Abstract
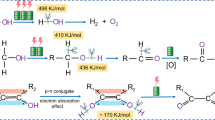
In recent years, electrocatalytic systems powered by renewable energy have gained prominence in regard to sustainable chemical production. A majority of published literature focuses on catalyst development in alkaline electrolytes, driven by advantages such as high ionic conductivity, low charge transfer resistance and rapid kinetics. Here we shed light on challenges arising from the use of alkaline electrolytes for product separation and electrolyte recovery. Delving into acid–base reaction chemistry, we identify the problematic synthesis of organic acids whereby the high-pH environment leads to dissociation or deprotonation, forming conjugate bases and water. Our analysis of an alkaline electrochemical process for glycerol oxidation highlights the significant economic hurdles, with >60% of capital costs, 70% of raw material (for example, potassium hydroside) costs and 64% of total energy costs attributed to downstream product separation. These challenges, related to acid–base reaction chemistry, are often overlooked at the catalyst development stage, resulting in a significant waste of research resources.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Data that support the findings of this study are provided with the manuscript and its Supplementary Information file. All data used in this study are available from the corresponding author on reasonable request.
References
Climate Action. United Nations www.un.org/en/climatechange/net-zero-coalition
Chemicals. International Energy Agency (IEA) www.iea.org/energy-system/industry/chemicals
Davis, S. J. et al. Net-zero emissions energy systems. Science 360, eaas9793 (2018).
De Luna, P. et al. What would it take for renewably powered electrosynthesis to displace petrochemical processes? Science 364, eaav3506 (2019).
Khan, M. A. et al. Techno-economic analysis of a solar-powered biomass electrolysis pathway for coproduction of hydrogen and value-added chemicals. Sustain. Energy Fuels 4, 5568–5577 (2020).
Jouny, M., Luc, W. & Jiao, F. General techno-economic analysis of CO2 electrolysis systems. Ind. Eng. Chem. Res. 57, 2165–2177 (2018).
Kim, H. J. et al. Coproducing value-added chemicals and hydrogen with electrocatalytic glycerol oxidation technology: experimental and techno-economic investigations. ACS Sustain. Chem. Eng. 5, 6626–6634 (2017).
Zhong, C. et al. A review of electrolyte materials and compositions for electrochemical supercapacitors. Chem. Soc. Rev. 44, 7484–7539 (2015).
Mahmood, N. et al. Electrocatalysts for hydrogen evolution in alkaline electrolytes: mechanisms, challenges, and prospective solutions. Adv. Sci. 5, 1700464 (2018).
Gu, J. et al. Modulating electric field distribution by alkali cations for CO2 electroreduction in strongly acidic medium. Nat. Catal. 5, 268–276 (2022).
Chorkendorff, I. & Niemantsverdriet, J. W. Concepts of Modern Catalysis and Kinetics (John Wiley & Sons, 2017); https://doi.org/10.1002/3527602658.fmatter
Li, T. & Harrington, D. A. An overview of glycerol electrooxidation mechanisms on Pt, Pd and Au. ChemSusChem 14, 1472–1495 (2021).
Gomes, J. F., Martins, C. A., Giz, M. J., Tremiliosi-Filho, G. & Camara, G. A. Insights into the adsorption and electro-oxidation of glycerol: self-inhibition and concentration effects. J. Catal. 301, 154–161 (2013).
Wang, L. et al. Deactivation of palladium electrocatalysts for alcohols oxidation in basic electrolytes. Electrochim. Acta 177, 100–106 (2015).
Araujo, H. R. et al. How the adsorption of Sn on Pt (100) preferentially oriented nanoparticles affects the pathways of glycerol electro-oxidation. Electrochim. Acta 297, 61–69 (2019).
Lin, J. L., Ren, J., Tian, N., Zhou, Z. Y. & Sun, S. G. In situ FTIR spectroscopic studies of ethylene glycol electrooxidation on Pd electrode in alkaline solution: the effects of concentration. J. Electroanal. Chem. 688, 165–171 (2013).
Schnaidt, J. W. Electrooxidation of C2 and C3 Molecules Studied By Combined In Situ ATR-FTIRS and Online DEMS. PhD Dissertation (Ulm University, 2013); https://oparu.uni-ulm.de/xmlui/handle/123456789/2625
Rafaïdeen, T., Baranton, S. & Coutanceau, C. Highly efficient and selective electrooxidation of glucose and xylose in alkaline medium at carbon supported alloyed PdAu nanocatalysts. Appl. Catal. B Environ. 243, 641–656 (2019).
Guo, J., Chen, R., Zhu, F. C., Sun, S. G. & Villullas, H. M. New understandings of ethanol oxidation reaction mechanism on Pd/C and Pd2Ru/C catalysts in alkaline direct ethanol fuel cells. Appl. Catal. B Environ. 224, 602–611 (2018).
Li, J. et al. Constraining CO coverage on copper promotes high-efficiency ethylene electroproduction. Nat. Catal. 2, 1124–1131 (2019).
El-Nowihy, G. H., Mohammad, A. M., Sadek, M. A., Khalil, M. M. H. & El-Deab, M. S. EIS-activity correlation for the electro-oxidation of ethylene glycol at nanoparticles-based electrocatalysts. J. Electrochem. Soc. 166, F364–F376 (2019).
Spendelow, J. S., Goodpaster, J. D., Kenis, P. J. A. & Wieckowski, A. Mechanism of CO oxidation on Pt(111) in alkaline media. J. Phys. Chem. B 110, 9545–9555 (2006).
Gómez-Marín, A. M. & Hernández-Ortiz, J. P. Langmuir–Hinshelwood mechanism including lateral interactions and species diffusion for CO electro-oxidation on metallic surfaces. J. Phys. Chem. C Nanometer. Interfaces 118, 2475–2486 (2014).
Thomas, F. & Fuller, J. N. H. Electrochemical Engineering (John Wiley & Sons, 2018).
Lind, E. Influence of Mass Transport on Glycerol Electrooxidation on Palladium in Alkaline Media (KTH Royal Institute of Technology, 2022).
Yang, Y. & Mu, T. Electrochemical oxidation of biomass derived 5-hydroxymethylfurfural (HMF): pathway, mechanism, catalysts and coupling reactions. Green Chem. 23, 4228–4254 (2021).
Morgan, A., Kavanagh, R., Lin, W. F., Hardacre, C. & Hu, P. Electrooxidation of methanol in an alkaline fuel cell: determination of the nature of the initial adsorbate. Phys. Chem. Chem. Phys. 15, 20170–20175 (2013).
Kwon, Y.; Lai, S. C.; Rodriguez, P.; Koper, M. T., Electrocatalytic oxidation of alcohols on gold in alkaline media: base or gold catalysis? J. Am. Chem. Soc. 133, 6914–6917 (2011).
Beyhan, S., Uosaki, K., Feliu, J. M. & Herrero, E. Electrochemical and in situ FTIR studies of ethanol adsorption and oxidation on gold single crystal electrodes in alkaline media. J. Electroanal. Chem. 707, 89–94 (2013).
Valter, M., Wickman, B. & Hellman, A. Solvent effects for methanol electrooxidation on gold. J. Phys. Chem. C Nanometer. Interfaces 125, 1355–1360 (2021).
Smith, M. B. Organic Chemistry: An Acid–Base Approach (CRC, 2022); https://doi.org/10.1201/9781003174929
Lower, S. K. Introduction to Acid–Base Chemistry (Simon Fraser Univ., 1999); www.chem1.com/acad/pdf/c1xacid1.pdf
Clayden, J., Greeves, N. & Warren, S. Organic Chemistry (Oxford Univ. Press, 2012).
March, J. et al. Advanced Organic Chemistry: Reactions, Mechanisms, and Structure (McGraw-Hill, 1977).
Na, J. et al. General technoeconomic analysis for electrochemical coproduction coupling carbon dioxide reduction with organic oxidation. Nat. Commun. 10, 5193 (2019).
Murcia Valderrama, M. A., van Putten, R. J. & Gruter, G. J. M. The potential of oxalic – and glycolic acid based polyesters (review). Towards CO2 as a feedstock (Carbon Capture and Utilization – CCU). Eur. Polym. J. 119, 445–468 (2019).
Zhou, X. et al. Glycolic acid production from ethylene glycol via sustainable biomass energy: integrated conceptual process design and comparative techno-economic–society–environment analysis. ACS Sustain. Chem. Eng. 9, 10948–10962 (2021).
Chen, Y. et al. Sustainable production of formic acid and acetic acid from biomass. Mol. Catal. 545, 113199 (2023).
Kaiser, D., Beckmann, L., Walter, J. & Bertau, M. Conversion of green methanol to methyl formate. Catalysts 11, 869 (2021).
Reksten, A. H., Thomassen, M. S., Møller-Holst, S. & Sundseth, K. Projecting the future cost of PEM and alkaline water electrolysers; a CAPEX model including electrolyser plant size and technology development. Int. J. Hydrog. Energy 47, 38106–38113 (2022).
The Future of Hydrogen (International Energy Agency, 2019); https://doi.org/10.1787/1e0514c4-en
Ruth, M., Mayyas, A. & Mann, M. Manufacturing competitiveness analysis for PEM and alkaline water electrolysis systems (2017); www.nrel.gov/docs/fy19osti/70380.pdf
Boukil, R. et al. Enhanced electrocatalytic activity and selectivity of glycerol oxidation triggered by nanoalloyed silver-gold nanocages directly grown on gas diffusion electrodes. J. Mater. Chem. A Mater. 8, 8848–8856 (2020).
Khan, M. A. et al. Zero-crossover electrochemical CO2 reduction to ethylene with co-production of valuable chemicals. Chem. Catal. 2, 2077–2095 (2022).
Yadegari, H. et al. Glycerol oxidation pairs with carbon monoxide reduction for low-voltage generation of C2 and C3 product streams. ACS Energy Lett. 6, 3538–3544 (2021)
Pookpunt, S., Ongsakul, W. & Madhu, N. A comprehensive techno-economic analysis for optimally placed wind farms. Electr. Eng. 102, 2161–2179 (2020).
Sukor, N. R., Shamsuddin, A. H., Mahlia, T. M. I. & Isa, M. F. M. Techno-economic analysis of CO2 capture technologies in offshore natural gas field: Implications to carbon capture and storage in Malaysia. Processes 8, 350 (2020).
H2A: hydrogen analysis production models. NREL www.nrel.gov/hydrogen/h2a-production-models.html (2018).
Pérez-Fortes, M., Schöneberger, J. C., Boulamanti, A., Harrison, G. & Tzimas, E. Formic acid synthesis using CO2 as raw material: techno-economic and environmental evaluation and market potential. Int. J. Hydrog. Energy 41, 16444–16462 (2016).
Kim, D. & Han, J. Comprehensive analysis of two catalytic processes to produce formic acid from carbon dioxide. Appl. Energy 264, 114711 (2020).
Potassium Sulfate Market. Allied Market Research www.alliedmarketresearch.com/potassium-sulfate-market-A49194 (2023).
Potassium sulfate market set to achieve USD 10.8 billion revenue by 2031 with 4.9% CAGR – TMR study. GlobeNewsWire www.globenewswire.com/news-release/2023/05/29/2677921/0/en/Potassium-Sulfate-Market-Set-to-Achieve-USD-10-8-Billion-Revenue-by-2031-with-4-9-CAGR-TMR-Study.html (2023).
Jain, R. M., Mody, K. H., Keshri, J. & Jha, B. Biological neutralization and biosorption of dyes of alkaline textile industry wastewater. Mar. Pollut. Bull. 84, 83–89 (2014).
Knexevic, K., Saracevic, E., Krampe, J. & Kreuzinger, N. Comparison of ion removal from waste fermentation effluent by nanofiltration, electrodialysis and ion exchange for a subsequent sulfuric acid recovery. J. Environ. Chem. Eng. 10, 108423 (2022).
Kim, Y. et al. The role of ruthenium on carbon-supported PtRu catalysts for electrocatalytic glycerol oxidation under acidic conditions. ChemCatChem 9, 1683–1690 (2017).
de Gyves, J. et al. Enhanced performance of glycerol electro-oxidation in alkaline media using bimetallic Au–Cu NPs supported by MWCNTs and reducible metal oxides. Front. Chem. 11, 1–18 (2023).
Yuda, A., Ashok, A. & Kumar, A. A comprehensive and critical review on recent progress in anode catalyst for methanol oxidation reaction. Catal. Rev. Sci. Eng. 64, 126–228 (2022).
Thia, L. et al. Copper-modified gold nanoparticles as highly selective catalysts for glycerol electro-oxidation in alkaline solution. ChemCatChem 8, 3272–3278 (2016).
Al-Attas, T. et al. Bioinspired multimetal electrocatalyst for selective methane oxidation. Chem. Eng. J. 474, 145827 (2023).
Vo, T.-G., Ho, P.-Y. & Chiang, C.-Y. Operando mechanistic studies of selective oxidation of glycerol to dihydroxyacetone over amorphous cobalt oxide. Appl. Catal. B Environ. 300, 120723 (2022).
Moklis, M. H., Shuo, C., Boonyubol, S. & Cross, J. S. Electrochemical valorization of glycerol via electrocatalytic reduction into biofuels: a review. ChemSusChem 26, e202300990 (2023).
Zhu, P. & Wang, H. High-purity and high-concentration liquid fuels through CO2 electroreduction. Nat. Catal. 4, 943–951 (2021).
She, X., Wang, Y., Xu, H., Chi Edman Tsang, S. & Ping Lau, S. Challenges and opportunities in electrocatalytic CO2 reduction to chemicals and fuels. Angew. Chem. Int. Ed. Engl. 61, e202211396 (2022) .
Chen, C., Li, Y. & Yang, P. Address the ‘alkalinity problem’ in CO2 electrolysis with catalyst design and translation. Joule 5, 737–742 (2021).
Rabinowitz, J. A. & Kanan, M. W. The future of low-temperature carbon dioxide electrolysis depends on solving one basic problem. Nat. Commun. 11, 10–12 (2020).
Xie, Y. et al. High carbon utilization in CO2 reduction to multi-carbon products in acidic media. Nat. Catal. 5, 564–570 (2022).
Acknowledgements
M.A.K. acknowledges funds received through Office of the Dean, Faculty of Engineering, University of Alberta in assistance with research-related expenditures. S.K.N. thanks Alberta Innovates and the Government of Alberta for their support through graduate scholarships.
Author information
Authors and Affiliations
Contributions
S.K.N. and M.A.K. conducted formal economic analysis. M.A.M.R. performed the literature review. S.K.N. and M.A.M.R. summarized the data and cowrote the first draft of the manuscript. K.K. helped with manuscript writing. S.K.N. and K.K. further revised the manuscript. M.R. and H.S.S. helped with literature review and manuscript editing. M.G.K. and M.A.K. reviewed and further edited the manuscript. M.A.K. conceptualized the work. M.G.K. and M.A.K. supervised all aspects of the work.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Catalysis thanks Miguel Modestino and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Tables 1–19, Note 1 and Figs. 1–3.
Rights and permissions
About this article
Cite this article
Nabil, S.K., Muzibur Raghuman, M.A., Kannimuthu, K. et al. Acid–base chemistry and the economic implication of electrocatalytic carboxylate production in alkaline electrolytes. Nat Catal 7, 330–337 (2024). https://doi.org/10.1038/s41929-024-01107-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41929-024-01107-6