Abstract
Purpose
To describe the risk factors, clinical features, histopathology, treatment, and outcomes of patients with orbital tumour extension of ocular surface squamous neoplasia (OSSN).
Methods
Retrospective study of 51 patients with orbital tumour extension (cases) and 360 patients without orbital extension (controls).
Results
Of 1,653 patients with OSSN, orbital tumour extension was noted in 51 (3%) cases. The risk factors for orbital tumour extension included outdoor occupation (p < 0.03; Odds ratio (OR) = 1.96), Human Immunodeficiency Virus (HIV) infection (p < 0.0001; OR = 5.81), prolonged duration of symptoms (p = 0.01; OR = 1.02), tumour bilaterality (p = 0.02; OR = 2.92), forniceal and tarsal conjunctival involvement, diffuse tumour (p < 0.0001; OR = 9.13), inferior quadrantic location (p < 0.0001; OR = 7.51), increased tumour thickness (p = 0.04; OR = 1.59), higher % of ocular surface involvement (p = 0.002; OR = 1.12), nodular (p = 0.002; OR = 2.61) and nodulo-ulcerative (p < 0.0001; OR = 11.05) tumour morphology, poorly differentiated tumours (p = 0.006; OR = 4.23); invasive squamous cell carcinoma (SCC) (p < 0.0001; OR = 29.76), spindle cell and mucoepidermoid variant (p = 0.02; OR = 16.94) tumours. At a mean follow-up period of 27 months, tumour recurrence in the socket was noted in 1 (2%), locoregional lymph node metastasis (LNM) in 15 (29%) patients, and nine (18%) patients died due to systemic metastasis (SM). T4 tumour at presentation was a risk factor for LNM (p = 0.01; Hazard ratio (HR) = 5.60) and SM (p = 0.0003; HR = 5.09).
Conclusion
Orbital extension of OSSN is rare. Outdoor occupation, HIV infection, larger and thicker tumours in the inferior quadrant with forniceal and/or tarsal conjunctival involvement with nodular or noduloulcerative morphology, poor tumour differentiation, SCC, spindle cell and mucoepidermoid variants on histopathology are at increased risk for orbital tumour extension.
Similar content being viewed by others
Introduction
Ocular surface squamous neoplasia (OSSN) encompassing dysplasia, carcinoma in situ, and invasive squamous cell carcinoma is the most common malignant tumour of the ocular surface [1, 2]. In a review of 9,633 ocular and periocular tumours in Asian Indians, OSSN was the second most common tumour amongst all tumours and the most common malignant ocular surface tumour, representing 10% of all tumours and 30% of all conjunctival tumours at the referral centre [2]. In a review of 5,002 conjunctival tumours at a referral centre in the United States, OSSN was the most common ocular surface malignancy representing 14% of all conjunctival tumours [2].
Orbital extension of OSSN is uncommon in developed countries. Negligence or non-compliance to treatment can result in orbital tumour extension of OSSN. A review of 612 cases of OSSN from the United States revealed orbital tumour extension in only four (<1%) cases [3]. The incidence of orbital extension of OSSN is expected to be higher in developing and underdeveloped countries. In Africa, OSSN is the most common indication for orbital exenteration (57–76% of all orbital exenterations) in adults [4], suggesting a higher prevalence of orbital tumour extension. The incidence of orbital tumour extension of OSSN is 35% in Africa [5]. A histopathological review of 776 OSSN cases from India revealed orbital tumour extension in 26 (4%) cases [6]. Given the rarity of orbital extension of OSSN, there are only two studies that evaluated the prognosis in such cases [7, 8]. In this study, we aim to discuss the presenting features, treatment and prognosis of OSSN patients with orbital tumour extension and to evaluate risk factors for orbital tumour extension in OSSN.
Methods
The study was conducted at the Ocular Oncology Services, LV Prasad Eye Institute, Hyderabad and Vijayawada, India. The Institutional Ethics committee of LV Prasad Eye Institute approved the study. The study adhered to the tenets of the Declaration of Helsinki. A search was conducted in the clinical and histopathology database for the diagnosis of “ocular surface squamous neoplasia (OSSN)” for the period January 2004 to December 2019. All patients with documented evidence of orbital tumour extension of OSSN were included in the study group, and those with no evidence of orbital tumour extension were included as controls.
All patients underwent clinical examination and photographic documentation. The following demographic data and history were collected from the medical records for both cases and controls: age, sex, presenting complaints, duration of symptoms, systemic disease, prior history of intervention for the tumour, and duration between prior intervention and diagnosis of orbital tumour extension. Human Immunodeficiency Virus (HIV) screening was done in all these cases. The tumour details included laterality, epicentre, extent, size, pattern, and associated features (keratin, pigmentation, feeder vessels).
Computed tomography (CT) or magnetic resonance imaging (MRI) of the orbit was done to confirm the orbital tumour extent. Orbital imaging features were noted based on description in medical records and review of CT/MRI photographs. The treatment details (definitive and adjuvant treatments) were noted for cases. Histopathology details included tumour variant, differentiation, and extent. Tumour variant and differentiation were also compared between cases and controls. The outcome measures included tumour recurrence, locoregional lymph node metastasis, systemic metastasis and death. The time interval between the diagnosis of orbital tumour extension of OSSN and any of these outcome measures were noted. Comparison between those with and without metastasis was performed to identify risk factors for metastasis/death.
The statistical analysis was performed using STATA v14.2 (StataCorp, College Station, TX, USA). Descriptive statistics included mean, median and range for continuous data and proportion for categorical data. A mixed effects model, with random intercepts at the level of subjects, was used in the comparison between study and control groups. Evaluation of factors associated with orbital extension was performed by mixed effects logistic regression with the estimation of odds ratio and 95% confidence intervals (CIs). Kaplan–Meier analysis was performed to estimate the probabilities of metastasis and death, and Cox proportional hazards regression model was used to evaluate associated risk factors. A p value of <0.05 was considered statistically significant.
Results
Of 1653 patients with OSSN, orbital tumour extension was noted in 51 (3%) cases (Fig. 1). A total of randomly chosen 360 OSSN subjects with no orbital tumour extension were included as controls.
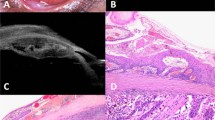
A A 51-year-old male with extensive ocular surface squamous neoplasia masking the right eye. B Worms eye view depicting proptosis in the right eye. C, D Computer tomography (CT) of the orbit demonstrating homogenous soft tissue lesion over the right ocular surface extending into the anterior orbit, without obvious intraocular invasion. E Photomicrograph showing scanner view of the ocular surface squamous neoplasia invading posterior orbital tissue (asterix) without any intraocular extension (Haematoxylin and eosin (H&E); scanner view). F Tumour arising from dysplastic conjunctiva (arrow) (H&E; x40). G Tumour cells with marked pleomorphism, intracytoplasmic keratinization and dysplastic squamous cells (H&E; x200). H Tumour cells seen invading orbital fibro-adipose tissue (asterix) and extraocular muscle (M) (H&E; x40).
The mean age at presentation of OSSN with orbital tumour extension was 50 years (median, 48 years; range, 6–88 years). History of prior intervention before the diagnosis of orbital tumour extension was noted in 19 (37%) patients. One patient had orbital tumour recurrence following extended enucleation for intraocular tumour extension, and one patient had undergone evisceration for a painful blind eye due to presumed undiagnosed OSSN. Of these 19 patients with history of prior intervention, 13 patients had undergone intervention(s) elsewhere prior to presentation to us, while the remaining six patients developed orbital tumour extension while undergoing treatment with us. The mean number of interventions was 2 (median, 1; range, 1–4). The mean interval between prior intervention and orbital tumour extension was 13 months (median, eight months; range, 1–60 months). Of the six patients who developed orbital tumour extension while undergoing treatment with us, four patients had microscopic tumour residue at the surgical margins and two patients had microscopic residual tumour at tumour base. Of the four patients with positive margins, two had received two cycles of topical mitomycin-C post-operatively, while two patients did not receive topical chemotherapy. The two patients with positive base were advised plaque radiotherapy but were non-complaint to treatment.
Predisposition for OSSN with a history of HIV infection or XP was noted in 22 (43%) patients. HIV infection was noted in 20 (39%) cases. Of these, 12 patients had a known history of HIV (mean duration since detection of HIV was 52 months (median, 36 months; range, 2–192 months)) and 8 were detected to be HIV positive on routine screening. Xeroderma pigmentosum was noted in two (4%) patients. There was no history of systemic cancer, evidence of immunosuppression, or other predisposing factors of OSSN in other patients.
Symptoms included mass (n = 39, 76%), watering (n = 9, 18%), pain (n = 5, 10%), decreased vision (n = 2, 4%), protrusion of eyeball (n = 2, 4%), and diplopia (n = 1, 2%). The mean duration of symptoms was 14 months (median, four months; range, <1–120 months). Orbital tumour extension of OSSN was unilateral in all cases though OSSN was bilateral in five (10%) cases. The tumour details are listed in Table 1 and compared with controls.
Orbital imaging was advised in all cases, but adequate details were available in only 42 (82%) cases. CT orbits was done in 40 cases and only two patients underwent MRI orbits. Proptosis was noted in 10 (24%) cases. Tumour extension was noted till the anterior (n = 19; 45%), mid (n = 21; 50%), or posterior (n = 2; 5%) orbit. The tumour involved inferior (n = 16; 38%), temporal (n = 11; 26%), superior (n = 3; 7%), nasal (n = 5; 12%), or diffuse (n = 7; 17%) orbital quadrants. Extraocular muscle involvement was noted in 32 (76%) cases, and inferior rectus (n = 18; 43%) was the most common muscle involved. Globe indentation was noted in 13 (31%) cases, while the globe was indistinguishable in two (5%) cases. Extraconal (n = 16; 38%) or intraconal (n = 5; 12%) fat involvement was noted. There was no evidence of intracranial extension in any case.
Orbital exenteration was performed in 44 (86%) cases while the remaining seven (14%) cases were non-compliant to treatment. Six (12%) patients underwent adjuvant external beam radiotherapy to the orbit. Most tumours were well differentiated (n = 21, 41%). Mucoepidermoid tumour variant was noted in three (6%) cases and spindle cell variant in one (2%) case. While orbital imaging indicated intraocular tumour extension in eight (19%) cases, histopathology confirmed intraocular tumour extension in 10 (23%) cases.
The risk factors for orbital tumour extension included outdoor occupation (p < 0.03; Odds ratio (OR) = 1.96), HIV infection (p < 0.0001; OR = 5.81), prolonged duration of symptoms (p = 0.01; OR = 1.02), tumour bilaterality (p = 0.02; OR = 2.92), forniceal and tarsal conjunctival involvement, diffuse tumour (p < 0.0001; OR = 9.13), inferior quadrantic location (p < 0.0001; OR = 7.51), increased tumour thickness (p = 0.04; OR = 1.59), higher % of ocular surface involvement (p = 0.002; OR = 1.12), nodular (p = 0.002; OR = 2.61) and nodulo-ulcerative (p < 0.0001; OR = 11.05) tumour morphology, poorly differentiated tumours (p = 0.006; OR = 4.23); invasive squamous cell carcinoma (SCC) (p < 0.0001; OR = 29.76), spindle cell and mucoepidermoid variant (p = 0.02; OR = 16.94) tumours. Forniceal and tarsal conjunctival tumour involvement and spindle cell variant of OSSN predicted “orbital tumour extension” perfectly. The factors suggestive of lower risk of orbital tumour extension included limbal tumour epicentre (p = 0.004; OR = 0.41), corneal tumour epicentre, temporal quadrantic location (p = 0.02; OR = 0.47), placoid tumour morphology, tumour pigmentation (p = 0.0001; OR = 0.23), well-differentiated tumours (p = 0.002; OR = 0.3), and tumours displaying conjunctival/corneal intraepithelial neoplasia. Corneal epicentre, placoid tumour morphology, and tumours with conjunctival/corneal intraepithelial neoplasia predicted “no orbital extension” perfectly (Table 2).
Tumour recurrence in the exenterated socket was noted in one case. He presented with noduloulcerative variant of OSSN, which was initially misdiagnosed as sclerokeratitis. Ultimately, diagnosis of OSSN was made 6 months later. Owing to intraocular tumour extension he was advised extended enucleation but the patient was lost to follow-up for 10 months when he presented with orbital tumour extension. Orbital exenteration followed by orbital external beam radiotherapy (EBRT) was performed. Histopathology revealed poorly differentiated mucoepidermoid variant of squamous cell carcinoma with lymphovascular tumour invasion. Ten months later, patient developed massive tumour recurrence in the socket with ipsilateral parotid gland metastasis. Patient eventually died seven months later due to intracranial tumour extension. Patients with orbital tumour extension had significantly higher rates of regional lymph node metastasis (29% vs <1%; p < 0.0001) and metastasis-related mortality (18% vs <1%; p < 0.0001) compared to controls over a mean follow-up period of 27 months (median, six months; range, 1–237 months) in cases vs 11 months (median, five months; range, 1 to 108 months) in controls. The mean time interval between orbital extension of OSSN and systemic metastasis/death was 22 months (median, 20 months; range; 3–48 months). The one, two, and four-years Kaplan–Meier estimate of regional lymph node metastasis and systemic metastasis/death in cases were 37% and 3%, 37% and 37%, and 64% and 62% respectively (Table 3 and Fig. 2). Older age (p = 0.049; Hazard ratio (HR) = 1.03) and T4 tumour at presentation (p = 0.01; HR = 5.60) were identified as the risk factors for regional lymph node metastasis, while T4 tumour at presentation (p = 0.0003; HR = 5.09) was the sole risk factor for systemic metastasis/death.
Discussion
Orbital extension of OSSN is rare. It mainly occurs due to negligence towards an early consult, incomplete treatment, or non-compliance to treatment. Appropriate surgical and/or medical treatment, achieving clear margins and base following surgical excision, appropriate adjuvant treatment, and regular follow-up after treatment of OSSN are pre-requisites for good outcomes. According to our study, the risk factors for orbital tumour extension included outdoor occupation, HIV infection, prolonged duration of symptoms, tumour bilaterality, forniceal and tarsal conjunctival involvement, diffuse tumour, inferior quadrantic location, increased tumour thickness, higher % of ocular surface involvement, nodular and nodulo-ulcerative tumour morphology, poorly differentiated tumours, invasive SCC, spindle cell and mucoepidermoid variant tumours. Forniceal and tarsal conjunctival tumour involvement and spindle cell variant of OSSN predicted “orbital tumour extension” perfectly. The factors suggestive of lower risk of orbital tumour extension included limbal tumour epicentre, corneal tumour epicentre, temporal quadrantic location, placoid tumour morphology, tumour pigmentation, well-differentiated tumours, and tumours displaying conjunctival/corneal intraepithelial neoplasia. Corneal epicentre, placoid tumour morphology, and tumours with conjunctival/corneal intraepithelial neoplasia predicted “no orbital extension” perfectly.
Recurrence following OSSN treatment can occur in 0–56% cases and usually occurs within the first two years [9]. The predictors for tumour recurrence after surgical excision include positive surgical margins and higher-grade tumours [10]. In our study, there was a history of prior intervention in 19 (37%) patients and the mean interval between prior intervention and tumour recurrence in the form of orbital tumour extension was 19 months. While details of positive surgical margins and grade of tumour were not known in 13 cases, six cases who underwent prior intervention with us revealed positive surgical margins/base and an invasive squamous cell carcinoma in all six cases. The use of adjuvant topical chemotherapy/immunotherapy for positive surgical margins and use of adjuvant plaque radiotherapy for positive surgical base can minimize the evidence of tumour recurrence [10, 11]. In our study, two patients developed tumour recurrence with extension of tumour to the orbit despite the use of adjuvant topical chemotherapy. However, these patients had a poor follow-up and presented only when the tumour extended to the orbit. These findings suggest that the patients with positive surgical margins require a close follow-up despite the use of topical chemotherapy/immunotherapy.
About 38% of OSSN patients can be associated with HIV infection [12, 13]. These cases are usually associated with aggressive tumours with a higher incidence of larger and thicker tumours, corneal, scleral, and orbital tumour extension [14]. The risk of orbital tumour extension in HIV positive cases is 9–13% compared to 3% in those without HIV [12,13,14]. In this study, 39% cases had HIV infection against 18% controls. Of these 20 HIV infected cases, eight were previously unaware of the HIV status and was detected only on routine screening. These findings suggest that HIV screening should be mandatory in all cases presenting with orbital extension of OSSN.
Orbital tumour extension can occur more commonly in aggressive histopathological variants of OSSN. A review of 21 cases of mucoepidermoid variant of OSSN revealed orbital tumour extension in six (29%) cases [15]. In our study, mucoepidermoid variant of OSSN was noted in three (6%) cases. Of these, one case had socket recurrence despite orbital exenteration and adjuvant EBRT indicating aggressive nature of the lesion. Spindle cell variant of OSSN is notorious for multiple tumour recurrences and intraocular tumour extension in 50% to 67% cases [16, 17]. However, orbital tumour extension of spindle cell variant of OSSN is uncommon. In this study, there was one case of spindle cell variant of OSSN who presented with orbital tumour extension without evidence of intraocular tumour extension. Based on our study, invasive SCC, spindle cell and mucoepidermoid variant tumours have higher risk of orbital tumour invasion.
Intraocular tumour extension of OSSN is rare and occurs in 1–15% cases of OSSN[17]. In a review of 23 OSSN cases with intraocular tumour extension, additional orbital tumour extension was noted in five (24%) cases [17] indicating a higher chance of co-existence of intraocular and orbital tumour extension. Orbital tumour recurrences can also occur after extended enucleation for intraocular tumour extension of OSSN [17]. In our study, additional intraocular tumour extension was noted in 10 (23%) cases on histopathology. One patient had orbital tumour recurrence following extended enucleation for intraocular tumour extension. Whenever intraocular tumour extension is noted in OSSN cases, careful examination should be done to rule out additional orbital extension at the same visit or subsequent visits during follow-up.
The preferred choice of treatment for patients with orbital tumour extension of OSSN is orbital exenteration [7, 18]. Neoadjuvant systemic chemotherapy (Paclitaxel + Carboplatin or 5-Fluorouracil + Carboplatin) is also described for these cases to facilitate eyelid-sparing orbital exenteration in patients with extensive lesion [19] or to avoid orbital exenteration in patients with potential vision [20]. In a study of three patients with orbital tumour extension of OSSN, one to four cycles of systemic chemotherapy achieved complete tumour regression and could avoid orbital exenteration in two cases [20]. In our study, all cases with orbital tumour extension of OSSN were advised orbital exenteration, of which 44 (86%) cases underwent the surgery. Adjuvant EBRT was also performed in cases with positive margins. Despite this aggressive approach, at least one patient with mucoepidermoid variant of OSSN developed tumour recurrence in the socket.
The risk of metastasis in OSSN is low with lower chances of mortality. The prevalence of locoregional metastasis in OSSN is 0.5–18%, and distant metastasis is 0–6% [8, 21,22,23,24]. In our study, metastasis and metastasis-related death occurred in <1% controls. In a study of 28 cases with secondary orbital extension of OSSN, six (20%) cases developed regional lymph node metastasis and seven (23%) died due to metastasis [7]. In another study of 24 cases with orbital invasion or invasion of adjacent structures, metastasis/death occurred in 7 (29%) cases [8]. In our study of 51 cases, regional lymph node metastasis was seen in 15 (29%) cases and metastasis related death in nine (18%) cases, suggesting poor prognosis in these cases. The risk factors for regional lymph node metastasis included older age and T4 tumour at presentation. The role of sentinel lymph node biopsy for early detection of regional lymph node metastasis needs to be explored. T4 tumour at presentation was a risk factor for systemic metastasis and death. Periodic systemic screening is important for early detection of systemic metastasis. The role of adjuvant chemotherapy in improving prognosis in these cases needs to be explored.
In conclusion, orbital tumour extension of OSSN can occur in 3% cases. Orbital tumour extension of OSSN should be suspected in patients with proptosis, limited extraocular movements, globe dystopia, or in eyes with indistinct posterior limit of the tumour. The risk factors for orbital tumour extension include outdoor occupation, HIV infection, prolonged duration of symptoms, tumour bilaterality, forniceal and tarsal conjunctival involvement, diffuse tumour, inferior quadrantic location, increased tumour thickness, higher % of ocular surface involvement, nodular and nodulo-ulcerative tumour morphology, poorly differentiated tumours, and aggressive histopathological variants of OSSN. Orbital imaging is useful in detection of orbital tumour extension. Based on current evidence, orbital exenteration is the preferred modality of treatment. Periodic lymph node examination and periodic systemic screening are mandatory in these cases. Orbital tumour extension of OSSN is associated with poor prognosis with higher chances of metastasis and death.
Summary
What was known before
-
Orbital tumour extension of ocular surface squamous neoplasia (OSSN) is rare.
-
Metastastic rate of OSSN is low.
What this study adds
-
Orbital tumour extension of ocular surface squamous neoplasia is associated with poor prognosis.
-
The metastastic rate in patients with orbital extension of OSSN is high at 29%.
References
Shields CL, Chien JL, Surakiatchanukul T, Sioufi K, Lally SE, Shields JA. Conjunctival tumors: review of clinical features, risks, biomarkers, and outcomes–The 2017 J. Donald M. Gass Lecture. Asia Pac. J Ophthalmol (Philos). 2017;6:109–20.
Kaliki S, Das AV. Ocular and periocular tumors in india: an eyesmart electronic medical record analysis of 9633 cases from a referral center. Middle East Afr J Ophthalmol. 2021;27:199–203.
Kao AA, Galor A, Karp CL, Abdelaziz A, Feuer WJ, Dubovy SR. Clinicopathologic correlation of ocular surface squamous neoplasms at Bascom Palmer Eye Institute: 2001 to 2010. Ophthalmology. 2012;119:1773–6.
Gichuhi S, Sagoo MS, Weiss HA, Burton MJ. Epidemiology of ocular surface squamous neoplasia in Africa. Trop Med Int Health. 2013;18:1424–43.
Fasina O. Ocular surface squamous neoplasia at a tertiary eye facility, Southwestern Nigeria: a 10-year review. Int Ophthalmol. 2021;41:3325–31.
Mishra DK, Taneja S, Singh V, Veena U, Kaliki S. Histopathological spectrum of ocular surface squamous neoplasia: A retrospective study of 776 lesions. Indian J Pathol Microbiol. 2022;65:3–7.
Johnson TE, Tabbara KF, Weatherhead RG, Kersten RC, Rice C, Nasr AM. Secondary squamous cell carcinoma of the orbit. Arch Ophthalmol. 1997;115:75–8.
Chauhan S, Sen S, Sharma A, Sharma A, Tandon R, Kashyap S, et al. American Joint Committee on Cancer Staging and clinicopathological high-risk predictors of ocular surface squamous neoplasia: a study from a tertiary eye center in India. Arch Pathol Lab Med. 2014;138:1488–94.
Hӧllhumer R, Williams S, Michelow P. Ocular surface squamous neoplasia: management and outcomes. Eye (Lond). 2021;35:1562–73.
Galor A, Karp CL, Oellers P, Kao AA, Abdelaziz A, Feuer W, et al. Predictors of ocular surface squamous neoplasia recurrence after excisional surgery. Ophthalmology. 2012;119:1974–81.
Arepalli S, Kaliki S, Shields CL, Emrich J, Komarnicky L, Shields JA. Plaque radiotherapy in the management of scleral-invasive conjunctival squamous cell carcinoma: an analysis of 15 eyes. JAMA Ophthalmol. 2014;132:691–6.
Kaliki S, Kamal S, Fatima S. Ocular surface squamous neoplasia as the initial presenting sign of human immunodeficiency virus infection in 60 Asian Indian patients. Int Ophthalmol. 2017;37:1221–8.
Rathi SG, Ganguly Kapoor A, Kaliki S Ocular surface squamous neoplasia in HIV-infected patients: current perspectives. HIV AIDS (Auckl) 201814;10:33-45.
Kamal S, Kaliki S, Mishra DK, Batra J, Naik MN. Ocular surface squamous neoplasia in 200 patients: a case-control study of immunosuppression resulting from Human Immunodeficiency Virus versus immunocompetency. Ophthalmology. 2015;122:1688–94.
Robinson JW, Brownstein S, Jordan DR, Hodge WG. Conjunctival mucoepidermoid carcinoma in a patient with ocular cicatricial pemphigoid and a review of the literature. Surv Ophthalmol. 2006;51:513–9.
Huntington AC, Langloss JM, Hidayat AA. Spindle cell carcinoma of the conjunctiva. An immunohistochemical and ultrastructural study of six cases. Ophthalmology. 1990;97:711–7.
Kaliki S, Jajapuram SD, Maniar A, Taneja S, Mishra DK. Ocular surface squamous neoplasia with intraocular tumour extension: a study of 23 patients. Eye (Lond). 2020;34:319–26.
Gökmen Soysal H, Ardiç F. Malignant conjunctival tumors invading the orbit. Ophthalmologica. 2008;222:338–43.
Nair AG, Kaliki S, Mishra DK, Reddy VA, Naik MN. Neoadjuvant chemotherapy for invasive squamous cell carcinoma of the conjunctiva: a case report. Indian J Ophthalmol. 2015;63:927–9.
Meel R, Dhiman R, Kashyap S, Agrawal S, Pushker N, Bakhshi S. Systemic neoadjuvant chemotherapy in non-resectable invasive squamous cell carcinoma of ocular surface. Eur J Ophthalmol. 2021:11206721211014376. Epub ahead of print.
El-Hadad C, Rubin ML, Nagarajan P, Ford JR, Xu S Jr, Ning J, et al. Prognostic factors for orbital exenteration, local recurrence, metastasis, and death from disease in conjunctival squamous cell carcinoma. Ophthalmic Plast Reconstr Surg. 2021;37:262–68.
Tabbara KF, Kersten R, Daouk N, Blodi FC. Metastatic squamous cell carcinoma of the conjunctiva. Ophthalmology. 1998;95:318–21.
Abt NB, Zhao J, Huang Y, Eghrari AO. Prognostic factors and survival for malignant conjunctival melanoma and squamous cell carcinoma over four decades. Am J Otolaryngol. 2019;40:577–82.
Singh S, Mohamed A, Kaliki S. Ocular surface squamous neoplasia: analysis based on the 8th American Joint Committee on Cancer classification. Int Ophthalmol. 2019;39:1283–91.
Funding
Support provided by The Operation Eyesight Universal Institute for Eye Cancer (SK) and Hyderabad Eye Research Foundation (SK), Hyderabad, India. The funders had no role in the preparation, review or approval of the manuscript.
Author information
Authors and Affiliations
Contributions
SK was responsible for conception of the study, data analysis, and drafting the manuscript. SK is accountable for all aspects of the work; RDW, VSV, AGK, DKM, and SJ were responsible for data acquisition and revision of the manuscript; AM was responsible for statistical analysis, data interpretation and revision of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kaliki, S., Wagh, R.D., Vempuluru, V.S. et al. Ocular surface squamous neoplasia with orbital tumour extension: risk factors and outcomes. Eye 37, 446–452 (2023). https://doi.org/10.1038/s41433-022-01955-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41433-022-01955-1