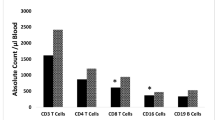
Abstract
Background
T-Cell Receptor Excision Circles based newborn screening (TREC-NBS) allows for early detection of T-cell lymphopenia in infants with primary immunodeficiency disorders (PIDD). The utility of abnormal TREC-NBS in infants without PIDD is not well studied. We sought to evaluate the association of abnormal TREC-NBS with mortality.
Methods
365,207 TREC-NBS from October 2011 to December 2014 were reviewed. 467 newborns had abnormal screens and did not meet the criteria for a PIDD diagnosis. Cases were matched to controls (1:3) based on gestational age, birth weight, neonatal intensive care unit status (NICU), and race. Data were obtained through NBS, birth and death certificates records from the Michigan Department of Health and Human Services (MDHHS) databases.
Results
Infants with abnormal TREC-NBS had higher mortality even when PIDD was ruled-out. Transient abnormal TREC-NBS was not associated with higher mortality, but unresolved or late abnormal TREC-NBS was associated with higher mortality. Infants with late abnormal TREC-NBS had severe prematurity, lower birth weight, lower Apgar scores, and higher percentage of congenital anomalies.
Conclusion
Infants with abnormal TREC-NBS may be at a higher risk of morbidity and mortality and should be carefully followed, especially if discharged home before a repeat screen can be completed.
Impact
-
This study explores the risk factors and mortality for newborns with secondary T-cell lymphopenia captured on T-Cell Receptor Excision Circles based newborn screening (TREC-NBS).
-
Abnormal TREC-NBS allows for prompt life-saving interventions for primary immunological conditions such as Severe Combined Immunodeficiency (SCID), but can also be associated with non-immunologic conditions.
-
Unresolved and late abnormal TREC-NBS is associated with higher mortality even without primary immunodeficiency, likely detected in infants with more severe prematurity, lower birth weight, and congenital anomalies.
-
TREC-NBS positive infants with secondary T-cell lymphopenia require special attention and close monitoring.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 14 print issues and online access
$259.00 per year
only $18.50 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Data availability
The data that support the findings of this study are available from Michigan Department of Health and Human Services but restrictions apply to the availability of these data, which were used under license for the current study, and so are not publicly available. Data are however available from the authors upon reasonable request and with permission of Michigan Department of Health and Human Services.
References
Chan, K. & Puck, J. M. Development of population-based newborn screening for severe combined immunodeficiency. J. Allergy Clin. Immunol. 115, 391–398 (2005).
Kelly, B. T., Tam, J. S., Verbsky, J. W. & Routes, J. M. Screening for severe combined immunodeficiency in neonates. Clin. Epidemiol. 5, 363–369 (2013).
Van der Spek, J., Groenwold, R. H. H., Van der Burg, M. & Van Montfrans, J. M. TREC Based Newborn Screening for Severe Combined Immunodeficiency Disease: A Systematic Review. J. Clin. Immunol. 35, 416–430 (2015).
Puck, J. M. Neonatal screening for severe combined immunodeficiency. Curr. Opin. Pediatr. 23, 667–673 (2011).
Puck, J. M. Laboratory Technology for Population-based Screening for SCID in Neonates: The Winner Is T-cell Receptor Excision Circles (TRECs). J. Allergy Clin. Immunol. 129, 607–616 (2013).
Amatuni, G. S. et al. Newborn Screening for Severe Combined Immunodeficiency and T-cell Lymphopenia in California, 2010–2017. Pediatrics 143, e20182300 (2019).
Nevid, M. et al. Non-immunologic Conditions Associated with Low TREC Values. J. Allergy Clin. Immunol. 145, AB214 (2020).
Puck, J. M. Newborn screening for severe combined immunodeficiency and T-cell lymphopenia. Immunol. Rev. 287, 241–252 (2019).
Accetta, D. J., Brokopp, C. D., Baker, M. W., Verbsky, J. & Routes, J. M. Cause of death in neonates with inconclusive or abnormal T-cell receptor excision circle assays on newborn screening. J. Clin. Immunol. 31, 962–967 (2011).
Koenig, J. M. & Christensen, R. D. The mechanism responsible for diminished neutrophil production in neonates delivered of women with pregnancy-induced hypertension. Am. J. Obstet. Gynecol. 165, 467–473 (1991).
Backes, C. H. et al. Maternal preeclampsia and neonatal outcomes. J. Pregnancy 2011, 214365 (2011).
Duijts, L. et al. Perinatal stress influences lymphocyte subset counts in neonates. The generation R study. Pediatr. Res. 63, 292–298 (2008).
Irvine, B., Dzakpasu, S. & León, J. A. Perinatal health indicators 2013: a surveillance report by the Public Health Agency of Canada’s Perinatal Surveillance System. Health Promot Chronic Dis. Prev. Can. 35, 23–24 (2015).
Ray, J. G., Park, A. L. & Fell, D. B. Mortality in Infants Affected by Preterm Birth and Severe Small-for-Gestational Age Birth Weight. Pediatrics 140, e20171881 (2017).
Sattolo, M. L. et al. Association of Birth Defects With Child Mortality Before Age 14 Years. JAMA Netw. Open 5, e226739 (2022).
Currier, R. & Puck, J. M. SCID newborn screening: What we’ve learned. J. Allergy Clin. Immunol. 147, 417–426 (2021).
Warny, M., Helby, J., Nordestgaard, B. G., Birgens, H. & Bojesen, S. E. Incidental lymphopenia and mortality: a prospective cohort study. CMAJ 192, E25–E33 (2020).
Wolf, J., Dahlenburg, K. & Borte, S. Immune Cellular Evaluation Following Newborn Screening For Severe T and B Cell Lymphopenia. EJIFCC 30, 396–406 (2019).
Manuck, T. A. Racial and ethnic differences in preterm birth: A complex, multifactorial problem. Semin Perinatol. 41, 511–518 (2017).
Johnson, J. D., Green, C. A., Vladutiu, C. J. & Manuck, T. A. Racial Disparities in Prematurity Persist among Women of High Socioeconomic Status. Am. J. Obstet. Gynecol. MFM 2, 100104 (2020).
Acknowledgements
We would like to thank the Michigan Primary Immunodeficiency Disorders Quality Improvement Committee (PIDQIC) members, especially Dr. Ulrich Duffner (Michigan State University/Helen DeVos Children’s Hospital), Dr. Sureyya Savasan (Children’s Hospital of Michigan/Central Michigan University College of Medicine), Dr. Nicholas Hartog (Helen DeVos Children’s), Mary Fredrickson (Children’s Hospital of Michigan) and Mary Kleyn (Michigan Department of Community Health) for their support of this project, insightful comments and review of the manuscript.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Jenny Huang, Ashwin Shankar, Isabel Hurden, Ronald Thomas and Pavadee Poowuttikul. The first draft of the manuscript was written by Jenny Huang and Ashwin Shankar, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
Drs. Pavadee Poowuttikul and Jenny Huang have research/grant support from Respiri for a project titled “The Use of Electronic Monitoring Device for Wheezing (Wheezo) and Remote Monitoring Program (RMP) to Improve Asthma Outcome in Inner-City Children” The other authors have indicated they have no potential conflicts of interest to disclose.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Huang, J., Shankar, A., Hurden, I. et al. Increased mortality in infants with abnormal T-cell receptor excision circles. Pediatr Res (2024). https://doi.org/10.1038/s41390-024-03121-7
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41390-024-03121-7