Abstract
Dendrobium officinale Kimura et Migo is a famous precious medicinal plant in China. Seed and seedling were cultivated with the mycorrhizal fungus Sebacina sp. CCaMK was initially cloned from D. officinale based on a SSH cDNA library of symbiotically germinated seeds with Sebacina sp. Phylogenetic analysis was performed among DoCCaMK and other CCaMKs. The particle bombardment technique was used to visualize DoCCaMK-GFP. qRT-PCR and western blot analysis were conducted to determine the tissue expression patterns of DoCCaMK with (SGS) and without (UGS) Sebacina sp. Furthermore, the effect of KN-93 on CCaMK expression was also examined. Using NMT the net Ca2+ fluxes and the CCaMK concentration were measured during D. officinale seed germination. DoCCaMK had the highest homology with Lilium longiflorum CCaMK. The DoCCaMK-GFP protein localized in the nucleus and cell membrane. CCaMK expression was significantly upregulated after symbiosis with Sebacina sp. KN-93 could be used as an inhibitor of CCaMK to inhibit D. officinale seed germination. Ca2+ influx and the concentration of the CCaMK in the SGS group was significantly more than that of the UGS group. The characterization of CCaMK provides certain genetic evidence for the involvement of this gene during seed germination and mycorrhizal cultivation in D. officinale.
Similar content being viewed by others
Introduction
Most plants form plant-microbe interactions known as symbioses1. For arbuscular mycorrhizal (AM) symbioses, fungi help plants acquire nutrients, especially via phosphorus absorption, and thus play a key role in the improvement of plant growth2. For nodulation, plants gain nitrogen from bacterial symbionts in the form of ammonia, which can be easily utilized3. On the other hand, the fungal and bacterial partners gain carbon nutrients from plants2. Therefore, plants benefit from both mycorrhization and nodulation.
In the most common type of mycorrhization of the endo-mycorrhiza, symbiotic fungi penetrate the plant cell2. Orchid mycorrhization (OM) and AM are both forms of endo-mycorrhization, thus, to some extent, they share certain common structural characteristics. For OM, symbiotic fungal mycelia penetrate and colonize the root cortex and subsequently form pelotons, which have been regarded as a typical structure in orchids. Furthermore, previous findings have shown that the mycorrhizal-symbiosis mycoheterotrophic orchids shares at least some common properties with AM-forming plants4.
CCaMK, with the full name calcium and calmodulin (CaM)-dependent protein kinase, belongs to the calcium/CaM-dependent protein kinase superfamily5. It is a serine/threonine (Ser/Thr) protein kinase that is composed of a kinase domain, a calmodulin (CaM)-binding domain and an EF-hand motif-containing neural visinin-like Ca2+-binding domain. The activity of the protein is subject to dual regulation by Ca2+ and Ca2+/CaM6. CCaMK is unique among Ca2+-regulated proteins and is able to bind both free Ca2+ and Ca2+ in complex with CaM. It binds to Ca2+ either directly through the EF-hand domain or indirectly via the CaM-binding domain7. In recent years, Non-invasive micro-test technique (NMT) is a relatively novel technique to be widely used to record transmembrane ion influx and efflux such as Ca2+ directly in a non-contact way by detecting the diffusion potentials outside of the cell membrane in plant physiological research8. CCaMKs are plant specific and are distinctly different from calcium-dependent protein kinases (CDPKs) and other serine/threonine kinases in plants, but they are very highly similar to mammalian CaM-dependent protein kinase II (CaMKII)5. As an antagonizing protein kinase compound, KN-93 possesses great specificity for the CaMKII class of eukaryotic kinases9 and can also moderately inhibit zoospore release, encystment and cyst germination10.
Dendrobium is one of the largest genera of Orchidaceae. Under natural conditions, seed germination and seedling development of Dendrobium require compatible endophytic mycorrhizal fungi to supply many kinds of nutrients in natural conditions11. Similar to other precious herbs12, Chinese medicinal plant, D. officinale has ornamental value and a broad range of therapeutic effects, such as immunomodulation and hepatoprotective activities13,14. It is also commonly used as a traditional valuable tonic hygienic food in China15. However, due to phytopathogens, pests, commercial overexploitation and anthropogenic interference in natural habitats, this medicinal orchid herb is increasingly endangered16. Seed germination is a determining factor in the propagation of pant species17. Currently, symbiotic germination and asymbiotic germination are two effective methods for orchid propagation. The former refers to seeds inoculated with mycorrhizal fungi in a relatively barren medium, while the latter refers to seeds sown on sugar-rich medium without fungi. However, symbiotic germination was superior to asymbiotic germination, according to previously reported experiments18. Thus, it is imperative to carry out seed and seedling germination and cultivation using techniques that engender symbioses between the host plant and mycorrhizal fungi. Seed germination and seedling propagation in D. officinale depend on mycorrhizal fungi, such as Sebacinaceae, Tulasnellaceae and Ceratobasidiaceae, for acquisition of carbohydrates and other nutrients. In recent years, due to symbiosis between mycorrhizal fungi and D. officinale seeds or seedlings, increasing attention has been paid to morphological features, biochemical reactions, secondary metabolites and so on19, yet the molecular mechanism of D. officinale seed germination and seedling growth promotion induced by mycorrhizal fungi is still unclear.
In a previous study, a suppression subtractive hybridization (SSH) cDNA library of symbiotically germinated and ungerminated D. officinale seeds was constructed, and as one of the differentially expressed genes, CCaMK was selected as one OM symbiosis-associated candidate gene.
Particle bombardment is a relatively easy and powerful method for the transient expression of genes in plant cells, and transient gene expression has been extensively used in the subcellular localization of fluorescent proteins20. Therefore, in this study, the characteristics, subcellular and histological localization, and expression levels of DoCCaMK during Sebacina sp. interaction with D. officinale seed germination and seedling growth were investigated. Furthermore, a DoCCaMK-GFP fusion protein was also detected using western blot analysis. Whether KN-93 has an antagonistic effect on DoCCaMK during D. officinale seed germination and seedling propagation with Sebacina sp. was also investigated. In addition, real-time measurement of Ca2+ fluxes during D. officinale seed germination was detected using NMT technique and CCaMK activity was also investigated. This study will reveal the characteristics of CCaMK and serve as the basis for uncovering the mechanism of CCaMK in OM symbiosis.
Results
Homology and phylogenetic analysis of DoCCaMK
According to the SSH cDNA library of symbiotically germinated D. officinale seed EST sequences, DoCCaMK was found to have high homology (67%) with the CCaMK of Maianthemum racemosum (MrCCaMK). The full-length cDNA of DoCCaMK (Do807) was 2071 bp long. As deduced by Compute pI/MW, DoCCaMK has 514 amino acids, its isoelectric point is 5.92, and its molecular weight is 57.51 kDa. The comparison results from BLASTX showed that DoCCaMK had the highest homology (81%) with Lilium longiflorum LlCCaMK (Q43531). It also had a high homology (78%) with Brachypodium distachyon BdCCaMK (XP_003566106) and Triticum aestivum TaCCaMK (ADK22086). The identification of conserved domains in the CCaMK protein predicted by InterProScan analyses showed that DoCCaMK possessed serine/threonine/dual-specificity protein kinase catalytic domains, a tyrosine-protein kinase catalytic domain and an EF-hand locus (Table 1).
Phylogenetic analysis
The phylogenetic analysis was performed using the Neighbour-joining method. The result showed that CCaMKs from monocotyledons and dicotyledons gathered into two separate groups. DoCCaMK and LlCCaMK both belonged to the monocotyledon group and gathered into one clade, which indicated a close genetic relationship between them (Fig. 1a).
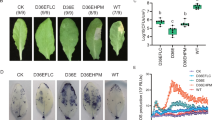
Phylogenetic tree and expression patterns of CCaMK in D. officinale by qRT-PCR analysis. In (a) Gm: Glycine max, Os: Oryza sativa, Bh: Brachypodium distachyon, LI: Lilium longiforum, Sb: Sorghum bicolor, Ta: T. aestivum, Zm: Zea mays, Lj: Lotus japonicas, Mt: Medicago truncatula, Pe: Pellia epiphylla. In (b) seed, root, stem and leaf represent different tissues of D. officinale without the mycorrhizal fungus, while I-seed and I-root indicate seed and root tissues infected with Sebacina sp. N-seed and N-root represent seed and root tissues infected with Sebacina sp. after KN-93 treatment. *P < 0.05 (compared to the UGS group).
DoCCaMK expression profile analysis
qRT-PCR analysis of the DoCCaMK gene at the transcriptional level revealed that in the D. officinale tissues without fungus group, CCaMK was constitutively expressed and that in the UGS group, it was downregulated in the root, stem and leaf tissues compared to that in the seed (Fig. 1b). CCaMK expression was significantly and inducibly upregulated 3.67- and 5.66-fold, respectively, in the seed and root by Sebacina sp. induction compared to that in the respective tissues in the UGS group. In the SGS group, after KN-93 was added, CCaMK expression levels in the root and seed were both significantly decreased. Furthermore, the expression of CCaMK in the KN-93 group was even significantly lower than that in the UGS group (Fig. 1b). The results indicated that CCaMK gene expression was induced by the mycorrhizal fungus, which plays important roles in the orchid mycorrhizal system.
Subcellular localization of the DoCCaMK-GFP fusion protein
Using the particle bombardment technique, a transient expression vector was transformed into onion epidermal cells. Then, the localization and distribution of DoCCaMK was observed. The DoCCaMK-GFP fusion protein was observed to be located in the onion epidermal cell nucleus and cell membrane (Fig. 2I) under a fluorescence microscope, while GFP was distributed throughout the onion cell in the control group (Fig. 2i). DoCCaMK-GFP and GFP were illustrated under bright field (Fig. 2II, Fig. 2ii). The DoCCaMK-GFP fusion protein and the GFP protein under white light and merge conditions were shown in Fig. 2III, Fig. 2iii. The results showed that DoCCaMK was localized in the cell nucleus and cell membrane.
DoCCaMK expression and western blot analysis
SDS-PAGE electrophoresis showed that the molecular weight of the recombinant DoCCaMK protein in vitro was 57 kDa, which was in accord with what Compute pI/MW predicted (Fig. 3a). Western blot analysis showed that the expression of CCaMK was different in different tissues of D. officinale and that the polyclonal antibody could specifically bind to CCaMK in symbiotically germinated seeds and roots induced by Sebacina sp., while it only slightly bound to CCaMK in the seed, stem and root tissues of the UGS group. The molecular weight of CCaMK in the symbiotically germinated seeds and roots was also approximately 57 kDa, which was in accordance with the recombinant DoCCaMK protein in vitro. Furthermore, the expression of CCaMK in the seed induced by Sebacina sp. was slightly less than that in the root induced by the same fungus (Fig. 3b).
Expression of DoCCaMK in different tissues of D. officinale by western blot analysis. In (a), M: Low molecular protein marker; in (a,b), R1: recombination protein. In (b), IR: D. officinale root symbiosis with Sebacina sp.; IS: D. officinale seeds in the SGS group; R: root; S1: seed; S2: stem; L: leaf; NS: KN-93 treatment seeds; NR: KN-93 treatment roots.
Effect of KN-93 on D. officinale seed germination
The dust-like seeds of D. officinale depend on mycorrhizal fungi for germination in the similar way as Gastrodia elata. At the beginning of germination, Orchids always form protocroms21. The developmental stages of seed germination were previously divided into five stages, according to Stewart et al.22. For example, at the germination stage, the embryo swells, enlarges and emerges from the coat, which is then named stage 2. When the embryo differentiates into a protomeristem, it is called the protocorm stage or stage 3, while at the time of stage 4, the first leaf emerges and the developmental process enters the seedling stage. In the present study, after cultivation for 35 d, D. officinale seeds in the SGS group germinated to stage 5, in which shoot tips began extending increasingly longer, and large amounts of rhizoids formed (Fig. 4a). However, in the control group, most of the seeds germinated to stage 2, in which most of the embryos became swollen, turned green and broke through the seed coats, but some only became slightly swollen, with their seed coats remaining intact and staying in stage 1 (Fig. 4b).
Effect of KN-93 on D. officinale seed germination after cultivation for 35 d. (a) CK, green protocorms with long shoot tips and rhizoids at 35 d after inoculation (stage 5). Scale bar: 1000 µm. (b) In the experimental group, most seeds swelled and geminated to stage 2, but some became only slightly swollen and remained at stage 1. Scale bar: 500 µm.
Real-time measurement of Ca2+ fluxes during D. officinale seed germination
As shown in Fig. 5, after seed germination for 35d, in the SGS, UGS and KN-93 complement groups, the result showed that the uptake of Ca2+ in the SGS group was evidently increased compared to that of the UGS and KN-93 complement groups. The mean Ca2+ influx was lower 229% and 320% in the seed of the UGS group and KN-93 complement group than that of the SGS group.
Real-time measurement of Ca2+ fluxes during D. officinale seed germination Ca2+ fluxes (pmol·cm−2·s−1) showed that the negative values standed for influx of Ca2+ and the positive values represented for efflux of Ca2+. The values of net Ca2+ flux were means ± SD (n = 6). After inoculation for 35d, Ca2+ influx in the seed continued for a period of time in different groups during D. officinale seed germination and the net Ca2+ uptake in the SGS group increased significantly than that of the UGS and KN-93-treatment group.
CCaMK concentration detection during D. officinale seed germination
The standard curve and regression equation were established (y = 61.975×−5.7033, R2 = 0.9989) in Fig. 6. The concentration of CCaMK in the SGS group was 14.17 U/mL, significantly higher than that of the control (UGS) group (P < 0.05) with 2.45 folds change. The CCaMK activity of the UGS group was 1.61 times that of the KN-93 group (Table 2).
Discussion
As an important signalling molecule in eukaryote cells, CCaMK plays a critical role in many physiological processes. In plants, it participates in development and hormone-related reactions, mainly involving light response regulation, stress and symbiosis. For example, during T. aestivum seed germination and seedling development, CCaMK overexpression decreases the sensitivity of the host plant to abscisic acid (ABA)13. In recent years, CCaMK has been proven to exert vital regulatory effects in the signal transduction of the symbiosis interaction between rhizobia or arbuscular mycorrhizal fungi and many plants23, while Arabidopsis has been reported to be the only non-mycorrhizal plant species, lacking the CCaMK gene24. The phylogenetic analysis in the present study showed that DoCCaMK had the highest homology with LlCCaMK, followed by that with BdCCaMK and TaCCaMK, with a homology of 78%. Therefore, plant CCaMKs are structurally conserved at both the protein and gene levels5.
It was found that the induction of the host genes essential for infection of AM fungi required nuclear localization of CCaMK23. A previous study also reported that ZmCCaMK is located in the nucleus25. Transient transformation of the epidermal cell layer of the onion bulb with the particle bombardment method and subcellular localization analysis of CCaMK fused with green fluorescent protein in the present study showed that DoCCaMK-GFP of D. officinale was located in the nucleus and the membrane, which was slightly different from what had been predicted before, with localization only in the nucleus25. As Ca2+ in the nucleus requires interactions between K+ channels, Ca2+ transporters and Ca2+ channels at the membrane of the nucleus26, CCaMK localizing in the cell membrane and nucleus of D. officinale may facilitate coordinated activities among ions.
It is well known that specific genes are involved in a so-called common symbiotic pathway (CSP)3. As CCaMK is considered to be a master decoder, a regulatory kinase and a key component of the CSP, it plays an essential role in deciphering the nuclear Ca2+ oscillations to control the proper transcriptional response2. Calcium oscillation requires CCaMK, and the activation of CCaMK is sufficient not only for AM and rhizobium-legume symbioses but also for actinomycetes to induce symbiotic processes27. The occurrence of calcium spiking, which caused the intracellular calcium ion concentration oscillates will response to nodulation or mycorrhization factors produced by the microbial symbionts28. In the present study, real-time Ca2+ fluxes measurement during D. officinale seed germination using NMT showed that Ca2+ influx in the SGS group was significantly more than that of the UGS group (Fig. 5). The Ca2+ uptake in the SGS group caused intracellular Ca2+ concentration to be increased and this may facilitate Ca2+ spiking formation and interaction with CCaMK. The more Ca2+ uptake from the environment, the more CCaMK will probably interact with this ion. In addition, the concentration of the CCaMK in the SGS group was much higher than that of the UGS group (Table 2), which indicated that the mycorrizal fungus Sebacina sp. could increase the CCaMK activity of the host plant.
CCaMK is highly expressed in tissues except for in roots, indicating its other roles in addition to the regulation of symbiosis29. Partly consistent with this observation, in the present study, the expression of CCaMK presented tissue specificity, and it was expressed relatively highly in the seed compared to that in the root, stem and leaf in asymbiotic group (Fig. 1b). CCaMK expression was low in the seed, root, and stem of D. officinale without interaction with a mycorrhizal fungus; however, it was upregulated 3.67 and 5.66 times, respectively, in the seed and root induced by Sebacina sp. compared to that in those tissues in the UGS group. In the SGS group, the expression level of CCaMK, especially in the root of D. officinale, was induced much higher than in other tissues, which indicated that Sebacina sp. directly and intensely contacted D. officinale roots and interacted with them in the co-culture system between D. officinale seedlings and the fungus; the result will be confirmed in the undergoing work. Additionally, at the protein level, DoCCaMK was only weakly expressed in the stem, seed and root, but it was highly expressed in the seed and root of the SGS group (Fig. 3b). Furthermore, DoCCaMK was expressed a bit more highly in the root relative to that in the seed induced by Sebacina sp., indicating that DoCCaMK probably affected symbiotic interaction with the root of the host plant more remarkably than that in the seed germination. The results indicated that DoCCaMK was most likely induced by the mycorrhizal fungus and participated in OM symbiosis.
In symbiotic seed germination, mutualistic fungi colonize orchid seeds and provide necessary nutrients for protocorm formation30. As a Ser/Thr protein kinase, CaMKII is well characterized and widely distributed and has been used as a model for CCaMK due to the significant structural and sequence similarities between the two proteins. For example, the CaM-binding domain of CCaMK shows high sequence identity (79%) to the α subunit of CaMKII, although there are also a few differences in the mechanism of action between them. Similar to CCaMK, autophosphorylation is also key to the regulation of CaMKII, and they both interpret a calcium spiking signal in a similar manner2. KN-93 is a synthesized inhibitor of CaMKII31. In our present study, we found that KN-93 could also inhibit D. officinale seed germination. During the same cultivation period as that for the control group without KN-93, we found that KN-93 could prevent seed germination to stage 5, halting development at stage 2 or even stage 1 (Fig. 4b). qRT-PCR and western blot analyses showed that in the KN-93 complement group, compared to that in the control group, CCaMK was downregulated not only at the mRNA level but also at the protein level in the SGS group (Figs. 1b and 3b). Therefore, KN-93 could inhibit CCaMK during D. officinale seed germination induced by Sebacina sp. In addition, in comparison to that of the SGS group, KN-93 could also decrease Ca2+ influx and reduce CCaMK activity during D. officinale seed germination. The results of the present study demonstrated that CCaMK participates in D. officinale seed germination. However, the interaction of other molecules with CCaMK in symbiotic germination of orchid seeds and the symbiotic mechanism between the orchid root and mycorrhizal fungus deserves further research.
Conclusions
CCaMK has been reported to play important roles in AM and nodulation symbioses, but little is known about its role in the OM. In this study, DoCCaMK-GFP fusion protein is found to locate in the cell nucleus and cell membrane. After Sebacina sp. colonization, CCaMK was highly expressed not only in the root, but also in the seed of D. officinale. In addition, during seed germination, the Ca2+ influx and the concentration of CCaMK was more in the SGS group than that of the UGS group. KN-93 can prohibit seed germination and inhibit CCaMK expression, furthermore, it can also reduce CCaMK activity and the Ca2+ influx during seed germination. This investigation of the characteristics and the expression patterns of CCaMK in D. officinale seed germination and seedling growth will provide key insight into the D. officinale-mycorrhizal fungal interaction in the future.
Methods
Plant materials, fungal strains and symbiotic germination
Capsules of D. officinale were collected from the Menghai experimental base of the Yunnan branch, Institute of Medicinal Plant Development, Chinese Academy of Medical Sciences & Peking Union Medical College Xishuangbanna, Yunnan, China. According to a previous study19, seeds in an in situ seed baiting technique experiment were divided into two groups. One group of seeds was cultured with Sebacina sp., which was isolated from collected germinated seeds according to a previous report18. The seeds and Sebacina sp. were cultivated in OMA medium (g L−1; oatmeal infusion-30 g and agar-15 g) and were defined as SGS. The other group of seeds that was cultured in OMA medium without any fungi was defined as UGS. The seeds were sown and cultured for 5 w in 20 cm diameter pots containing growth medium (bark:pebble:coarse humus = 3:1:1), as previously described19. Then, the cultivated samples in the SGS and UGS groups were all collected and immediately frozen in liquid nitrogen prior to RNA extraction.
Co-culture system between D. officinale seedlings and Sebacina sp
Sebacina sp. was cultivated in sawdust-based medium containing corn (166.0 g L−1), sawdust (668.0 g L−1), wheat bran (166.0 g L−1), KH2PO4 (2.0 g L−1), MgSO4·7 H2O (0.1 g L−1) and sucrose (10.0 g L−1). The substrate was then divided into 100 g aliquots in plastic bottles and autoclaved at 122 °C for 120 min32. In the SGS group, after Sebacina sp. was cultured for 1 month, 3 g of the fungal cultures was then transferred and inoculated next to the roots of three D. officinale seedlings in each flowerpot, which were 11 cm in diameter. The components of the medium in the flowerpot were sterile bark, small pebbles and rough humus, with a ratio of 3:1:1. In the UGS group, 3 g of the sawdust-based medium without fungus was added next to D. officinale. The experiment was repeated three times, with 30 replicates in each group. After cultivation for 60 d in a greenhouse at 25 °C, the root, stem and leaf in the SGS and UGS groups were collected and frozen in liquid nitrogen prior to RNA extraction and qRT-PCR analysis.
Total RNA extraction and complementary DNA synthesis
The total RNA of each frozen sample was extracted using an EASYspin Plus Kit (Aidlab, Beijing, China) according to the manufacturer’s protocol.
According to the Clontech SMARTer PCR cDNA Synthesis Kit, the samples in the SGS and UGS groups were used as the template, and ds cDNA was synthesized. Based on the EST sequence of the SSH cDNA library, the primers were designed for cDNA fragment amplification (Table 3). PCRs were conducted, and the final amplified PCR products were purified using a QIAquick PCR purification Kit (Qiagen, Hiden, Germany) and directly cloned into the pGEM-T Easy vector (Promega, Madison, WI, USA), which was then transformed into competent Escherichia coli DH5α cells (Trans, Beijing, China). Transformed cells were then screened by the blue/white colony method19.
Cloning and characterization of the full-length DoCCaMK gene from D. officinale
Isolation of the 3′/5′-end of CCaMK was performed using a SMART RACE cDNA Amplification Kit (Clontech, Japan) according to the manufacturer’s protocol. Based on the sequences of the SSH cDNA Library, four gene-specific primers (GSPs) were designed (Table 3).
The amplified products were purified, cloned into the pGEM-T Easy vector (Promega, Madison, WI, USA) and transformed into DH5α cells. Positive recombinant plasmids were screened and sequenced by Beijing Genewiz, Inc. After clustering and splicing with the core sequence, the ORFs of each sequence were obtained using BLASTX and ORF finder. Primers targeting the ORF determined in RACE screens were used to sequence and confirm the first reconstituted RACE sequences19. The nucleotide sequences of CCaMK reported in the present study are available in the GenBank database under accession number MG545931.
Effect of KN-93 on D. officinale seed germination and seedling growth
The seeds and roots of D. officinale cultured with Sebacina sp. as mentioned above were designated as the control group samples, while the samples of the group that received KN-93 (Sigma-Aldrich Co., St. Louis, MO, USA) treatment at a final concentration of 0.1 μmol/L in OMA medium were referred to as the experimental group samples. In addition, the seeds in the UGS group in OMA medium standed for the negative control group. After inoculation for 35 d, the seed germination in these three groups was subjected to morphological examination using a stereoscopic microscope (Leica DM2500). Additionally, the roots and the seeds in each group were collected for qRT-PCR and western blot analysis. The experiment was repeated three times, with 30 replicates in each group.
qRT-PCR analysis of DoCCaMK expression
According to the cDNA sequences of CCaMK in D. officinale, the primers were designed for qRT-PCR using Primer Premier 5.0 (Table 3). To analyse gene expression characteristics, different tissues of D. officinale were collected as follows: germinated seeds in the SGS group with and without KN-93 treatment, ungerminated seeds in the UGS group after cultivation in OMA medium for 35 d, and roots of seedlings with and without KN-93 treatment after cultivation with Sebacina sp. for 60 d. The roots, stems and leaves of the seedlings without fungus were also collected. qRT-PCR was conducted using a real-time SYBR Green kit (Takara, Dalian, China) and an ABI 7500 Real-Time PCR System (Applied Biosystems, Foster City, CA, USA). GAPDH was used as a reference control. The reaction was performed using the following conditions: denaturation at 95 °C for 30 s, followed by 40 cycles of amplification (95 °C for 5 s, 60 °C for 40 s). The gene expression ratio was evaluated by the comparative 2−ΔΔCt method of relative gene expression quantification19.
Phylogenetic analysis
To understand the phylogeny of DoCCaMK, the CCaMK sequences of several plants or crops, such as Glycine max, Oryza sativa, Lotus japonicas, Medicago truncatula and so on, were subjected to phylogenetic analysis. A phylogenetic tree was constructed using MEGA software version 6.0, using Pellia epiphylla CCaMK as an outgroup.
Construction of a plant expression vector and transient expression of a DoCCaMK-GFP fusion protein
Using a 163-hGFP plant expression vector containing 239 amino acids of GFP, an enhanced CCaMK-GFP fusion expression vector was constructed. Plasmid vector DNA (3 µg) was added to 6 µl of a suspension of gold nanoparticles (50 mg mL−1) with a diameter of 1.0 µm and mixed evenly. Next, 0.1 M spermidine (4 μL) and 6 μL of CaCl2 (12.5 M) were also added and blended thoroughly for 3 min, and then, the mixture was incubated in an ice bath for 15 min. After centrifugation at 12,000 rpm for 10 s, the liquid supernatant was discarded, and ethanol was added and vortexed. Finally, the gold nanoparticle sediment was collected, and ethanol was added to suspend the precipitation for further studies.
Onion bulb scale leaves were cut into strips of approximately 2 cm × 2 cm and were cultured in the centre of MS medium. The fusion expression vector containing the target gene and GFP in the control group was bombarded by the particle bombardment transforming method (1100 psi) to transform the onion epidermis20. After transformation, the onion epidermis was cultured in darkness for 24 h and photographed using a Zeiss laser scanning confocal microscope (LSM 510 System, Zeiss, Germany).
Recombinant protein expression and purification and western blot analysis
Using the pET-28a plant expression vector, the CCaMK-28a eukaryotic expression vector was constructed, and the specific primer sequences (CCaMK-TE) are listed in Table 3. CCaMK-TE-Pet-28a was expressed in E. coli BL21 (DE3) competent cells that were grown in 500 mL of LB broth containing 50 μg/mL kanamycin and shaking at 180 rpm at 37 °C until the OD 600 reached 0.8. Next, sterilized IPTG was added at a final concentration of 0.4 mM, followed by shaking at 250 rpm at 37 °C for 4 h. The cultures were then harvested by centrifugation at 10,000 × g (high-speed tabletop centrifuge, Sorvall ST8, Thermo Fisher Scientific Corporation, MA, USA) for 10 min at 4 °C. The pellets were re-suspended in 30 mL of lysis buffer containing 20 mM HEPES, 1 mM NaCl, 2 mM BME, 0.3 mg mL−1 lysozyme, and 5 mM imidazole (pH 8); incubated on ice for 30 min; frozen at −80 °C for 10 min; and thawed at 4 °C. After 3 repetitions, the protein was subjected to ultrasonication at 4 °C for 10 min. After centrifugation, the supernatant was collected, purified using a column packed with 1 mL of Ni-NTA resin in a gravity flow chromatography step and equilibrated with lysis buffer. The purified protein was eluted using a gradient of imidazole (10, 20, 80 and 300 mM) in lysis buffer. The purity of the protein samples was estimated by SDS-PAGE33.
According to the “one-step plant active protein extraction kit” (Sangon Biotech, Shanghai, China), different tissues of D. officinale were collected as mentioned above in the “qRT-PCR analysis of DoCCaMK expression” section. After sodium dodecyl sulfate polyacrylamide gel electrophoresis, DoCCaMK was electrotransferred onto a PVDF membrane (Millipore, USA), which was blocked for 4 h with a blocking solution containing 5% non-fat powdered milk. After incubation with a New Zealand rabbit anti-CCaMK primary antibody and an HRP-linked secondary antibody, the PVDF membrane was then developed using an enhanced chemiluminescence detection kit (TransGen Biotech, China) and analysed with a Gel Doc XR + imaging system (Bio-Rad, USA)33.
Real-time Ca2+ fluxes measurement of D. officinale seed germination using NMT
In this study, cellular Ca2+ was measured on the surface of the germinated or ungerminated seed during seed germination on 35d in the SGS, UGS and the samples of the SGS group that received KN-93 as mentioned above using Non-invasive Micro-test Technology System (NMT100 Series, Younger USA LLC, Amherst, MA, USA). Ca2+-sensitive microsensor was purchased from NMT Service Center, Xuyue Beijing (Beijing) Sci. & Tech Co. Ltd. The experimental voltage is +600 mV. The primary position (M1) of Ca2+-sensitive microsensor was placed 30 μm from the germinated or ungerminated seed surface, and further away position (M2) is 60 μm34.
CCaMK concentration detection during D. officinale seed germination
CCaMK standard substance with different concentrations of 200, 100, 50, 25, 12.5, 6.25 and 3.12 (U/ml) was studied. The protocol of the standard curve and the CCaMK concentration detection of the samples were conducted according to the instruction of “Cat No.2Pl-KMLJ91921p plant (SnRKs CCaMK) Camilo ELISA kit”, Camilo biology.
CCaMK activity of D. officinale seed during germination was detected and three groups were divided in the experiment. Among them, the samples of the group that received KN-93 treatment at a final concentration of 0.1 μmol/L in OMA medium with Sebacina sp. were referred to as the KN-93 complement group. After germination for 35d, 0.2 g of the ungerminated or geminated seeds in triple of each group of the UGS (control), SGS and KN-93 complement groups were respectively collected from the petri dishes, weighed accurately and then grinded with liquid nitrogen. The CCaMK standard substance diluent was regarded as the negative control and the deionized water was treated as the blank control. Subsequently, 1.8 mL PBS buffer solution (0.01 M, pH 7.4) were added and centrifuged at 4 °C, 8000 rpm for 30 min. The supernate of the samples or the CCaMK standard with different concentrations (100 μL) were added to the coated plate and incubated at 37 °C for 90 min. After the plate was washed by TBS washing solution twice and 100 μL of SnRKs CCaMK antibody was added and the plate was incubated at 37 °C for 60 min. Subsequently, the plate was washed for three times and 100 μL of enzyme combo except for the blank control and incubated in the dark for 30 min at 37 °C. The plate was washed for 5 times and 100 μL TMB color solution was added and the reaction lasted for no more than 30 min. At last, 100 μL stop solution was added and the OD (450 nm) was measured in 10 min with EnSpire Multimode Reader (PerkinElmer, America).
Data analysis
The data were analyzed with one-way ANOVA and the statistical analyse was performed using SPSS 11.0. Data were presented as means ± SD from at least three independent experiments. P values < 0.05 were considered significant difference.
References
Spatafora, J. W. et al. A phylum-level phylogenetic classification of zygomycete fungi based on genome-scale data. Mycologia 108, 1028–1046 (2016).
Harrison, J. Characterisation of CCaMK in symbiosis signalling. Thesis submitted to the University of East Anglia for the degree of Doctor of Philosophy 41–51 (2015).
Oldroyd, G. E. D. Speak, friend, and enter: signalling systems that promote beneficial symbiotic associations in plants. Nature Reviews Microbiology 11, 252–263 (2013).
Suetsugu, K. et al. Comparison of green and albino individuals of the partially mycoheterotrophic orchid Epipactis helleborine on molecular identities of mycorrhizal fungi, nutritional modes and gene expression in mycorrhizal roots. Molecular Ecology 26, 1652–1669 (2017).
Wang, J. P., Jean-Pierre, M., You-Ping, X. & Xin-Zhong, C. Phylogeny of Plant Calcium and Calmodulin-Dependent Protein Kinases (CCaMKs) and Functional Analyses of Tomato CCaMK in Disease Resistance. Frontiers in Plant Science 6 (2015).
Gleason, C. et al. Nodulation independent of rhizobia induced by a calcium-activated kinase lacking autoinhibition. Nature 441, 1149–1152 (2006).
Miller, J. B. et al. Calcium/Calmodulin-Dependent Protein Kinase Is Negatively and Positively Regulated by Calcium, Providing a Mechanism for Decoding Calcium Responses during Symbiosis Signaling. Plant Cell 25, 5053–5066 (2013).
Li, T., Yuan, L., Zhang, J., Jiao, J. J. & Qi, J. S. Real-time measurement of Ca2+ flux in hippocampal slice with non-invasive microtest technique. Acta Physiologica Sinica 69(4), 467–476 (in Chinese) (2017).
Sumi, M. et al. The newly synthesized selective Ca2+/calmodulin dependent protein kinase II inhibitor KN-93 reduces dopamine contents in PC12h cells. Res. Commun 181, 968 (1991).
Judelson, H. S. & Roberts, S. Novel Protein Kinase Induced during Sporangial Cleavage in the Oomycete Phytophthora infestans. Eukaryotic Cell 1, 687–695 (2002).
Khamchatra Using in situ seed baiting technique to isolate and identify endophytic and mycorrhizal fungi from seeds of a threatened epiphytic orchid, dendrobium friedericksianum rchb.f. (orchidaceae). Agriculture & Natural Resources 50(1), 8–13 (2016).
Guo, L. C. et al. Differentiation of the Chinese minority medicinal plant genus Berchemia spp. by evaluating three candidate barcodes. Springerplus 5, 658 (2016).
Yan, L. et al. The Genome of Dendrobium officinale Illuminates the Biology of the Important Traditional Chinese Orchid Herb. Molecular Plant 8, 922–934 (2015).
He, C. M. et al. Molecular cloning and functional analysis of thephosphomannomutase(PMM) gene fromDendrobium officinaleand evidence for the involvement of an abiotic stress response during germination. Protoplasma 254, 1693–1704 (2017).
Ding, G. et al. Preliminary geoherbalism study ofDendrobium officinalefood by DNA molecular markers. European Food Research & Technology 227, 1283–1286 (2008).
Teixeira da Silva, J. A. et al. Symbiotic in vitro seed propagation ofDendrobium: fungal and bacterial partners and their influence on plant growth and development. Planta 242, 1–22 (2015).
Rajjou, L. et al. Seed Germination and Vigor. Annual Review of Plant Biology 63, 507–533 (2012).
Wang, H., Fang, H., Wang, Y., Duan, L. & Guo, S. In situ seed baiting techniques inDendrobium officinaleKimuraet Migo andDendrobium nobileLindl.: the endangered Chinese endemicDendrobium(Orchidaceae). World Journal of Microbiology & Biotechnology 27, 2051–2059 (2011).
Zhao, M. M., Zhang, G., Zhang, D. W., Hsiao, Y. Y. & Guo, S. X. ESTs Analysis Reveals Putative Genes Involved in Symbiotic Seed Germination in Dendrobium officinale. Plos One 8, e72705 (2013).
Osaki, Y. & Kodama, Y. Particle bombardment and subcellular protein localization analysis in the aquatic plant Egeria densa. Peer J 5, e3779 (2017).
Chen, L., Wang, Y.-C., Qin, L.-Y., He, H.-Y. & Zhang, H.-B. Dynamics of fungal communities during Gastrodia elata growth. BMC Microbiology 19 (2019).
Stewart, S. L., Zettler, L., Minso, J. & Brown, P. M. Symbiotic germination and reintroduction of Spiranthes brevilabris Lindley, an endangered orchid native to Florida. Selbyana 24, 64–70 (2003).
Takeda, N., Maekawa, T. & Hayashi, M. Nuclear-Localized and Deregulated Calcium- and Calmodulin-Dependent Protein Kinase Activates Rhizobial and Mycorrhizal Responses in Lotus japonicus. Plant Cell 24, 810–822 (2012).
Wang, B. et al. Presence of three mycorrhizal genes in the common ancestor of land plants suggests a key role of mycorrhizas in the colonization of land by plants. New Phytologist 186, 514–525 (2010).
Ma, F. et al. Nitric oxide-activated calcium/calmodulin-dependent protein kinase regulates the abscisic acid-induced antioxidant defence in maize. Journal of Experimental Botany 63, 4835–4847 (2012).
Charpentier, M. et al. Nuclear-localized cyclic nucleotide-gated channels mediate symbiotic calcium oscillations. Science 352, 1102–1105 (2016).
Jin, Y., Liu, H., Luo, D., Yu, N. & Wang, E. DELLA proteins are common components of symbiotic rhizobial and mycorrhizal signaling pathways. Nature Communications 7, 12433 (2016).
Chabaud, M. et al. Arbuscular mycorrhizal hyphopodia and germinated spore exudates trigger Ca2+ spiking in the legume and nonlegume root epidermis. New Phytol 189, 347–355 (2011).
Zhu, Y., Yan, J., Liu, W., Liu, L. & Zhang, A. Phosphorylation of a NAC transcription factor by ZmCCaMK regulates abscisic acid-induced antioxidant defense in maize. Plant Physiology 171, pp.00168.02016 (2016).
Li, Y. Y. et al. Immunolocalization and Changes of Hydroxyproline-Rich Glycoproteins During Symbiotic Germination of Dendrobium officinale. Frontiers in Plant Science 9, 552 (2018).
Wang, W. L. et al. KN-93, A CaMK II Inhibitor, Suppresses Ventricular Arrhythmia Induced by LQT2 Without Decreasing TDR. Journal of huazhong university of science and technology, medical science in English and German 33, 636–639 (2013).
Xing, Y. M. et al. Sclerotial Formation of Polyporus umbellatus by Low Temperature Treatment under Artificial Conditions. Plos One 8, e56190 (2013).
Liu, M., Zhang, D., Xing, Y. & Guo, S. Cloning and expression of three thaumatin-like protein genes from Polyporus umbellatus. Acta Pharmaceutica Sinica B, S2211383516303872.(2017).
Babourina, O., Newman, I. & Shabala, S. Blue light-induced kinetics of H and Ca2+ fluxes in etiolated. wild-type and phototropin-mutant Arabidopsis seedlings. PNAS 99, 2433–2438 (2002).
Acknowledgements
This research was financially supported by the CAMS Innovation Fund for Medical Sciences (CIFMS) (2017-I2M-3-013), National Natural Sciences Foundation of China (No. NSFC 81573527) and Peking Union Medical College Discipline Construction Project (Tsinghua 211-201920100901).
Author information
Authors and Affiliations
Contributions
S.X.G. and J.C.: experimental design and manuscript review; Y.M.X.: manuscript writing, manuscript review and performed part of the experiments; M.M.Z.: performed experiments and manuscript review; L.C.G. and B.L.: data analysis. All authors have read and approved the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Xing, YM., Zhao, MM., Guo, LC. et al. Identification and expression of DoCCaMK during Sebacina sp. symbiosis of Dendrobium officinale. Sci Rep 10, 9733 (2020). https://doi.org/10.1038/s41598-020-66616-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-66616-3
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.