Abstract
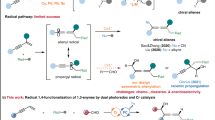
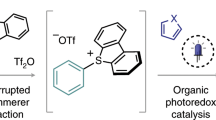
Photoredox catalysis, especially in combination with transition metal catalysis, can produce redox states of transition metal catalysts to facilitate challenging bond formations that are not readily accessible in conventional redox catalysis. For arene functionalization, metallophotoredox catalysis has successfully made use of the same leaving groups as those valuable in conventional cross-coupling catalysis, such as bromide. Yet the redox potentials of common photoredox catalysts are not sufficient to reduce most aryl bromides, so synthetically useful aryl radicals are often not directly available. Therefore, the development of a distinct leaving group more appropriately matched in redox potential could enable new reactivity manifolds for metallophotoredox catalysis, especially if arylcopper(iii) complexes are accessible, from which the most challenging bond-forming reactions can occur. Here we show the conceptual advantages of aryl thianthrenium salts for metallophotoredox catalysis, and their utility in site-selective late-stage aromatic fluorination.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
Crystallographic data for the structures reported in this article have been deposited at the Cambridge Crystallographic Data Centre (CCDC) under deposition numbers CCDC 1900278 (4), 1900279 (25-TT), 1900280 (40-TT), 1900276 (41-TT) and 1900277 (43-TT) (Supplementary Figs. 42–46). Copies of the data can be obtained free of charge via https://www.ccdc.cam.ac.uk/structures/. All other data that support the findings of this study are available within the article and its Supplementary Information, or from the corresponding author upon reasonable request.
References
Twilton, J. et al. The merger of transition metal and photocatalysis. Nat. Rev. Chem. 1, 0052 (2017).
Tellis, J. C. et al. Single-electron transmetalation via photoredox/nickel dual catalysis: unlocking a new paradigm for sp3–sp2 cross-coupling. Acc. Chem. Res. 49, 1429–1439 (2016).
Ghosh, I., Marzo, L., Das, A., Shaikh, R. & König, B. Visible light mediated photoredox catalytic arylation reactions. Acc. Chem. Res. 49, 1566–1577 (2016).
Skubi, K. L., Blum, T. R. & Yoon, T. P. Dual catalysis strategies in photochemical synthesis. Chem. Rev. 116, 10035–10074 (2016).
Zuo, Z. et al. Merging photoredox with nickel catalysis: coupling of α-carboxyl sp3-carbons with aryl halides. Science 345, 437–440 (2014).
Tellis, J. C., Primer, D. N. & Molander, G. A. Single-electron transmetalation in organoboron cross-coupling by photoredox/nickel dual catalysis. Science 345, 433–436 (2014).
Stache, E. E., Rovis, T. & Doyle, A. G. Dual nickel- and photoredox-catalyzed enantioselective desymmetrization of cyclic meso-anhydrides. Angew. Chem. Int. Ed. 56, 3679–3683 (2017).
Corcoran, E. B. et al. Aryl amination using ligand-free Ni(ii) salts and photoredox catalysis. Science 353, 279–283 (2016).
Terrett, J. A., Cuthbertson, J. D., Shurtleff, V. W. & MacMillan, D. W. C. Switching on elusive organometallic mechanisms with photoredox catalysis. Nature 524, 330–334 (2015).
Welin, E. R., Le, C., Arias-Rotondo, D. M., McCusker, J. K. & MacMillan, D. W. C. Photosensitized, energy transfer-mediated organometallic catalysis through electronically excited nickel(ii). Science 355, 380–385 (2017).
Lee, E., Hooker, J. M. & Ritter, T. Nickel-mediated oxidative fluorination for PET with aqueous [18F] fluoride. J. Am. Chem. Soc. 134, 17456–17458 (2012).
Ribas, X. & Casitas, A. in Ideas in Chemistry and Molecular Sciences: Where Chemistry Meets Life (ed. B. Pignataro) 31−57 (Wiley-VCH, 2010).
Casitas, A., Canta, M., Solà, M., Costas, M. & Ribas, X. Nucleophilic aryl fluorination and aryl halide exchange mediated by a Cui/Cuiii catalytic cycle. J. Am. Chem. Soc. 133, 19386–19392 (2011).
Le, C., Chen, T. Q., Liang, T., Zhang, P. & MacMillan, D. W. C. A radical approach to the copper oxidative addition problem: trifluoromethylation of bromoarenes. Science 360, 1010–1014 (2018).
Ghosh, I., Ghosh, T., Bardagi, J. & König, B. Reduction of aryl halides by consecutive visible light-induced electron transfer processes. Science 346, 725–728 (2014).
Ghosh, I. & König, B. Chromoselective photocatalysis: controlled bond activation through light-color regulation of redox potentials. Angew. Chem. Int. Ed. 55, 7676–7679 (2016).
Nguyen, J. D., D'Amato, E. M., Narayanam, J. M. R. & Stephenson, C. R. J. Engaging unactivated alkyl, alkenyl and aryl iodides in visible-light-mediated free radical reactions. Nat. Chem. 4, 854–859 (2012).
Costentin, C., Robert, M. & Savéant, J. M. Fragmentation of aryl halide π anion radicals. Bending of the cleaving bond and activation vs driving force relationships. J. Am. Chem. Soc 126, 16051–16057 (2004).
Yang, S., Chen, M. & Tang, P. Visible‐light photoredox‐catalyzed and copper‐promoted trifluoromethoxylation of arenediazonium tetrafluoroborates. Angew. Chem. Int. Ed. 58, 7840–7844 (2019).
Ichiishi, N., Canty, A. J., Yates, B. F. & Sanford, M. S. Cu-catalyzed fluorination of diaryliodonium salts with KF. Org. Lett. 15, 5134–5137 (2013).
Berger, F. et al. Site-selective and versatile aromatic C−H functionalization by thianthrenation. Nature 567, 223–228 (2019).
Ye, F. et al. Aryl sulfonium salts for site-selective late-stage trifluoromethylation. Angew. Chem. Int. Ed. 58, 14615–14619 (2019).
Engl, P. S. et al. C–N cross-couplings for site-selective late-stage diversification via aryl sulfonium salts. J. Am. Chem. Soc. 141, 13346–13351 (2019).
Lowry, M. S. et al. Single-layer electroluminescent devices and photoinduced hydrogen production from an ionic iridium(iii) complex. Chem. Mater. 17, 5712–5719 (2005).
Hammerich, O. & Parker, V. D. The reversible oxidation of aromatic cation radicals to dications. Solvents of low nucleophilicity. Electrochim. Acta 18, 537–541 (1973).
Rupp, H., Verplaetse, J. & Lontie, R. Binuclear copper electron paramagnetic resonance signals of α-methemocyanin of helix pomatia. Z. Naturforsch. 35 c, 188–192 (1980).
Andrieux, C. P., Savéant, J.-M., Tallec, A., Tardivel, R. & Tardy, C. Concerted and stepwise dissociative electron transfers. Oxidability of the leaving group and strength of the breaking bond as mechanism and reactivity governing factors illustrated by the electrochemical reduction of α-substituted acetophenones. J. Am. Chem. Soc. 119, 2420–2429 (1997).
Kampmeier, J. A. et al. Regioselectivity in the reductive bond cleavage of diarylalkylsulfonium salts: variation with driving force and structure of sulfuranyl radical intermediates. J. Am. Chem. Soc. 131, 10015–10022 (2009).
Laage, D., Burghardt, I., Sommerfeld, T. & Hynes, J. T. On the dissociation of aromatic radical anions in solution. 1. Formulation and application to p-cyanochlorobenzene radical anion. J. Phys. Chem. A 107, 11271–11291 (2003).
Burghardt, I., Laage, D. & Hynes, J. T. On the dissociation of aromatic radical anions in solution. 2. Reaction path and rate constant analysis. J. Phys. Chem. A 107, 11292–11306 (2003).
Laage, D., Burghardt, I., Sommerfeld, T. & Hynes, J. T. On the dissociation of aromatic radical anions in solution. ChemPhysChem 4, 61–66 (2003).
Fier, P. S., Luo, J. & Hartwig, J. F. Copper-mediated fluorination of arylboronate esters. Identification of a copper(iii) fluoride complex. J. Am. Chem. Soc. 135, 2552–2559 (2013).
Ye, Y. & Sanford, M. S. Mild copper-mediated fluorination of aryl stannanes and aryl trifluoroborates. J. Am. Chem. Soc. 135, 4648–4651 (2013).
Ye, Y., Schimler, S. D., Hanley, P. S. & Sanford, M. S. Cu(OTf)2-mediated fluorination of aryltrifluoroborates with potassium fluoride. J. Am. Chem. Soc. 135, 16292–16295 (2013).
Watson, D. A. et al. Formation of ArF from LPdAr(F): catalytic conversion of aryl triflates to aryl fluorides. Science 325, 1661–1664 (2009).
Yamamoto, K. et al. Palladium-catalysed electrophilic aromatic C–H fluorination. Nature 554, 511–514 (2018).
Acknowledgements
We thank C. Costentin (Université Paris VII) for helpful discussions on the electrochemical analysis. We thank O. Rüdiger (MPI CEC) for providing the electrochemical set-up and for helpful discussions, S. Marcus and D. Kampen (MPI KOFO) for mass spectrometry analysis and A. Dreier and J. Rust (MPI KOFO) for the crystal structure analysis.
Author information
Authors and Affiliations
Contributions
J.L., J.C. and R.S. developed the fluorination reaction. W.-S.H., M.B.P, J.L., J.C., F.B., S.C., A.S. and C.G. contributed to the mechanistic studies. F.B. initiated the approach to the project. All the authors wrote the manuscript. T.R. directed the project.
Corresponding author
Ethics declarations
Competing interests
A patent application (German patent number EP18204755.5), dealing with the use of TT and its derivatives for C–H functionalization has been filed, and F.B. and T.R. may benefit from royalty payments.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary information
Experimental procedures, product characterization, X-ray crystallographic analysis and mechanistic studies.
Crystallographic data
Crystallographic data for compound 25-TT. CCDC reference 1900279
Crystallographic data
Crystallographic data for compound 4. CCDC reference 1900278
Crystallographic data
Crystallographic data for compound 41-TT. CCDC reference 1900276
Crystallographic data
Crystallographic data for compound 43-TT. CCDC reference 1900277
Crystallographic data
Crystallographic data for compound 40-TT. CCDC reference 1900280
Rights and permissions
About this article
Cite this article
Li, J., Chen, J., Sang, R. et al. Photoredox catalysis with aryl sulfonium salts enables site-selective late-stage fluorination. Nat. Chem. 12, 56–62 (2020). https://doi.org/10.1038/s41557-019-0353-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-019-0353-3
This article is cited by
-
Unified metal-free intermolecular Heck-type sulfonylation, cyanation, amination, amidation of alkenes by thianthrenation
Nature Communications (2024)
-
Chemoselective umpolung of thiols to episulfoniums for cysteine bioconjugation
Nature Chemistry (2024)
-
Difluoromethyl phenoxathiinium salt: A new general and versatile difluoromethylating reagent with divergent ·CF2H, CF2H+, and:CF2 reactivities
Science China Chemistry (2024)
-
Photoinduced radical–ionic dihalogen transfer to carbon–carbon multiple bonds using oxime-based surrogates
Nature Synthesis (2023)
-
O-, N- and C-bicyclopentylation using thianthrenium reagents
Nature Synthesis (2023)