell proliferation. Annu. Rev. Cell Dev. Biol. 27, 441–464 (2011).
Xu, S. et al. Uptake of oxidized lipids by the scavenger receptor CD36 promotes lipid peroxidation and dysfunction in CD8+ T cells in tumors. Immunity 54, 1561–1577 (2021).
Ma, X. et al. CD36-mediated ferroptosis dampens intratumoral CD8+ T cell effector function and impairs their antitumor ability. Cell Metab. 33, 1001–1012 (2021).
Combes, A. J. et al. Discovering dominant tumor immune archetypes in a pan-cancer census. Cell 185, 184–203 (2022).
Baginska, J. et al. The critical role of the tumor microenvironment in shaping natural killer cell-mediated anti-tumor immunity. Front. Immunol. 4, 490 (2013).
Bridgewater, J. A. et al. Systemic chemotherapy with or without cetuximab in patients with resectable colorectal liver metastasis (New EPOC): long-term results of a multicentre, randomised, controlled, phase 3 trial. Lancet Oncol. 21, 398–411 (2020).
Jonker, D. J. et al. Cetuximab for the treatment of colorectal cancer. N. Engl. J. Med. 357, 2040–2048 (2007).
Larson, R. C. et al. CAR T cell killing requires the IFNγR pathway in solid but not liquid tumours. Nature 604, 563–570 (2022).
Mayassi, T. et al. Chronic inflammation permanently reshapes tissue-resident immunity in celiac disease. Cell 176, 967–981 (2019).
Guo, X.-Z. J. et al. Lung γδ T cells mediate protective responses during neonatal influenza infection that are associated with type 2 immunity. Immunity. 49, 531–544 (2018).
Agaronyan, K. Tissue remodeling by an opportunistic pathogen triggers allergic inflammation. Immunity 55, 895–911 (2022).
Jin, C. et al. Commensal microbiota promote lung cancer development via γδ T cells. Cell 176, 998–1013 (2019).
Yu, X. et al. The surface protein TIGIT suppresses T cell activation by promoting the generation of mature immunoregulatory dendritic cells. Nat. Immunol. 10, 48–57 (2009).
Marin-Acevedo, J. A. et al. Next generation of immune checkpoint therapy in cancer: new developments and challenges. J. Hematol. Oncol. 11, 39 (2018).
Ge, Z., Peppelenbosch, M. P., Sprengers, D. & Kwekkeboom, J. TIGIT, the next step towards successful combination immune checkpoint therapy in cancer. Front. Immunol. 12, 699895 (2021).
Harjunpää, H. & Guillerey, C. TIGIT as an emerging immune checkpoint. Clin. Exp. Immunol. 200, 108–119 (2020).
Pizzolato, G. et al. Single-cell RNA sequencing unveils the shared and the distinct cytotoxic hallmarks of human TCRVδ1 and TCRVδ2 γδ T lymphocytes. Proc. Natl Acad. Sci. USA 116, 11906–11915 (2019).
Freud, A. G. et al. NKp80 defines a critical step during human natural killer cell development. Cell Rep. 16, 379–391 (2016).
Vitale, M. et al. Identification of NKp80, a novel triggering molecule expressed by human NK cells. Eur. J. Immunol. 31, 233–242 (2001).
Kuttruff, S. et al. NKp80 defines and stimulates a reactive subset of CD8 T cells. Blood 113, 358–369 (2009).
Di Lorenzo, B. et al. Broad cytotoxic targeting of acute myeloid leukemia by polyclonal delta one T cells. Cancer Immunol. Res. 7, 552–558 (2019).
Lenz, H.-J. et al. Impact of consensus molecular subtype on survival in patients with metastatic colorectal cancer: results from CALGB/SWOG 80405 (Alliance). J. Clin. Oncol. 37, 1876–1885 (2019).
Terrén, I. et al. Metabolic changes of interleukin-12/15/18-stimulated human NK cells. Sci. Rep. 11, 6472 (2021).
Cooper, M. A. et al. Cytokine-induced memory-like natural killer cells. Proc. Natl Acad. Sci. USA 106, 1915–1919 (2009).
Zhou, T. et al. IL-18BP is a secreted immune checkpoint and barrier to IL-18 immunotherapy. Nature 583, 609–614 (2020).
Scharping, N. E. et al. The tumor microenvironment represses T cell mitochondrial biogenesis to drive intratumoral T cell metabolic insufficiency and dysfunction. Immunity 45, 374–388 (2016).
Yu, Y.-R. et al. Disturbed mitochondrial dynamics in CD8+ TILs reinforce T cell exhaustion. Nat. Immunol. 21, 1540–1551 (2020).
Qiu, J. et al. Acetate promotes T cell effector function during glucose restriction. Cell Rep. 27, 2063–2074 (2019).
Buck, M. D., O’Sullivan, D. & Pearce, E. L. T cell metabolism drives immunity. J. Exp. Med. 212, 1345–1360 (2015).
Michelet, X. et al. Metabolic reprogramming of natural killer cells in obesity limits antitumor responses. Nat. Immunol. 19, 1330–1340 (2018).
Krishnan, S. et al. Amphiregulin-producing γδ T cells are vital for safeguarding oral barrier immune homeostasis. Proc. Natl Acad. Sci. USA 115, 10738–10743 (2018).
Zaiss, D. M. W., Gause, W. C., Osborne, L. C. & Artis, D. Emerging functions of amphiregulin in orchestrating immunity, inflammation, and tissue repair. Immunity 42, 216–226 (2015).
Liu, J. et al. An integrated TCGA pan-cancer clinical data resource to drive high-quality survival outcome analytics. Cell 173, 400–416 (2018).
Therneau, T., Atkinson, B., & Ripley, B. Rpart: recursive partitioning. R package version 4.1-3 (2013).
Therneau, T. A package for survival analysis in R. R package version 3.5-5 (2023).
Maykel, J. et al. NOD-scidIl2rg tm1Wjl and NOD-Rag1 null Il2rg tm1Wjl: a model for stromal cell–tumor cell interaction for human colon cancer. Dig. Dis. Sci. 59, 1169–1179 (2014).
Aryee, K.-E. et al. Enhanced development of functional human NK cells in NOD-scid-IL2rgnull mice expressing human IL15. FASEB J. 36, e22476 (2022).
Katano, I. et al. Long-term maintenance of peripheral blood derived human NK cells in a novel human IL-15- transgenic NOG mouse. Sci. Rep. 7, 17230 (2017).
Acknowledgements
This work was supported by National Institutes of Health grant no. 5R01AI134861 and a BWH IGNITE Grant and NIH R24 OD026440 (to M.B). D.B. acknowledges funding from Science Foundation Ireland under the Strategic Research Programme Precision Oncology Ireland (18/SPP/3522), the Ireland East Hospital Group and the National Maternity Hospital Foundation. Biobanking activities at UCD-GOG are supported by the UCD Clinical Research Centre. We thank our collaborators in the Evergrande Flagship Project for early access to the scRNA-seq datasets. We thank our patients and their families for participating in this research project.
Author information
Authors and Affiliations
Contributions
C.H. and L.L. conceptualized the study. C.H., H.K., M.A.B. and L.L. devised the methodology. C.H., H.M., H.K., B.M. and J.S. were responsible for the software. H.M. and K.S. validate the data. C.H., H.M., J.S., B.M. and L.L. carried out the formal analysis. C.H., H.M., A.Z., P.S.L., K.S., D.D., B.K., C.L.M. and A.N.S. carried out the investigation. A.C.A., D.W., D.B., M.A.B. and L.L. managed the resources. C.H., H.M., A.Z., D.W. and D.B. curated the data. C.H. and L.L. wrote the original paper. C.H., H.M., D.W., D.B., M.A.B. and L.L. reviewed and edited the paper. C.H., H.M. and L.L. visualized the data. M.A.B. and L.L. supervised the study. L.L. was responsible for project administration. D.W., D.B., M.A.B. and L.L. were responsible for funding.
Corresponding author
Ethics declarations
Competing interests
L.L. is a member of the scientific advisory board for MiNK Therapeutics and a consultant for Bayer. A.C.A. is a member of the scientific advisory board for Tizona Therapeutics, Trishula Therapeutics, Compass Therapeutics, Zumutor Biologics, ImmuneOncia and Nekonal Sarl. A.C.A. is also a paid consultant for iTeos Therapeutics, Larkspur Biosciences and ExcepGen. The interests of L.L. and A.C.A. were reviewed and managed by the Brigham and Women’s Hospital and Partners Healthcare in accordance with their conflict-of-interest policies. The other authors declare no competing interests.
Peer review
Peer review information
Nature Cancer thanks Florian Greten and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Prognostic value of Vd1 T cells in solid tumors.
(a-d) Progression free survival analysis of ovarian (OV), lung (LUAD), endometrial (UCEC) and colorectal (COAD) cancer patients from the TCGA database, stratified by high and low expression of γδ TCR genes TRDV1 & TRDV2. (e) Absolute numbers of Vδ1 and Vδ2T cells in endometrial tumors and healthy tissue (f) Absolute numbers of Vδ1 and Vδ2T cells in colon tumors and healthy tissue. Data presented as mean ± SEM. Data were analyzed using Cox Proportional Hazards Model (A-D), Wilcoxon matched pairs test two tailed (E-F). A–D n = 379-575, E-F n = 5 patients, * p < 0.05.
Extended Data Fig. 2 Identification of clusters.
scRNAseq was performed 7 CRC patients using the 10x platform. Data was analyzed in R using the Seurat package. (a) Gating strategy for the isolation of NK cells, γδ T cells, MAIT cells and adaptive T cells. (b) Top 20 differentially expressed genes in clusters from CRC samples. (c) Lineage markers used to identify clusters.
Extended Data Fig. 3 Expression of effector molecules, immune checkpoints, & cytotoxicity receptors.
(a) Heatmap of effector gene expression in cell clusters from CRC samples. (b) Heatmap of immune checkpoint expression in cell clusters from CRC samples. (c) Heatmap of cytotoxicity receptor expression in cell clusters from CRC samples. (d) Production of IL17A in healthy colon and tumor γδ T cell subsets after PMA stimulation. Data presented as mean ± SEM. Data were analyzed using Friedman test, with Dunn’s multiple comparison test two tailed (n = 6 (Vδ3), 12 (Vδ2), 13 (Vδ1) patients, * p < 0.05, ** p < 0.01, ** p < 0.001).
Extended Data Fig. 4 γδ T cells are not the major source of AREG in healthy colon, healthy endometrium, or endometrial tumors.
scRNAseq was performed on 2 endometrial tumors, 2 healthy endometrial samples and 5 healthy colon samples using the 10x platform. Data was analyzed in R using the Seurat package. (a) UMAP representation of cell clusters from endometrial tumors. (b) Violin plots of AREG expression in cell clusters from endometrial tumors. (c) UMAP representation of cell clusters from healthy endometrium. (d) Violin plots of AREG expression in cell clusters from healthy endometrium. (e) UMAP representation of cell clusters from healthy colon. (f) Violin plots of AREG expression in cell clusters from healthy colon.
Extended Data Fig. 5 Cell types expressing AREG in colorectal cancer patients.
(a) Proportion of cells in each cluster which express AREG in colorectal cancer. Data presented as mean ± SEM.
Extended Data Fig. 6 γδ T cells in individual patients from CRC cohort.
(a) Absolute number of γδ T cells from each patient in the Flagship CRC cohort (MMRp-red, MMRd-black). (b) Proportion of γδ T cells in lymphocyte clusters in each patient in the Flagship CRC cohort (MMRp-red, MMRd-black). (c) Proportion of γδ T cells expressing AREG in each patient in the Flagship CRC cohort (MMRp-orange, MMRd-blue) (n = 62).
Extended Data Fig. 7 Analysis of γδ subsets in CRC.
scRNAseq Data was analyzed from deVries et al. 2023. (a, b) γδ T cells were analyzed and subsetted on Vδ1T cells by expression of the TRDV1 gene. (c) AREG and IFNG gene expression in TRDV1+ cells. (d) Correlation between the expression of AREG and IFNG in TRDV1+ cells. scRNAseq was performed 7 CRC patients using the 10x platform. Data was analyzed in R using the Seurat package. (e) Expression of immune checkpoint molecules in γδ T cell subsets from CRC samples. (f) Expression of activatory receptors in γδ T cell subsets from CRC samples. (g) Correlation plots of KRLF1, TIGIT & AREG in γδ T cells in Flagship CRC dataset.
Extended Data Fig. 8 Expanded Vδ1T cells promote growth of HCT116 colon cells through AREG production.
Vδ1T cells were expanded using a published protocol. (a) Expression of NKp80 & TIGIT pre- and post-expansion. A scratch assay was performed using HCT116 cells with or without 1 × 105 Vδ1T cells for 24hrs. (b) Percentage of scratch area at 0, 6 & 24 hours, untreated or treated with Vδ1T cells. (c) Percentage of Vδ1T cells expressing AREG after 24 hours co-incubation with scratched and unscratched HCT116 cells. (d) Concentration of AREG in supernatants of HCT116 cells without Vδ1 treatment (Control) or with Vδ1T cells with or without cell scratching. (e) Representative images of scratch assay using SW480 treated with Vδ1T cells ± cetuximab (αEGFR), aIL1β, or αTCRγδ (3 independent experiments). Data presented as mean ± SEM. Data were analyzed using Wilcoxon matched pairs test two tailed or Friedman test, with Dunn’s multiple comparison test two tailed (A n = 6 donors, B n = 8 donors, C n = 5 donors, D n = 3 donors, * p < 0.05, **, p < 0.01).
Extended Data Fig. 9 Optimization of Vδ1T cell expansion.
Isolated Vd1 T cells were cultured for 7days with aCD3 ± Il1b, IL21, IFNγ, IL4 or IL15. Cells were then stimulated for 4 hours with PMA (a) Percentage of Vd1 T cells producing IFNγ. γδ T cells were isolated by positive selection MACS and cultured for 21 days in varying conditions using a combination of cytokines, including IL-2, IL-12, IL-15, IL-18, αCD3 and αCD2. (b) Growth curves of Vδ1T cells using cytokine cocktail based on IL-12/IL-15 combination. (c) Growth curves of Vδ1T cells using cytokine cocktail based on IL-12/IL-18 combination. (d) Growth curves of Vδ1T cells using cytokine cocktail based on IL-15/IL-18 combination. (e) Growth curves of Vδ1T cells using cytokine cocktail based on IL-12/IL-15/IL-18 combination. Conditions which resulted in cell expansion were assessed for cell phenotype. (f) Percentage of live cells. (g) Percentage of Vδ1T cells. (h-k) Percentage of TIGIT, LAG3, CTLA4, TIM3 positive cells. Data presented as mean ± SEM. Data were analyzed using Friedman test, with Dunn’s multiple comparison test two tailed. (A n = 7 donors, B-K n = 2 donors, * p < 0.05, **, p < 0.01,).
Extended Data Fig. 10 Gen 1 cells are functionally distinct from Gen 2 cells, and Gen 2 cells prevent tumor growth in vitro.
RNAseq was performed on unexpanded Vδ1T cells, Gen 1, and Gen 2 cells, with and without PMA stimulation for 4hrs. (a) Heatmap of top 100 genes expressed in Vδ1 T cells, Gen 1 & Gen 2 cells with and without stimulation. (b) KEGG pathway analysis of differentially expressed gene between Gen 1 and Gen 2 cells. (c) Expression of genes associated with tissue residency and cell trafficking (n = 2). Data presented as mean ± SEM. (d) Percentage of scratch area of HCT116 cells imaged after 6 or 24 hours coincubation with Gen 1 or Gen 2 cells. (e) Percentage of Vδ1T cells expressing degranulation marker CD107a after 24-hour co-incubation with HCT116 cells. (f) Percentage of Vδ1T cells producing IFNγ after 24-hour co-incubation with HCT116 cells. (g) Percentage of Vδ1T cells producing AREG after 24-hour co-incubation with HCT116 cells. Data presented as mean ± SEM. Data were analyzed using Wilcoxon matched pairs test two tailed or Friedman test, with Dunn’s multiple comparison test two tailed (A n = 8 donors, B n = 5 donors, C n = 6 donors, D n = 5 donors, * p < 0.05, **, p < 0.01).
Supplementary information
Source data
Source Data Fig. 1
FACS data.
Source Data Fig. 2
FACS data.
Source Data Fig. 3
FACS data.
Source Data Fig. 4
Gene expression and FACS data.
Source Data Fig. 5
FACS data and scratch assay image analysis data.
Source Data Fig. 6
Cell counts, FACS data and scratch assay image analysis data.
Source Data Fig. 7
FACS data, Seahorse data and gene expression.
Source Data Fig. 8
Tumor size and FACS data.
Source Data Extended Data Fig. 1
Gene expression and FACS data.
Source Data Extended Data Fig. 2
Gene expression data.
Source Data Extended Data Fig. 3
Gene expression and FACS data.
Source Data Extended Data Fig. 4
Gene expression data.
Source Data Extended Data Fig. 5
Gene expression data.
Source Data Extended Data Fig. 6
Gene expression data.
Source Data Extended Data Fig. 7
Gene expression data.
Source Data Extended Data Fig. 8
FACS, scratch assay image analysis and ELISA data.
Source Data Extended Data Fig. 9
FACS and cell count data.
Source Data Extended Data Fig. 10
Gene expression, FACS and scratch assay image analysis data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Harmon, C., Zaborowski, A., Moore, H. et al. γδ T cell dichotomy with opposing cytotoxic and wound healing functions in human solid tumors. Nat Cancer 4, 1122–1137 (2023). https://doi.org/10.1038/s43018-023-00589-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s43018-023-00589-w
This article is cited by
-
CTLs heterogeneity and plasticity: implications for cancer immunotherapy
Molecular Cancer (2024)
-
γδ T cells as unconventional targets of checkpoint blockade
Nature Cancer (2024)
-
PD-1 defines a distinct, functional, tissue-adapted state in Vδ1+ T cells with implications for cancer immunotherapy
Nature Cancer (2024)
-
The two sides of the γδ T cell coin
Nature Cancer (2023)