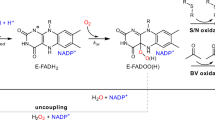
Abstract
Hydroxytryptophan serves as a chemical precursor to a variety of bioactive specialized metabolites, including the human neurotransmitter serotonin and the hormone melatonin. Although the human and animal routes to hydroxytryptophan have been known for decades, how bacteria catalyze tryptophan indole hydroxylation remains a mystery. Here we report a class of tryptophan hydroxylases that are involved in various bacterial metabolic pathways. These enzymes utilize a histidine-ligated heme cofactor and molecular oxygen or hydrogen peroxide to catalyze regioselective hydroxylation on the tryptophan indole moiety, which is mechanistically distinct from their animal counterparts from the nonheme iron enzyme family. Through genome mining, we also identify members that can hydroxylate the tryptophan indole ring at alternative positions. Our results not only reveal a conserved way to synthesize hydroxytryptophans in bacteria but also provide a valuable enzyme toolbox for biocatalysis. As proof of concept, we assemble a highly efficient pathway for melatonin in a bacterial host.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The data that support the findings of this study are available within the main text and its Supplementary Information files. Data are also available from the corresponding author upon request. Source data are provided with this paper.
References
Alkhalaf, L. M. & Ryan, K. S. Biosynthetic manipulation of tryptophan in bacteria: pathways and mechanisms. Chem. Biol. 22, 317–328 (2015).
Axelrod, J. & Weissbach, H. Enzymatic O-methylation of N-acetylserotonin to melatonin. Science 131, 1312–1312 (1960).
Maeda, H. & Dudareva, N. The shikimate pathway and aromatic amino acid biosynthesis in plants. Ann. Rev. Plant Biol. 63, 73–105 (2012).
Lee, J.-H., Wood, T. K. & Lee, J. Roles of indole as an interspecies and interkingdom signaling molecule. Trends Microbiol. 23, 707–718 (2015).
Kaur, H. et al. The antibiotic darobactin mimics a β-strand to inhibit outer membrane insertase. Nature 593, 125–129 (2021).
Fricke, J., Blei, F. & Hoffmeister, D. Enzymatic synthesis of psilocybin. Angew. Chem. Int. Ed. Engl. 56, 12352–12355 (2017).
Caputi, L. et al. Missing enzymes in the biosynthesis of the anticancer drug vinblastine in Madagascar periwinkle. Science 360, 1235–1239 (2018).
Daniels, P. N. et al. A biosynthetic pathway to aromatic amines that uses glycyl-tRNA as nitrogen donor. Nat. Chem. 14, 71–77 (2022).
Hong, B. et al. Biosynthesis of strychnine. Nature 607, 617–622 (2022).
Adak, S., Lukowski, A. L., Schäfer, R. J. B. & Moore, B. S. From tryptophan to toxin: nature’s convergent biosynthetic strategy to aetokthonotoxin. J. Am. Chem. Soc. 144, 2861–2866 (2022).
Yu, J. et al. Parallel pathways for serotonin biosynthesis and metabolism in C. elegans. Nat. Chem. Biol. https://doi.org/10.1038/s41589-022-01148-7 (2022).
Fitzpatrick, P. F. Tetrahydropterin-dependent amino acid hydroxylases. Annu. Rev. Biochem. 68, 355–381 (1999).
Walther, D. J. et al. Synthesis of serotonin by a second tryptophan hydroxylase isoform. Science 299, 76 (2003).
Barry, S. M. et al. Cytochrome P450-catalyzed l-tryptophan nitration in thaxtomin phytotoxin biosynthesis. Nat. Chem. Biol. 8, 814–816 (2012).
Dong, C. et al. Tryptophan 7-halogenase (PrnA) structure suggests a mechanism for regioselective chlorination. Science 309, 2216–2219 (2005).
Winkelblech, J. et al. Tryptophan C5-, C6- and C7-prenylating enzymes displaying a preference for C-6 of the indole ring in the presence of unnatural dimethylallyl diphosphate analogues. Adv. Synth. Catal. 357, 975–986 (2015).
Tsunematsu, Y. et al. Distinct mechanisms for spiro-carbon formation reveal biosynthetic pathway crosstalk. Nat. Chem. Biol. 9, 818–825 (2013).
Farrow, S. C. et al. Cytochrome P450 and O-methyltransferase catalyze the final steps in the biosynthesis of the anti-addictive alkaloid ibogaine from Tabernanthe iboga. J. Biol. Chem. 293, 13821–13833 (2018).
Chang, F.-Y., Ternei, M. A., Calle, P. Y. & Brady, S. F. Discovery and synthetic refactoring of tryptophan dimer gene clusters from the environment. J. Am. Chem. Soc. 135, 17906–17912 (2013).
Muliandi, A. et al. Biosynthesis of the 4-methyloxazoline-containing nonribosomal peptides, JBIR-34 and -35, in Streptomyces sp. Sp080513GE-23. Chem. Biol. 21, 923–934 (2014).
Hoshino, T. Violacein and related tryptophan metabolites produced by Chromobacterium violaceum: biosynthetic mechanism and pathway for construction of violacein core. Appl. Microbiol. Biotechnol. 91, 1463–1475 (2011).
Wang, H. et al. Metabolic pathway engineering for high-level production of 5-hydroxytryptophan in Escherichia coli. Metab. Eng. 48, 279–287 (2018).
Liu, J., Ng, T., Rui, Z., Ad, O. & Zhang, W. Unusual acetylation-dependent reaction cascade in the biosynthesis of the pyrroloindole drug physostigmine. Angew. Chem. Int. Ed. Engl. 53, 136–139 (2014).
Smith, D. R. M. et al. An unusual flavin-dependent halogenase from the metagenome of the marine sponge Theonella swinhoei WA. ACS Chem. Biol. 12, 1281–1287 (2017).
Shi, X. et al. Enzymatic tailoring in luzopeptin biosynthesis involves cytochrome P450-mediated carbon–nitrogen bond desaturation for hydrazone formation. Angew. Chem. Int. Ed. Engl. 60, 19821–19828 (2021).
Hirose, Y. et al. Involvement of common intermediate 3-hydroxy-l-kynurenine in chromophore biosynthesis of quinomycin family antibiotics. J. Antibiot. 64, 117–122 (2011).
Zallot, R., Oberg, N. & Gerlt, J. A. The EFI web resource for genomic enzymology tools: leveraging protein, genome, and metagenome databases to discover novel enzymes and metabolic pathways. Biochemistry 58, 4169–4182 (2019).
Pogorevc, D. et al. Biosynthesis and heterologous production of argyrins. ACS Synth. Biol. 8, 1121–1133 (2019).
Kim, E. et al. Characterization of the ohmyungsamycin biosynthetic pathway and generation of derivatives with improved antituberculosis activity. Biomolecules 9, 672 (2019).
Forouhar, F. et al. Molecular insights into substrate recognition and catalysis by tryptophan 2,3-dioxygenase. Proc. Natl Acad. Sci. USA 104, 473–478 (2007).
Basran, J. et al. The mechanism of formation of n-formylkynurenine by heme dioxygenases. J. Am. Chem. Soc. 133, 16251–16257 (2011).
Shin, I. et al. Stepwise O-atom transfer in heme-based tryptophan dioxygenase: role of substrate ammonium in epoxide ring opening. J. Am. Chem. Soc. 140, 4372–4379 (2018).
Basran, J., Booth, E. S., Lee, M., Handa, S. & Raven, E. L. Analysis of reaction intermediates in tryptophan 2,3-dioxygenase: a comparison with indoleamine 2,3-dioxygenase. Biochemistry 55, 6743–6750 (2016).
Geng, J., Weitz, A. C., Dornevil, K., Hendrich, M. P. & Liu, A. Kinetic and spectroscopic characterization of the catalytic ternary complex of tryptophan 2,3-dioxygenase. Biochemistry 59, 2813–2822 (2020).
Sacramento, J. J. D. & Goldberg, D. P. Oxidation of an indole substrate by porphyrin iron(III) superoxide: relevance to indoleamine and tryptophan 2,3-dioxygenases. Chem. Commun. 56, 3089–3092 (2020).
Mondal, P., Rajapakse, S. & Wijeratne, G. B. Following nature’s footprint: mimicking the high-valent heme-oxo mediated indole monooxygenation reaction landscape of heme enzymes. J. Am. Chem. Soc. 144, 3843–3854 (2022).
Wei, Y. et al. Directed evolution of a tryptophan 2,3-dioxygenase for the diastereoselective monooxygenation of tryptophans. Angew. Chem. Int. Ed. Engl. 59, 3043–3047 (2020).
Connor, K. L., Colabroy, K. L. & Gerratana, B. A heme peroxidase with a functional role as an l-tyrosine hydroxylase in the biosynthesis of anthramycin. Biochemistry 50, 8926–8936 (2011).
Tang, M.-C., Fu, C.-Y. & Tang, G.-L. Characterization of SfmD as a heme peroxidase that catalyzes the regioselective hydroxylation of 3-methyltyrosine to 3-hydroxy-5-methyltyrosine in saframycin A biosynthesis. J. Biol. Chem. 287, 5112–5121 (2012).
Shin, I., Wang, Y. & Liu, A. A new regime of heme-dependent aromatic oxygenase superfamily. Proc. Natl Acad. Sci. USA 118, e2106561118 (2021).
Shin, I. et al. A novel catalytic heme cofactor in SfmD with a single thioether bond and a bis-His ligand set revealed by a de novo crystal structural and spectroscopic study. Chem. Sci. 12, 3984–3998 (2021).
Sambrook, J. Molecular Cloning: A Laboratory Manual (CSHL Press, 2001).
Kieser, T. Practical Streptomyces Genetics (John Innes Foundation, 2000).
Du, Y.-L., Dalisay, D. S., Andersen, R. J. & Ryan, K. S. N-Carbamoylation of 2,4-diaminobutyrate reroutes the outcome in padanamide biosynthesis. Chem. Biol. 20, 1002–1011 (2013).
Barr, I. & Guo, F. Pyridine hemochromagen assay for determining the concentration of heme in purified protein solutions. Bio Protoc. 5, e1594 (2015).
Acknowledgements
This work was supported by National Key R&D Program of China (2018YFA0903200) and the National Natural Science Foundation of China (32122005) to Y.-L.D.
Author information
Authors and Affiliations
Contributions
X.S. and G.Z. carried out bioinformatic, genetic and biochemical work and performed metabolic analysis, compound isolation and structure elucidation. H.L., Z.Z. and W.L. carried out metabolic engineering, metabolic analysis and flask fermentation. X.S., M.W. and Y.-L.D. designed the study, analyzed results and wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Chemical Biology thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 In vitro biochemical assays and kinetic study of Luz15 with Trp in the presence of O2 and ascorbate.
a, Time-course studies of the in vitro reaction of Luz15 by HPLC. The conversion rate at 24 h is 12%. b, Kinetic study of Luz15 with Trp in the presence of O2 and ascorbate (25 mM). The kinetic parameters are Km = 3.1 ± 4 mM and kcat = 2.2 ± 0.1 h−1. Assays were run in triplicate and kinetic values throughout this paper are reported as mean ± s.d. of three replicates. Note: the kinetic study of Luz15 with H2O2 was not performed due to the rapid inactivation of Luz15 (the formation of the product plateaued after the first minute of turnover). However, these values were determined for other more stable Luz15 homologues (see the below Extended Data Figs).
Extended Data Fig. 2 In vitro biochemical assays and kinetic studies of PsmG.
a, Time-course studies of the in vitro reaction of PsmG with Trp in the presence of O2 and ascorbate by HPLC. The conversion rate at 24 h is 5%. b, Kinetic study of PsmG with Trp in the presence of H2O2 (3 mM). The kinetic parameters are Km = 406 ± 77 μM and kcat = 10.8 ± 0.5 min−1. c, Kinetic study of PsmG with Trp in the presence of O2 and ascorbate (25 mM). The kinetic parameters are Km = 1.7 ± 0.4 mM and kcat = 2.1 ± 0.2 h−1. Assays were run in triplicate, and data are reported as mean ± s.d. of three replicates.
Extended Data Fig. 3 In vitro biochemical assays and kinetic studies of KerI.
a, Time-course studies of the in vitro reaction of KerI with Trp in the presence of O2 and ascorbate by HPLC. The conversion rate at 24 h is 22%. b, Kinetic study of KerI with Trp in the presence of H2O2 (3 mM). The kinetic parameters are Km = 2.6 ± 0.3 mM and kcat = 87 ± 5 min−1. c, Kinetic study of KerI with Trp in the presence of O2 and ascorbate (25 mM). The kinetic parameters are Km = 448 ± 57 μM and kcat = 2.4 ± 0.1 h−1. Assays were run in triplicate and data are reported as mean ± s.d. of three replicates. It is worth mentioning that, among the Trp 5-hydroxylases we identified, KerI is the most stable protein under in vitro conditions. KerI does not precipitate at the concentrations of H2O2 we used (up to 4 mM) and has the highest conversion rate.
Extended Data Fig. 4 In vivo production and identification of 4-HTP and 6-HTP.
a, Metabolic profiling of the E. coli strain expressing arg5 and SaT6H (a Trp 6-hydoxylase, see later in the main text) by HPLC analysis (detection wavelength: 254 nm). Assays were run in triplicate and representative results are shown. b, Mass value for the strain-specific peak from E. coli (ΔtnaA)/pET22b-arg5. c, Mass value for the strain-specific peak from E. coli (ΔtnaA)/pET22b-SaT6H. d, Structure elucidation of isolated 4-HTP and 6-HTP. Key COSY and HMBC correlations are displayed.
Extended Data Fig. 5 In vitro biochemical assays of OhmK and Arg5 in the presence of O2 and ascorbate.
a, Time-course studies of the in vitro reaction of OhmK with Trp in the presence of O2 and ascorbate by HPLC. b, Time-course studies of the in vitro reaction of Arg5 with Trp in the presence of O2 and ascorbate by HPLC. c, The reaction product 4-HTP undergoes further side reactions to form blue compounds in the reaction with H2O2. This blueing reaction likely occurs through a similar oxidative oligomerization mechanism to that of psilocin (4-hydroxy-N,N-dimethyltryptamine) from mushrooms (please also see Supplementary Fig. 24). Note: the kinetic parameters of the Trp 4-hydroxylases were not determined due to the above oligomerization side reaction with H2O2, and the low conversion rates with O2 and ascorbate (approximately one turnover). Assays were run in triplicate, and representative results are shown.
Extended Data Fig. 6 In vitro biochemical assays and kinetic studies of SaT6H.
a, Time-course studies of the in vitro reaction of SaT6H with Trp in the presence of O2 and ascorbate by HPLC. The conversion rate at 30 h is 86%. b, Kinetic study of SaT6H with Trp in the presence of H2O2 (3 mM). The kinetic parameters are Km = 4.3 ± 0.9 mM and kcat = 113 ± 12 min−1.c, Kinetic study of SaT6H with Trp in the presence of O2 and ascorbate (25 mM). The kinetic parameters are Km = 1.1 ± 0.2 mM and kcat = 23 ± 2 h−1. Assays were run in triplicate, and data are reported as mean ± s.d. of three replicates.
Extended Data Fig. 7 In vitro biochemical assays and kinetic studies of AxT6H.
a, Time-course studies of the in vitro reaction of AxT6H with Trp in the presence of O2 and ascorbate by HPLC. The conversion rate at 30 h is 37%. b, Kinetic study of AxT6H with Trp in the presence of H2O2 (3 mM). The kinetic parameters are Km = 1.3 ± 0.4 mM and kcat = 31 ± 5 min−1.c, Kinetic study of AxT6H with Trp in the presence of O2 and ascorbate (25 mM). The kinetic parameters are Km = 994 ± 99 μM and kcat = 2.5 ± 0.1 h−1. Assays were run in triplicate, and data are reported as mean ± s.d. of three replicates.
Supplementary information
Supplementary Information
Supplementary Figs. 1–61 and Tables 1–7.
Supplementary Data 1
DNA sequences for synthetic genes.
Supplementary Data 2
Statistical source data for Supplementary Fig. 35.
Source data
Source Data Figs. 3 and 5
Statistical source data.
Source Data Extended Data Figs. 1, 2, 3, 6 and 7
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shi, X., Zhao, G., Li, H. et al. Hydroxytryptophan biosynthesis by a family of heme-dependent enzymes in bacteria. Nat Chem Biol 19, 1415–1422 (2023). https://doi.org/10.1038/s41589-023-01416-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41589-023-01416-0