Abstract
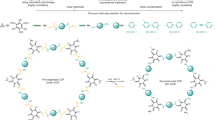
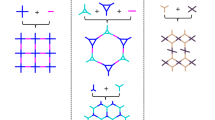
Mono- or few-layer sheets of covalent organic frameworks (COFs) represent an attractive platform of two-dimensional materials that hold promise for tailor-made functionality and pores, through judicious design of the COF building blocks. But although a wide variety of layered COFs have been synthesized, cleaving their interlayer stacking to obtain COF sheets of uniform thickness has remained challenging. Here, we have partitioned the interlayer space in COFs by incorporating pseudorotaxane units into their backbones. Macrocyclic hosts based on crown ethers were embedded into either a ditopic or a tetratopic acylhydrazide building block. Reaction with a tritopic aldehyde linker led to the formation of acylhydrazone-based layered COFs in which one basal plane is composed of either one layer, in the case of the ditopic macrocyclic component, or two adjacent layers covalently held together by its tetratopic counterpart. When a viologen threading unit is introduced, the formation of a host–guest complex facilitates the self-exfoliation of the COFs into crystalline monolayers or bilayers, respectively.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
X-ray crystallographic data have been deposited at the Cambridge Crystallographic Data Centre (http://www.ccdc.cam.ac.uk/) with CCDC reference numbers 1863696 (single-crystal Cy1-MVPF6), and 2006788 (single-crystal CyHz0-MVPF6). A copy of the data can be obtained free of charge via https://www.ccdc.cam.ac.uk/structures/. All other data supporting the findings of this study are available within the paper and its Supplementary Information. Data are also available from the corresponding author upon reasonable request.
References
Colson,, J. W. & Dichtel,, W. R. Rationally synthesized two-dimensional polymers. Nat. Chem. 5, 453–465 (2013).
Ding, S.-Y. & Wang, W. Covalent organic frameworks (COFs): from design to applications. Chem. Soc. Rev. 42, 548–568 (2013).
Huang, N., Wang, P. & Jiang, D. Covalent organic frameworks: a materials platform for structural and functional designs. Nat. Rev. Mater. 1, 16068 (2016).
Segura, J. L., Mancheno, M. J. & Zamora, F. Covalent organic frameworks based on Schiff-base chemistry: synthesis, properties and potential applications. Chem. Soc. Rev. 45, 5635–5671 (2016).
Diercks, C. S. & Yaghi, O. M. The atom, the molecule, and the covalent organic framework. Science 355, eaal1585 (2017).
Li,, X., Yadav,, P. & Loh,, K. P. Function-oriented synthesis of two-dimensional (2D) covalent organic frameworks – from 3D solids to 2D sheets. Chem. Soc. Rev. 49, 4835–4866 (2020).
Xu, H.-S. et al. Single crystal of a one-dimensional metallo-covalent organic framework. Nat. Commun. 11, 1434 (2020).
Côté, A. P. et al. Porous, crystalline, covalent organic frameworks. Science 310, 1166–1170 (2005).
El-Kaderi, H. M. et al. Designed synthesis of 3D covalent organic frameworks. Science 316, 268–272 (2007).
Lyle, S. J., Waller, P. J. & Yaghi, O. M. Covalent organic frameworks: organic chemistry extended into two and three dimensions. Trends Chem. 1, 172–184 (2019).
Berlanga, I. et al. Delamination of layered covalent organic frameworks. Small 7, 1207–1211 (2011).
Bunck, D. N. & Dichtel, W. R. Bulk synthesis of exfoliated two-dimensional polymers using hydrazone-linked covalent organic frameworks. J. Am. Chem. Soc. 135, 14952–14955 (2013).
Chandra, S. et al. Chemically stable multilayered covalent organic nanosheets from covalent organic frameworks via mechanical delamination. J. Am. Chem. Soc. 135, 17853–17861 (2013).
Peng, Y. et al. Ultrathin two-dimensional covalent organic framework nanosheets: preparation and application in highly sensitive and selective DNA detection. J. Am. Chem. Soc. 139, 8698–8704 (2017).
Khayum, M. A. et al. Chemically delaminated free-standing ultrathin covalent organic nanosheets. Angew. Chem. Int. Ed. 55, 15604–15608 (2016).
Colson, J. W. et al. Oriented 2D covalent organic framework thin films on single-layer graphene. Science 332, 228–231 (2011).
Matsumoto, M. et al. Lewis-acid-catalyzed interfacial polymerization of covalent organic framework films. Chem 4, 308–317 (2018).
Hao, Q. et al. Confined synthesis of two-dimensional covalent organic framework thin films within superspreading water layer. J. Am. Chem. Soc. 140, 12152–12158 (2018).
Zhou, T.-Y., Lin, F., Li, Z.-T. & Zhao, X. Single-step solution-phase synthesis of free-standing two-dimensional polymers and their evolution into hollow spheres. Macromolecules 46, 7745–7752 (2013).
Dienstmaier, J. F. et al. Synthesis of well-ordered COF monolayers: surface growth of nanocrystalline precursors versus direct on-surface polycondensation. ACS Nano 5, 9737–9745 (2011).
Xu, L. et al. Surface-confined single-layer covalent organic framework on single-layer graphene grown on copper foil. Angew. Chem. Int. Ed. 53, 9564–9568 (2014).
Chen, C. et al. Local electronic structure of a single-layer porphyrin-containing covalent organic framework. ACS Nano 12, 385–391 (2018).
Joshi, T. et al. Local electronic structure of molecular heterojunctions in a single-layer 2D covalent organic framework. Adv. Mater. 0, 1805941 (2018).
Lindoy L. F. The Chemistry of Macrocyclic Ligand Complexes (Cambridge Univ. Press, 1989).
Stoddart, J. F. The chemistry of the mechanical bond. Chem. Soc. Rev. 38, 1802–1820 (2009).
Rambo, B. M., Gong, H.-Y., Oh, M. & Sessler, J. L. The “Texas-Sized” Molecular Box: A versatile building block for the construction of anion-directed mechanically interlocked structures. Acc. Chem. Res. 45, 1390–1401 (2012).
Stoddart, J. F. Mechanically interlocked molecules (MIMs)—molecular shuttles, switches, and machines (Nobel Lecture). Angew. Chem., Int. Ed. 56, 11094–11125 (2017).
Li, Q. et al. Docking in metal–organic frameworks. Science 325, 855 (2009).
Vukotic, V. N., Harris, K. J., Zhu, K., Schurko, R. W. & Loeb, S. J. Metal–organic frameworks with dynamic interlocked components. Nat. Chem. 4, 456–460 (2012).
Li, X. et al. Rapid, scalable construction of highly crystalline acylhydrazone two-dimensional covalent organic frameworks via dipole-induced antiparallel stacking. J. Am. Chem. Soc. 142, 4932–4943 (2020).
Allwood B. L., Spencer N., Shahriari-Zavareh H., Stoddart J. F. & Williams D. J. Complexation of Paraquat by a bisparaphenylene-34-crown-10 derivative. J. Chem. Soc. Chem. Commun. 1064–1066 (1987).
Li, X. et al. Tuneable near white-emissive two-dimensional covalent organic frameworks. Nat. Commun. 9, 2335 (2018).
Anelli, P. L. et al. Molecular meccano. 1. [2]Rotaxanes and a [2]catenane made to order. J. Am. Chem. Soc. 114, 193–218 (1992).
Poizat, O., Sourisseau, C. & Mathey, Y. Vibrational study of the methyl viologen dication MV2+ and radical cation MV˙+ in several salts and as an intercalate in some layered MPS3 compounds. J. Chem. Soc. Faraday Trans. 80, 3257–3274 (1984).
Shearer, C. J., Slattery, A. D., Stapleton, A. J., Shapter, J. G. & Gibson, C. T. Accurate thickness measurement of graphene. Nanotechnology 27, 125704 (2016).
Acknowledgements
K.P.L. acknowledges NRF-CRP grant “Two-Dimensional Covalent Organic Framework: Synthesis and Applications,” grant number NRF-CRP16-2015-02, funded by National Research Foundation, Prime Minister’s Office, Singapore. We acknowledge the use of the imaging facilities at the Center for BioImaging Sciences. Funding for the K2-IS camera is provided by the Singapore National Research Foundation’s Competitive Research Program (NRF-CRP16-2015-05). We thank S. Xiangyan for the solid-state NMR measurement. We thank C. Hyun and T. J. Shin for the synchrotron-based GIXRD measurements.
Author information
Authors and Affiliations
Contributions
Under the supervision of K.P.L., X.L. designed and performed most of the experiments, including synthesis and characterization of COFs and their model compounds. X.L., H.-S.X. and Q.G. discussed the manuscript. K.L. performed the AFM measurements. Under the supervision of U.M., S.W.C. performed and analysed the TEM of COFs, and N.J. performed and analysed SAED of COFs. X.Z. analysed the TEM results. H.X. performed the Q-plus AFM measurement. Under the supervision of S.Y.Q., J.Q. performed the DFT theoretical calculations. I.-H.P. resolved the single-crystal structures of the model compounds. X.L. and K.P.L. co-wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary text, Figs. 1–35
Supplementary Table 1
Supplementary Tables 1–7
Supplementary Data 2
Single-crystal file of Cy1-MVPF6
Supplementary Data 3
Single-crystal file of CyHz0-MVPF6
Rights and permissions
About this article
Cite this article
Li, X., Xu, HS., Leng, K. et al. Partitioning the interlayer space of covalent organic frameworks by embedding pseudorotaxanes in their backbones. Nat. Chem. 12, 1115–1122 (2020). https://doi.org/10.1038/s41557-020-00562-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-020-00562-5
This article is cited by
-
Linkage conversions in single-crystalline covalent organic frameworks
Nature Chemistry (2024)
-
Covalent organic framework monolayer: Accurate syntheses and advanced application
Nano Research (2024)
-
Covalent organic frameworks
Nature Reviews Methods Primers (2023)
-
Hierarchies of Hofstadter butterflies in 2D covalent organic frameworks
npj 2D Materials and Applications (2023)
-
Hydrogen-bonding and π-π interaction promoted solution-processable covalent organic frameworks
Nature Communications (2023)