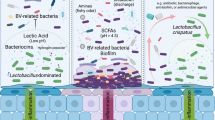
Abstract
Bacterial vaginosis (BV) is a common vaginal dysbiosis characterized by a malodorous discharge and irritation. The imbalance of the vaginal microbiota plays a key role in the development of BV. It has been demonstrated that Gardnerella vaginalis (GV), a facultative anaerobic bacillus, is involved in BV. Due to the rising number of antimicrobial-resistant species, recurrence of BV is becoming more frequent in women; thus, alternative treatments to antibiotics are needed. Natural substances have recently shown a great efficacy for the treatment of vaginal dysbiosis. Thus, this study aimed to investigate the beneficial effect of a product containing pea protein (PP), grape seed extract (GS) and lactic acid (LA) in an in vivo model of Gardnerella vaginalis-induced vaginosis by intravaginal administration of GV suspension (1 × 106 CFU/20 µL saline). Our results demonstrated that the product containing PP, GS and LA significantly reduced GV proliferation. More specifically, it significantly preserved tissue architecture and reduced neutrophil infiltration, inflammatory markers and sialidase activity when used both as a pre- or a post-treatment. Moreover, the product displayed strong bioadhesive properties. Therefore, our data suggested that the product containing PP, GS and LA could be used as alternative preventive or curative treatment for the management of BV.
Similar content being viewed by others
Introduction
Bacterial vaginosis (BV) is the most common vaginal dysbiosis in women of childbearing age1, and it is estimated to occur in 5–70% of women1,2. BV is caused by an imbalance of the vaginal microbiota2 and is characterized by a thin, gray/off-white, malodorous adherent vaginal discharge3,4. The abnormal vaginal discharge results in part from the degradation of the protective vaginal mucin gel, induced by mucin-degrading enzymes produced by BV-associated bacteria5. BV has been associated with urinary tract infections, higher risk of sexually transmitted infections, infertility, preterm birth, intrauterine and intraamniotic infections, as well as cervical infections, dysplasia, and cancer6,7,8,9. It has been demonstrated that vaginal microbiota of healthy women is composed mainly of lactobacilli which maintain the acidic milieu of the vagina through lactic acid production inhibiting the growth of harmful pathogens10. Alteration of the vaginal microbiota causes change in pH, which allows a variety of anaerobes and facultative bacteria to overgrow and cause chronic infection as well as abnormal vaginal discharge11,12. Bacteria that colonize vaginal mucosa are mainly Gram-positive cocci or anaerobic species such as Prevotella13, Mobiluncus14, and the facultative anaerobic bacillus Gardnerella vaginalis (GV)15. Particularly, GV has been extensively studied because it has been found from the vaginal samples of almost all women with BV16,17,18. Over the past decades, several classifications have been developed to delineate subgroups or “clades” of GV19. In 2019, Vaneecoutte et al.20 identified three other BV-associated Gardnerella species based on whole genome sequence comparison, G. leopoldii, G. piotii, and G. swidsinskii. Plummer and colleagues21 revealed that G. leopoldii, and G. swidsinskii were not strongly associated with BV or the absence of Lactobacillus, supporting the existence of symbiotic and pathogenic subtypes of Gardnerella spp.22. Nevertheless, a recent study conducted by Hill et al.23 demonstrated that a higher abundance of G. swidsinskii is associated to several BV symptoms, including abnormal and malodourous vaginal discharge. BV is characterized by a polymicrobial biofilm, a community of microorganisms primarily constituted by GV clusters, strongly adhered to the vaginal epithelium24. The interactions between bacteria in multi-species biofilms contribute to enhance antimicrobial tolerance as standard antibiotics are unable to completely eradicate the vaginal biofilm, leading to relapses24. As biofilms allow Gardnerella spp. to persist in recurrent BV, it is important to identify new treatments capable to counteract BV-biofilm24,25. Currently, the therapeutic agents available for the management of BV are topical agents containing lactic acid, clindamycin and/or orally administered antibiotics26, among others. However, oral administration of clindamycin (CLIN) and/or metronidazole (MET) long-term could negatively impact the gut microbiome and increase BV recurrence27, due to the rising number of antimicrobial resistant species28; therefore, alternative treatments are needed for a better management of BV26,29. In the last decade, more attention was given to the use of natural substances for the treatment of vaginal dysbiosis, including BV25,30,31,32. Pea protein (PP) is derived from the plant of Pisum sativum; it is rich in fiber and represents an important source of essential amino acids such as lysine33. PP has shown to exert several health benefits including protective action on mucosal tissues34. Grape seed extract (GS) has gained great interest since it is rich in antioxidants and oligomeric proanthocyanidins34, while lactic acid (LA) is well-known for its ability to maintain the physiological vaginal pH and regulate the vaginal immune response10. Based on these findings, the aim of this study was to evaluate the efficacy of a product containing PP, GS and LA in an in vivo model of GV-induced bacterial vaginosis.
Results
Effect of PP, GS and LA on GV adhesion on vaginal tissue
At the end of experiment, serial dilutions of vaginal washes onto NYC-III agar plates containing 1 mg/mL streptomycin and 4 mg/L amphotericin (both from Sigma- Aldrich) were plated to evaluate GV counts. Both the pre- and post-treatment with PP, GS and LA were significantly effective in reducing bacterial counts compared to the GV group. Notably, the pre-treatment was more effective in treating the infection reaching a reduction of the bacterial count by 35.85% compared to the post-treatment group which showed a reduction of 11%. (Fig. 1A, B).
Effect of the product containing PP, GS, and LA on % CFU evaluation. The treatment with the product containing PP, GS and LA was able to reduce GV growth in a pre-treatment condition (A) and also in post-treatment condition (B). (A) ***p < 0.001 vs Sham; ###p < 0.001 vs GV group; (B) ***p < 0.001 vs Sham; #p < 0.05 vs GV group.
Effect of PP, GS and LA on histological damage induced by GV infection
Histological examination of vaginal tissues revealed characteristic pathological changes 7 days after infection (Figs. 2C, 3C), compared to sham group (Figs. 2A, 3A). The product containing PP, GS and LA was able to significantly preserve tissue architecture both as a pre-treatment (Fig. 2D) and post-treatment (Fig. 3D). No significance difference was observed in the sham groups treated with PP, GS and LA (Figs. 2B, 3B). However, the decrease in the histological score was more evident in the pre-treatment group (Fig. 2E) compared to post-treatment group (Fig. 3E).
Effect of the product containing PP, GS, and LA on histological damage in pre-treatment condition. The treatment with the product containing PP, GS and LA (D) given 1 h before GV infection was able to significantly preserve tissue architecture compared to GV group (C). No significance difference was observed in the sham group treated with PP, GS and LA group (B) compared to sham group alone (A). (E) ***p < 0.001 vs Sham; ###p < 0.001 vs GV group.
Effect of the product containing PP, GS, and LA on histological damage in post-treatment condition. The treatment with the product containing PP, GS and LA (D) was able to reduce histological damage post GV infection (C). No significance difference was observed in the sham group treated with PP, GS and LA (B) compared to sham group alone (A). (E) ***p < 0.001 vs Sham; #p < 0.05 vs GV group.
Effect of PP, GS and LA on myeloperoxidase activity of GV infection
MPO activity, an index of polymorfonucleate infiltration associated to inflammation, was evaluated in vaginal tissue homogenates from GV-infected mice. Our results demonstrated that the GV-infected mice group was characterized by an elevated MPO activity at 7 days post-infection. However, the product containing PP, GS and LA was able to significantly decrease MPO activity in pre-treatment condition as shown in the Fig. 4A. Additionally, our data demonstrated that the product containing PP, GS and LA was able to reduce MPO activity post GV infection, as shown in the Fig. 4B.
Effect of the product containing PP, GS and LA on MPO activity. The product containing PP, GS and LA significantly decreased MPO activity in pre-treatment condition compared to GV group (A). Moreover, the product containing PP, GS and LA was able to reduce MPO activity post GV infection (B). (A) ***p < 0.001 vs Sham; ###p < 0.001 vs GV group. (B)***p < 0.001 vs Sham; #p < 0.05 vs GV group.
Effect of PP, GS, and LA on sialidase activity of GV infection
Sialidase activity is an important hallmark feature of bacterial vaginosis35 and was evaluated using vaginal washes from mice. The obtained results showed that the pre-treatment with the product containing PP, GS and LA was able to significantly inhibit sialidase enzyme activity compared to GV infected mice group as shown in the Fig. 5. Moreover, we evaluated the effect of the product containing PP, GS and LA on sialidase activity also after GV infection, showing that PP, GS and LA significantly decreased sialidase enzyme activity compared to GV infected mice group (Fig. 5).
Effect of the product containing PP, GS and LA on sialidase activity. The product containing PP, GS and LA significantly decreased sialidase enzyme activity compared to GV infected mice group in pre-treatment and post-treatment conditions. ***p < 0.001 vs Sham; #p < 0.05 vs GV group; ##p < 0.01 vs GV group.
Effect of PP, GS and LA on mucoadhesion of GV infection
The everted vaginal sac assay evaluated the bioadhesive interaction of the cream with everted vaginal tissue in infected and non-infected mice. The results were presented as percent binding, which was derived from subtracting the weight of the washed and lyophilized cream particles remaining in the tube after incubation from the original tare weight of the cream particles. A high percentage of binding indicated strong bioadhesion of the particles to mucosal tissue. The treatment with a product containing PP, GS and LA showed a significant binding percentage, respectively to 41% in absence of G. vaginalis infection and 49.6% in presence of G. vaginalis infection compared to control group (saline + glucose), demonstrating a strong bioadhesion to vaginal mucosal tissue36 (Fig. 6).
Mucoadhesive effect of the product containing PP, GS, and LA. The treatment with the product containing PP, GS and LA showed a significant binding percentage, respectively to respectively to 41% in absence of G. vaginalis infection and 49.6% in presence of G. vaginalis infection compared to control group (saline + glucose). ***p < 0.001 vs control group.
Discussion
BV is caused by an overgrowth of some bacteria naturally found in the vaginal microbiota3,37. The vaginal mucosa represents an important and complex ecosystem dominated by Lactobacillus strains and containing a small number of fungi and balanced microbial communities which are vital for the health of the female reproductive tract38. However, even though 30% of women possess a healthy vaginal microbiota, a lack of significant concentrations of Lactobacillus species has been found compared to other facultative or strictly anaerobic bacteria39,40. The vaginal mucosal barrier is a multifaceted system comprising of a mucus layer and an epithelium layer. The latter guarantees mechanical protection against harmful pathogens41 as it embodies antimicrobial peptides (AMPs) associated with the innate and the adaptive immune system which represent the first line of defence against pathogen invasion42.The standard treatment for BV includes the use of antibiotics such as: metronidazole (MET), clindamycin (CLIN) or alternatively tinidazole (TI), which can be administered orally or intravaginally43. A systematic review conducted by Oduyebo et al.44 revealed that CLIN and MET, either in oral or vaginal formulations, were equally effective in treating BV, and that CLIN caused a lower rate of adverse events than MET. Studies suggest a lack of efficacy of traditional antimicrobial treatments and high recurrence rates of BV, of 30% and 76% within 4 weeks and 6 months, respectively, due to their inability to completely eradicate the vaginal biofilm. In addition, the negative effect of antibiotics on healthy vaginal microbiota impact therapeutical efficacy and symptom relief even further43,45. This ecosystem can be altered by several factors, including alterations of CO2/O2 levels and pH in the vaginal lumen. Considering an imbalance of vaginal mucosal barrier plays a pivotal role in BV pathogenesis10, novel treatments strengthening the vaginal barrier could represent an alternative solution to avoid and/or reduce the onset and progression of GV infections. Thus, there is a great need to source and develop new compounds to combat them46,47. Traditionally, natural compounds have been used for the treatment of vaginal dysbiosis43. These include plant extracts, such as Prangos ferulacea, Myrtus communis, Zataria multiflora, essential oils, small antimicrobial peptides of animal origin, bacteriocins and various groups of plant compounds (such as triterpenoids, phenols, and flavonoids) with antimicrobial and antiviral activity46,48. In particular, studies conducted by Machado and Rosca25,32 demonstrated the potential use of Thymbra capitata essential oil (EO) as a novel therapeutic agent in the treatment of BV thanks to its antimicrobial effect against several bacterial species associated with BV including Gardnerella species. The natural products can be used in combination with MET or probiotics to improve the efficacy of antimicrobial therapy and reduce the recurrence of vaginal infections43. Natural compounds are preferred over synthetic drugs to avoid any contraindications or side effects related to the intake of synthetic substances, which makes them attractive to patients46. In this context, recent reports have highlighted the health-promoting properties of some natural compounds in the management of BV25,34. A previous study performed by Esposito et al.34 demonstrated the beneficial effects of grape seed extract and pea protein in a murine model of vulvovaginal candidiasis, showing the ability of these substances to create a protective barrier on vaginal mucosa against fungal infection. Furthermore, LA has also gained considerable interest in the field of medicine due to its ability to maintain an acidic environment (pH 3.5–4.5) in the vagina to restrict the growth of fungi such as C. glabrata and C. albicans47,49. In particular, vaginal gels containing LA have an optimal adherence to the mucosa and enhance the vaginal defence mechanisms50. In addition, the optimal safety profile of PP, GS, and LA was demonstrated34,47,51 suggesting their possible use as a therapeutic alternative for the management of vaginal dysbiosis including BV. Based on these findings, the efficacy to manage BV-associated symptoms utilizing the aforementioned substances were investigated. Firstly, we evaluated GV proliferation through CFU counts, demonstrating that the product containing PP, GS and LA was able to significantly reduce vaginal bacterial growth in both pre-treatment and post-treatment conditions due to its ability to form a protective mucomimetic barrier on the vaginal mucosa. Furthermore, it has been demonstrated that BV causes alternations to the vaginal tissue35, characterized by the presence of edema, epithelial thickness and polymorphonuclear (PMN) cell infiltration35. In this context, our results demonstrated that the pre-treatment with the product containing PP, GS and LA was able to preserve vaginal tissue architecture, reducing epithelium thickness, compared to the GV infection group. Moreover, PP, GS and LA exerted a protective effect also in post-treatment condition. The development of histological damage after GV infection was accompanied by an increase of MPO activity. Here the treatment with PP, GS and LA demonstrated to be effective in reducing MPO activity, with a marked decrease in the pre-treatment group compared to the post-treatment group.
A relevant hallmark feature of BV is the presence of high levels of sialidase activity in vaginal fluid compared to specimens from women with normal microbiota35. Production of sialidase enzyme by isolated BV-associated bacteria grown in culture strongly suggests that BV-associated sialidases are bacterial in origin52. Bacterial sialidases have been characterized as virulence factors in bacterial infections of various mucosal sites52,53. Here, PP, GS and LA significantly inhibit sialidase activity, particularly at 1 h before GV infection. Additionally, the bioadhesive properties of the product containing PP, GS and LA with vaginal mucosa were evaluated. Mucosal membranes of human organism, including vaginal tissues, are characterized by an epithelial layer whose surface is covered by mucus36,38. The mucus contains glycoproteins, lipids, inorganic salts and 95% water by mass, which makes it a highly hydrated system and relatively permeable36. Our data demonstrated that the product containing PP, GS and LA possesses a strong bioadhesive potential to vaginal mucosal tissue, exerting a muco-protective barrier function.
Although many natural products have been identified for the treatment of BV, some of them, such as probiotics, have not been shown to be effective when administered alone29. It has been demonstrated that a single strain or mixture of Lactobacilli exerts many benefits in BV patients through lactic acid production; however, probiotics only contain bacterial strains without other potential beneficial factors, which poses a limitation for BV management29. Notably, our study demonstrates the beneficial effects of PP, GS, and LA, which provide a barrier-forming mechanism of action and can be considered an alternative therapeutic strategy for the treatment of BV to re-establish the pH and integrity of the vaginal mucosa. Preclinical models come with a set of limitations lacking translational reproduction of human diseases. Nonetheless, the association of PP, GS and LA could pave the way to better understanding new strategies of modulating bacterial infections, thus reducing the frequency and severity of symptoms in BV patients. It would be intriguing to further understand how PP, GS and LA could restore the balance of the vaginal microbiota by assessing resident microbial communities. Understanding the effects of PP, GS and LA in other bacteria-associated vaginal dysbiosis, such as Prevotella and Mobiluncus species or Chlamydia trachomatis and Neisseria gonorrhoea infections54 may also provide a deeper understanding on ways to maintain vaginal health, reduce the risk of relapse and improve the quality of life in BV patients. In conclusion, the obtained data demonstrated that the pre-treatment with a product containing PP, GS, and LA could decrease the engraftment of GV; while if used as a post treatment, it represents an impediment for the colonization of GV in the vaginal environment. Moreover, our results suggest that the product is able to preserve vaginal tissue architecture and avoid invasion and bacterial growth if used as both preventive or curative treatment for the management of BV. Thus, PP, GS and LA could represent an alternative therapeutic strategy to prevent and treat BV, significantly decreasing the risk of developing antimicrobial resistance that may come with conventional therapies.
Material and methods
Animals
Seven-week-old female C57BL/6 mice weighing 19–22 g were housed in wire cages under climate-controlled conditions (at 50 ± 10% humidity and 20–22 °C) in accordance with ARRIVE guidelines, fed with standard laboratory chow, and water ad libitum. The experimental protocol was approved by the Ethics Committee of University of Messina (n°409/2022-PR). The Animal experiments followed Italian regulations on protection of animals used for experimental and other scientific purposes (DM 116192) as well as EU regulations (OJ of EC L 358/1 12/18/1986). The animals used for this study were randomly selected from those suitable and available at that time.
Gardnerella vaginalis growth condition
Gardnerella vaginalis (GV) KCTC5096 was obtained from the Korean Collection for Type Cultures (KCTC, Daejeon, Korea). The GV strain was cultured in a modified brain–heart infusion (mBHI; Difco, Detroit, MI, USA) broth containing 10% (v/v) horse serum (Life Technologies Co., Grand Island, NY, USA), 1% (w/v) yeast extract (Difco, Detroit, MI, USA), 0.1% (w/v) maltose, and 0.1% (w/v) glucose, and cultivated at 37 °C for 48 h under anaerobic conditions (BD GasPakTM EZ pouch systems, BS, USA)55.
Gardnerella vaginalis-induced vaginosis model
Mice were treated subcutaneously with β-Estradiol-3-benzoate (0.5 mg/0.1 mL) 72 h before GV infection, and then a suspension of GV (1 × 106 CFU /20 µL saline) was administered intravaginally35,56. The control groups were treated with saline instead of the GV suspension. The product containing PP, GS and LA was administered intravaginally once a day for 7 consecutive days 1 h before GV-infection at the dose of 0.2 g/mouse. For another set of experiments, the product containing PP, GS and LA was administered once a day for 7 consecutive days after infection at the same dose. GV-infected control group was treated with saline (vehicle). Mice were sacrificed as previously described by Valentim et al.57 24 h after the final test item treatment. After placing the animal in the sevoflurane chamber to induce unconsciousness, cervical dislocation was performed57. GV was chosen to perform the BV model because it is considered one of the most common bacteria involved in BV17,18.
Experimental groups
Mice were divided into 5 experimental groups:
-
group 1: mice received vehicle (saline) without GV infection for 7 days (n = 5)
-
group 2: mice received PP, GS and LA without GV infection for 7 days (n = 5)
-
group 3: mice received PP, GS and LA 1 h before GV infection for 7 days (pre-treatment) (n = 10)
-
group 4: mice were infected with GV without PP, GS, and LA treatment (n = 10)
-
group 5: mice received PP, GS and LA beginning the day after infection GV for 7 days (post-treatment) (n = 10).
CFU evaluation
Serial dilutions of vaginal washes onto NYC-III agar plates + 1 mg/mL streptomycin and 4 mg/L amphotericin (both from Sigma-Aldrich) were performed to evaluate GV proliferation as previously described58. The plates were incubated at 37 °C under anaerobic conditions. This process was carried out with two replicates in at least three independent assays. The final concentration of bacteria-free supernatants per well is reported based on the 10% v/v dilution of the bacterial culture density (CFU/ml) and then expressed as %59.
Histological evaluation
Histological evaluation was performed as previously described by Lanza et al.60. The vaginal tissues were fixed with 10% neutral formalin, embedded in paraffin, and sectioned at 7 µm. Sections were stained with hematoxylin–eosin (H&E). All sections were evaluated using an AxioVision microscope (Axostar Plus equipped with Axio-Cam MRc, Zeiss, GE, Germany). The histological results are shown at 10× magnification (100 μm of the Bar scale). The parameters evaluated to assess tissue architecture were: epithelial thickness, edema and polymorphonuclear (PMN) cell infiltration35,58,61. Infection degree was scored as (1) absent (score of 0), meaning no yeast elements; (2) mild (score of 1), meaning 1–30 yeast elements in the vaginal lumen and cornified epithelial layer, or ≥ 5 in the upper 1/3 layer of the vaginal mucosa; (3) moderate (score of 2), meaning 31–60 yeast elements in the vaginal lumen and cornified epithelial layer or ≥ 5 in the upper 2/3 layer of the vaginal mucosa; and (4) severe (score of 3), meaning > 60 yeast elements in the vaginal lumen and cornified epithelial layer or ≥ 5 in the whole layer of the vaginal mucosa62. The slides were analyzed by a pathologist blinded to the treatment groups.
Myeloperoxidase (MPO) activity
MPO activity, an index of polymorphonuclear cell accumulation, was determined as previously described by Lanza et al.60. The rate of change in absorbance was measured spectrophotometrically at 650 nm. MPO activity was measured as the quantity of enzyme degrading 1 mM of peroxide min − 1 at 37 °C and was expressed in units per gram weight of wet tissue.
Sialidase activity assay
The activity of sialidase, an enzyme commonly used as diagnostic marker of BV63, was evaluated as previously described64. In the presence of BV, bacterial sialidase plays an important role in biofilm formation on vaginal epithelium63. To quantitatively assay the vaginal wash samples for sialidase activity, 100 µL of the specimen were incubated on a microplate at room temperature and placed on a shaker with 100 µL of the substrate 2-(3′-methoxyphenyl)-N-acetyl-d-neuraminic acid (Sigma, St. Louis) dissolved in 0.05 mol/L sodium acetate, pH 5.0.16. After 1 h of incubation 50 µL of 4 mmol/L 4-aminoantipyrine and 50 µL of 6 mmol/L potassium ferricyanide were added. A duplicate sample lacking the substrate served as sample blank. Absorbances were read at 492 nm.
Mucoadhesion test
Mucoadhesion test was performed as previously described by Gremião et al.36. The everted vaginal sac assay evaluated the bioadhesive interaction of a product containing PP, GS, and LA with everted vaginal tissue in infected and non-infected mice.
Materials
The product containing PP, GS and LA was kindly provided by DEVINTEC SAGL (Lugano, Switzerland). All chemicals were obtained from the highest grade of commercial sources. All stock solutions were prepared in non-pyrogenic saline (0.9% NaCl; Baxter, Liverpool, UK).
Statistical evaluation
All data were expressed as the mean ± SEM. The results were analysed with GraphPad 7 software, using one-way analysis of variance (ANOVA), followed by a Bonferroni post hoc test for multiple comparisons. A p-value of less than 0.05 was considered significant.
Data availability
The authors declare that all data and materials supporting the findings of this study are available within the article.
References
Ranjit, E., Raghubanshi, B. R., Maskey, S. & Parajuli, P. Prevalence of bacterial vaginosis and its association with risk factors among nonpregnant women: A hospital based study. Int. J. Microbiol. 2018, 8349601. https://doi.org/10.1155/2018/8349601 (2018).
Javed, A., Parvaiz, F. & Manzoor, S. Bacterial vaginosis: An insight into the prevalence, alternative treatments regimen and it’s associated resistance patterns. Microb. Pathog. 127, 21–30. https://doi.org/10.1016/j.micpath.2018.11.046 (2019).
Kairys, N. & Garg, M. Bacterial vaginosis. In StatPearls (Treasure Island, 2022).
Engberts, M. K., Boon, M. E., van Haaften, M. & Heintz, A. P. Symptomatic candidiasis: Using self sampled vaginal smears to establish the presence of Candida, Lactobacilli, and Gardnerella vaginalis. Diagn. Cytopathol. 35, 635–639. https://doi.org/10.1002/dc.20708 (2007).
Olmsted, S. S., Meyn, L. A., Rohan, L. C. & Hillier, S. L. Glycosidase and proteinase activity of anaerobic gram-negative bacteria isolated from women with bacterial vaginosis. Sex Transm. Dis. 30, 257–261. https://doi.org/10.1097/00007435-200303000-00016 (2003).
Morrill, S., Gilbert, N. M. & Lewis, A. L. Gardnerella vaginalis as a cause of bacterial vaginosis: Appraisal of the evidence from in vivo models. Front. Cell Infect. Microbiol. 10, 168. https://doi.org/10.3389/fcimb.2020.00168 (2020).
Sharami, S. H., Afrakhteh, M. & Shakiba, M. Urinary tract infections in pregnant women with bacterial vaginosis. J. Obstet. Gynaecol. 27, 252–254. https://doi.org/10.1080/01443610701194846 (2007).
Kovachev, S. M. Cervical cancer and vaginal microbiota changes. Arch. Microbiol. 202, 323–327. https://doi.org/10.1007/s00203-019-01747-4 (2020).
Harmanli, O. H., Cheng, G. Y., Nyirjesy, P., Chatwani, A. & Gaughan, J. P. Urinary tract infections in women with bacterial vaginosis. Obstet. Gynecol. 95, 710–712. https://doi.org/10.1016/s0029-7844(99)00632-8 (2000).
Amabebe, E. & Anumba, D. O. C. The vaginal microenvironment: The physiologic role of Lactobacilli. Front. Med. (Lausanne) 5, 181. https://doi.org/10.3389/fmed.2018.00181 (2018).
Valenti, P. et al. Role of lactobacilli and lactoferrin in the mucosal cervicovaginal defense. Front. Immunol. 9, 376. https://doi.org/10.3389/fimmu.2018.00376 (2018).
van de Wijgert, J. The vaginal microbiome and sexually transmitted infections are interlinked: Consequences for treatment and prevention. PLoS Med. 14, e1002478. https://doi.org/10.1371/journal.pmed.1002478 (2017).
Randis, T. M. & Ratner, A. J. Gardnerella and prevotella: Co-conspirators in the pathogenesis of bacterial vaginosis. J. Infect. Dis. 220, 1085–1088. https://doi.org/10.1093/infdis/jiy705 (2019).
McKenzie, R., Maarsingh, J. D., Laniewski, P. & Herbst-Kralovetz, M. M. Immunometabolic analysis of Mobiluncus mulieris and Eggerthella sp. Reveals novel insights into their pathogenic contributions to the hallmarks of bacterial vaginosis. Front. Cell Infect. Microbiol. 11, 759697. https://doi.org/10.3389/fcimb.2021.759697 (2021).
Spiegel, C. A. et al. Gardnerella vaginalis and anaerobic bacteria in the etiology of bacterial (nonspecific) vaginosis. Scand. J. Infect. Dis. Suppl. 40, 41–46 (1983).
Hardy, L. et al. The presence of the putative Gardnerella vaginalis sialidase A gene in vaginal specimens is associated with bacterial vaginosis biofilm. PLoS ONE 12, e0172522. https://doi.org/10.1371/journal.pone.0172522 (2017).
van de Wijgert, J. H. et al. The vaginal microbiota: What have we learned after a decade of molecular characterization?. PLoS ONE 9, e105998. https://doi.org/10.1371/journal.pone.0105998 (2014).
Fredricks, D. N., Fiedler, T. L., Thomas, K. K., Oakley, B. B. & Marrazzo, J. M. Targeted PCR for detection of vaginal bacteria associated with bacterial vaginosis. J. Clin. Microbiol. 45, 3270–3276. https://doi.org/10.1128/JCM.01272-07 (2007).
Castro, J., Jefferson, K. K. & Cerca, N. Genetic heterogeneity and taxonomic diversity among gardnerella species. Trends Microbiol. 28, 202–211. https://doi.org/10.1016/j.tim.2019.10.002 (2020).
Vaneechoutte, M. et al. Emended description of Gardnerella vaginalis and description of Gardnerella leopoldii sp. nov., Gardnerella piotii sp. nov. and Gardnerella swidsinskii sp. nov., with delineation of 13 genomic species within the genus Gardnerella. Int. J. Syst. Evol. Microbiol. 69, 679–687. https://doi.org/10.1099/ijsem.0.003200 (2019).
Plummer, E. L. et al. Gardnerella vaginalis clade distribution is associated with behavioral practices and nugent score in women who have sex with women. J. Infect. Dis. 221, 454–463. https://doi.org/10.1093/infdis/jiz474 (2020).
Qin, H. & Xiao, B. Research progress on the correlation between gardnerella typing and bacterial vaginosis. Front. Cell Infect. Microbiol. 12, 858155. https://doi.org/10.3389/fcimb.2022.858155 (2022).
Hill, J. E., Albert, A. Y. K. & Group, V. R. Resolution and cooccurrence patterns of Gardnerella leopoldii, G. swidsinskii, G. piotii, and G. vaginalis within the vaginal microbiome. Infect. Immun. 2019, 87. https://doi.org/10.1128/IAI.00532-19 (2019).
Machado, D., Castro, J., Palmeira-de-Oliveira, A., Martinez-de-Oliveira, J. & Cerca, N. Bacterial vaginosis biofilms: Challenges to current therapies and emerging solutions. Front. Microbiol. 6, 1528. https://doi.org/10.3389/fmicb.2015.01528 (2015).
Rosca, A. S. et al. Six bacterial vaginosis-associated species can form an in vitro and ex vivo polymicrobial biofilm that is susceptible to Thymbra capitata essential oil. Front. Cell Infect. Microbiol. 12, 824860. https://doi.org/10.3389/fcimb.2022.824860 (2022).
Bradshaw, C. S. & Sobel, J. D. Current treatment of bacterial vaginosis-limitations and need for innovation. J. Infect. Dis. 214(Suppl 1), S14-20. https://doi.org/10.1093/infdis/jiw159 (2016).
Zimmermann, P. & Curtis, N. The effect of antibiotics on the composition of the intestinal microbiota—a systematic review. J. Infect. 79, 471–489. https://doi.org/10.1016/j.jinf.2019.10.008 (2019).
Faught, B. M. & Reyes, S. Characterization and treatment of recurrent bacterial vaginosis. J. Womens Health (Larchmt) 28, 1218–1226. https://doi.org/10.1089/jwh.2018.7383 (2019).
Wu, S., Hugerth, L. W., Schuppe-Koistinen, I. & Du, J. The right bug in the right place: Opportunities for bacterial vaginosis treatment. NPJ Biofilms Microbiomes 8, 34. https://doi.org/10.1038/s41522-022-00295-y (2022).
Dover, S. E., Aroutcheva, A. A., Faro, S. & Chikindas, M. L. Natural antimicrobials and their role in vaginal health: A short review. Int. J. Probiot. Prebiot. 3, 219–230 (2008).
Sousa, L. G. V. et al. Synergistic effects of carvacrol, alpha-terpinene, gamma-terpinene, rho-cymene and linalool against Gardnerella species. Sci. Rep. 12, 4417. https://doi.org/10.1038/s41598-022-08217-w (2022).
Machado, D. et al. Thymbra capitata essential oil as potential therapeutic agent against Gardnerella vaginalis biofilm-related infections. Future Microbiol. 12, 407–416. https://doi.org/10.2217/fmb-2016-0184 (2017).
Mathai, J. K., Liu, Y. & Stein, H. H. Values for digestible indispensable amino acid scores (DIAAS) for some dairy and plant proteins may better describe protein quality than values calculated using the concept for protein digestibility-corrected amino acid scores (PDCAAS). Br. J. Nutr. 117, 490–499. https://doi.org/10.1017/S0007114517000125 (2017).
Esposito, E. et al. Effect of pea protein plus grape seed dry extract on a murine model of Candida albicans induced vaginitis. Future Microbiol. 13, 1375–1382. https://doi.org/10.2217/fmb-2018-0102 (2018).
Gilbert, N. M., Lewis, W. G. & Lewis, A. L. Clinical features of bacterial vaginosis in a murine model of vaginal infection with Gardnerella vaginalis. PLoS ONE 8, e59539. https://doi.org/10.1371/journal.pone.0059539 (2013).
Carvalho, F. C., Bruschi, M. L., Evangelista, R. C. & Gremiao, M. P. D. Mucoadhesive drug delivery systems. Braz. J. Pharm. Sci. 46, 1–17. https://doi.org/10.1590/S1984-82502010000100002 (2010).
Bagnall, P. & Rizzolo, D. Bacterial vaginosis: A practical review. JAAPA 30, 15–21. https://doi.org/10.1097/01.JAA.0000526770.60197.fa (2017).
Medina-Colorado, A. A. et al. Vaginal ecosystem modeling of growth patterns of anaerobic bacteria in microaerophilic conditions. Anaerobe 45, 10–18. https://doi.org/10.1016/j.anaerobe.2017.04.014 (2017).
Ma, B., Forney, L. J. & Ravel, J. Vaginal microbiome: Rethinking health and disease. Annu. Rev. Microbiol. 66, 371–389. https://doi.org/10.1146/annurev-micro-092611-150157 (2012).
Doyle, R. et al. A lactobacillus-deficient vaginal microbiota dominates postpartum women in rural Malawi. Appl. Environ. Microbiol. 2018, 84. https://doi.org/10.1128/AEM.02150-17 (2018).
Plesniarski, A., Siddik, A. B. & Su, R. C. The microbiome as a key regulator of female genital tract barrier function. Front. Cell Infect. Microbiol. 11, 790627. https://doi.org/10.3389/fcimb.2021.790627 (2021).
Yarbrough, V. L., Winkle, S. & Herbst-Kralovetz, M. M. Antimicrobial peptides in the female reproductive tract: A critical component of the mucosal immune barrier with physiological and clinical implications. Hum. Reprod. Update 21, 353–377. https://doi.org/10.1093/humupd/dmu065 (2015).
Tomas, M., Palmeira-de-Oliveira, A., Simoes, S., Martinez-de-Oliveira, J. & Palmeira-de-Oliveira, R. Bacterial vaginosis: Standard treatments and alternative strategies. Int. J. Pharm. 587, 119659. https://doi.org/10.1016/j.ijpharm.2020.119659 (2020).
Oduyebo, O. O., Anorlu, R. I. & Ogunsola, F. T. The effects of antimicrobial therapy on bacterial vaginosis in non-pregnant women. Cochrane Database Syst. Rev. https://doi.org/10.1002/14651858.CD006055 (2009).
Reiter, S. & Kellogg Spadt, S. Bacterial vaginosis: A primer for clinicians. Postgrad. Med. 131, 8–18. https://doi.org/10.1080/00325481.2019.1546534 (2019).
Stan, D. et al. Natural compounds with antimicrobial and antiviral effect and nanocarriers used for their transportation. Front. Pharmacol. 12, 723233. https://doi.org/10.3389/fphar.2021.723233 (2021).
Bulgasem, B. Y., Lani, M. N., Hassan, Z., Wan Yusoff, W. M. & Fnaish, S. G. Antifungal activity of lactic acid bacteria strains isolated from natural honey against pathogenic Candida species. Mycobiology 44, 302–309. https://doi.org/10.5941/MYCO.2016.44.4.302 (2016).
Boukhatem, M. N. & Setzer, W. N. Aromatic herbs, medicinal plant-derived essential oils, and phytochemical extracts as potential therapies for coronaviruses: Future perspectives. Plants (Basel) 2020, 9. https://doi.org/10.3390/plants9060800 (2020).
Rosenfeld, W. D. & Clark, J. Vulvovaginitis and cervicitis. Pediatr. Clin. N. Am. 36, 489–511. https://doi.org/10.1016/s0031-3955(16)36682-2 (1989).
Barnhart, K. T. et al. In vivo distribution of a vaginal gel: MRI evaluation of the effects of gel volume, time and simulated intercourse. Contraception 70, 498–505. https://doi.org/10.1016/j.contraception.2004.06.013 (2004).
Paterniti, I. et al. A new approach for the treatment of recurrent vulvovaginal candidiasis with a combination of pea protein, grape seed extract, and lactic acid assessed in vivo. J. Fungi (Basel) 2022, 8. https://doi.org/10.3390/jof8121251 (2022).
Briselden, A. M., Moncla, B. J., Stevens, C. E. & Hillier, S. L. Sialidases (neuraminidases) in bacterial vaginosis and bacterial vaginosis-associated microflora. J. Clin. Microbiol. 30, 663–666. https://doi.org/10.1128/jcm.30.3.663-666.1992 (1992).
Lewis, A. L. & Lewis, W. G. Host sialoglycans and bacterial sialidases: A mucosal perspective. Cell Microbiol. 14, 1174–1182. https://doi.org/10.1111/j.1462-5822.2012.01807.x (2012).
Bautista, C. T. et al. Association of bacterial vaginosis with chlamydia and gonorrhea among women in the US army. Am. J. Prev. Med. 52, 632–639. https://doi.org/10.1016/j.amepre.2016.09.016 (2017).
Kim, H., Kim, Y. & Kang, C. H. In vivo confirmation of the antimicrobial effect of probiotic candidates against Gardnerella vaginalis. Microorganisms 2021, 9. https://doi.org/10.3390/microorganisms9081690 (2021).
Jang, S. E. et al. Lactobacillus rhamnosus HN001 and Lactobacillus acidophilus La-14 attenuate Gardnerella vaginalis-infected bacterial vaginosis in mice. Nutrients 2017, 9. https://doi.org/10.3390/nu9060531 (2017).
Valentim, A. M., Guedes, S. R., Pereira, A. M. & Antunes, L. M. Euthanasia using gaseous agents in laboratory rodents. Lab Anim 50, 241–253. https://doi.org/10.1177/0023677215618618 (2016).
Choi, S. I., Won, G., Kim, Y., Kang, C. H. & Kim, G. H. Lactobacilli strain mixture alleviates bacterial vaginosis through antibacterial and antagonistic activity in gardnerella vaginalis-infected C57BL/6 mice. Microorganisms 2022, 10. https://doi.org/10.3390/microorganisms10020471 (2022).
Anton, L. et al. Gardnerella vaginalis alters cervicovaginal epithelial cell function through microbe-specific immune responses. Microbiome 10, 119. https://doi.org/10.1186/s40168-022-01317-9 (2022).
Lanza, M. et al. Properties of a new food supplement containing actinia equina extract. Antioxidants (Basel) 2020, 9. https://doi.org/10.3390/antiox9100945 (2020).
O’Brien, V. P. et al. Low-dose inoculation of Escherichia coli achieves robust vaginal colonization and results in ascending infection accompanied by severe uterine inflammation in mice. PLoS ONE 14, 19941. https://doi.org/10.1371/journal.pone.0219941 (2019).
Zhang, J. E. et al. Feasibility of histological scoring and colony count for evaluating infective severity in mouse vaginal candidiasis. Exp. Anim. 62, 205–210. https://doi.org/10.1538/expanim.62.205 (2013).
Wu, S. et al. A biochemiluminescent sialidase assay for diagnosis of bacterial vaginosis. Sci. Rep. 9, 20024. https://doi.org/10.1038/s41598-019-56371-5 (2019).
Robinson, L. S., Schwebke, J., Lewis, W. G. & Lewis, A. L. Identification and characterization of NanH2 and NanH3, enzymes responsible for sialidase activity in the vaginal bacterium Gardnerella vaginalis. J. Biol. Chem. 294, 5230–5245. https://doi.org/10.1074/jbc.RA118.006221 (2019).
Funding
The study was supported financially by DEVINTEC, SAGL, Switzerland.
Author information
Authors and Affiliations
Contributions
M.L., A.F., G.C. and S.A.S. conducted the experiment. S.C., I.P., and E.E. conceived the experiment. A.P.C., and M.C. analysed the results. M.L. and S.A.S. drafted the manuscript. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lanza, M., Scuderi, S.A., Capra, A.P. et al. Effect of a combination of pea protein, grape seed extract and lactic acid in an in vivo model of bacterial vaginosis. Sci Rep 13, 2849 (2023). https://doi.org/10.1038/s41598-023-28957-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-28957-7
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.