Abstract
Background
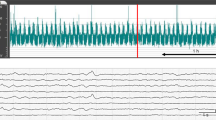
We previously reported that hydrogen (H2) gas combined with therapeutic hypothermia (TH) improved short-term neurological outcomes in asphyxiated piglets. However, the effect on seizure burden was unclear. Using amplitude-integrated electroencephalography (aEEG), we compared TH + H2 with TH alone in piglets 24 h after hypoxic–ischemic (HI) insult.
Methods
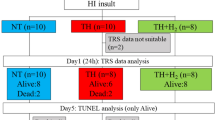
After a 40-min insult and resuscitation, 36 piglets ≤24 h old were divided into three groups: normothermia (NT, n = 14), TH alone (33.5 ± 0.5 °C, 24 h, n = 13), and TH + H2 (2.1–2.7% H2 gas, 24 h, n = 9). aEEG was recorded for 24 h post-insult and its background pattern, status epilepticus (SE; recurrent seizures lasting >5 min), and seizure occurrence (Sz; occurring at least once but not fitting the definition of SE) were evaluated. Background findings with a continuous low voltage and burst suppression were considered abnormal.
Results
The percentage of piglets with an abnormal aEEG background (aEEG-BG), abnormal aEEG-BG+Sz and SE was lower with TH + H2 than with TH at 24 h after HI insult. The duration of SE was shorter with TH + H2 and significantly shorter than with NT.
Conclusions
H2 gas combined with TH ameliorated seizure burden 24 h after HI insult.
Impact
-
In this asphyxiated piglet model, there was a high percentage of animals with an abnormal amplitude-integrated electroencephalography background (aEEG-BG) after hypoxic-ischemic (HI) insult, which may correspond to moderate and severe hypoxic-ischemic encephalopathy (HIE).
-
Therapeutic hypothermia (TH) was associated with a low percentage of piglets with EEG abnormalities up to 6 h after HI insult but this percentage increased greatly after 12 h, and TH was not effective in attenuating seizure development.
-
H2 gas combined with TH was associated with a low percentage of piglets with an abnormal aEEG-BG and with a shorter duration of status epilepticus at 24 h after HI insult.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 14 print issues and online access
$259.00 per year
only $18.50 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during this study are available from the corresponding author on reasonable request.
References
Gluckman, P. D. et al. Selective head cooling with mild systemic hypothermia after neonatal encephalopathy: multicentre randomised trial. Lancet 365, 663–670 (2005).
Shankaran, S. et al. Childhood outcomes after hypothermia for neonatal encephalopathy. N. Engl. J. Med. 366, 2085–2092 (2012).
Jacobs, S. E. et al. Cooling for newborns with hypoxic ischaemic encephalopathy. Cochrane Database Syst. Rev. 2013, CD003311 (2013).
Azzopardi, D. et al. Implementation and conduct of therapeutic hypothermia for perinatal asphyxial encephalopathy in the UK-analysis of national data. PLoS ONE 7, e38504 (2012).
Glass, H. C. et al. Risk factors for EEG seizures in neonates treated with hypothermia: a multicenter cohort study. Neurology 82, 1239–1244 (2014).
Boylan, G. B., Kharoshankaya, L. & Wusthoff, C. J. Seizures and hypothermia: importance of electroencephalographic monitoring and considerations for treatment. Semin. Fetal Neonatal Med. 20, 103–108 (2015).
Zhou, K. Q. et al. Treating seizures after hypoxic-ischemic encephalopathy-current controversies and future directions. Int. J. Mol. Sci. 22, 7121 (2021).
Lin, Y. K., Hwang-Bo, S., Seo, Y. M. & Youn, Y. A. Clinical seizures and unfavorable brain MRI patterns in neonates with hypoxic ischemic encephalopathy. Medicine 100, e25118 (2021).
Basti, C. et al. Seizure burden and neurodevelopmental outcome in newborns with hypoxic-ischemic encephalopathy treated with therapeutic hypothermia: a single center observational study. Seizure 83, 154–159 (2020).
Glass, H. C. et al. Contemporary profile of seizures in neonates: a prospective cohort study. J. Pediatr. 174, 98–103.e101 (2016).
Ohsawa, I. et al. Hydrogen acts as a therapeutic antioxidant by selectively reducing cytotoxic oxygen radicals. Nat. Med. 13, 688–694 (2007).
Domoki, F. et al. Hydrogen is neuroprotective and preserves cerebrovascular reactivity in asphyxiated newborn pigs. Pediatr. Res. 68, 387–392 (2010).
Htun, Y. et al. Hydrogen ventilation combined with mild hypothermia improves short-term neurological outcomes in a 5-day neonatal hypoxia-ischaemia piglet model. Sci. Rep. 9, 4088 (2019).
Nakamura, S. et al. Impact of hydrogen gas inhalation during therapeutic hypothermia on cerebral hemodynamics and oxygenation in the asphyxiated piglet. Sci. Rep. 13, 1615 (2023).
Brophy, G. M. et al. Guidelines for the evaluation and management of status epilepticus. Neurocrit. Care 17, 3–23 (2012).
Molloy, E. J. et al. Neuroprotective therapies in the NICU in term infants: present and future. Pediatr. Res. 93, 1819–1827 (2023).
Kharoshankaya, L. et al. Seizure burden and neurodevelopmental outcome in neonates with hypoxic–ischemic encephalopathy. Dev. Med. Child Neurol. 58, 1242–1248 (2016).
Nemeth, J. et al. Molecular hydrogen affords neuroprotection in a translational piglet model of hypoxic-ischemic encephalopathy. J. Physiol. Pharmacol. 67, 677–689 (2016).
El-Dib, M. & Soul, J. S. The use of phenobarbital and other anti-seizure drugs in newborns. Semin. Fetal Neonatal Med. 22, 321–327 (2017).
Bittigau, P. et al. Antiepileptic drugs and apoptotic neurodegeneration in the developing brain. Proc. Natl Acad. Sci. USA 99, 15089–15094 (2002).
Forcelli, P. A., Janssen, M. J., Vicini, S. & Gale, K. Neonatal exposure to antiepileptic drugs disrupts striatal synaptic development. Ann. Neurol. 72, 363–372 (2012).
Htun, Y., Nakamura, S. & Kusaka, T. Hydrogen and therapeutic gases for neonatal hypoxic-ischemic encephalopathy: potential neuroprotective adjuncts in translational research. Pediatr. Res. 89, 753–759 (2020).
Fellman, V. & Raivio, K. O. Reperfusion injury as the mechanism of brain damage after perinatal asphyxia. Pediatr. Res. 41, 599–606 (1997).
Nagatani, K. et al. Effect of hydrogen gas on the survival rate of mice following global cerebral ischemia. Shock 37, 645–652 (2012).
Miller, S. P. et al. Seizure-associated brain injury in term newborns with perinatal asphyxia. Neurology 58, 542–548 (2002).
Nakamura, S. et al. Cerebral blood volume combined with amplitude-integrated EEG can be a suitable guide to control hypoxic/ischemic insult in a piglet model. Brain Dev. 35, 614–625 (2013).
Nakamura, M. et al. Cerebral blood volume measurement using near-infrared time-resolved spectroscopy and histopathological evaluation after hypoxic-ischemic insult in newborn piglets. Int. J. Dev. Neurosci. 42, 1–9 (2015).
Thoresen, M. et al. A piglet survival model of posthypoxic encephalopathy. Pediatr. Res. 40, 738–748 (1996).
Haaland, K., Loberg, E. M., Steen, P. A. & Thoresen, M. Posthypoxic hypothermia in newborn piglets. Pediatr. Res. 41, 505–512 (1997).
Bjorkman, S. T. et al. Hypoxic/ischemic models in newborn piglet: comparison of constant FiO2 versus variable FiO2 delivery. Brain Res. 1100, 110–117 (2006).
Htun, Y. et al. Conflicting findings on the effectiveness of hydrogen therapy for ameliorating vascular leakage in a 5-day post hypoxic-ischemic survival piglet model. Sci. Rep. 13, 10486 (2023).
Acknowledgements
We thank medical students at the Faculty of Medicine, Kagawa University, Kagawa, Japan for their assistance in this study.
Funding
This study was financially supported by JSPS KAKENHI grants (19K08253 (S.N.), 19K08349 (K.K.), 22K15923 (Y.N.), and 22K07822 (T.K.), 22K15899 (T.W.), 22K15922 (A.M.), and 23K07332 (S.N.)), and Kagawa University Faculty of Medicine School of Medicine Alumni Association Sanjukai Research Aid R1-1 (S.N.).
Author information
Authors and Affiliations
Contributions
T.T., S.N. and T.K. were involved in the initial study design and wrote the main text. K.K., Y.N., T.K. and S.N. obtained the necessary financial support for this project and provided study materials. T.T. and Y.S. were primarily responsible for evaluating the aEEG findings. Y.N., T. Mitsuie, E.I., K.I., M.A., K.K. and N.F. carried out the animal experiments and recorded blood gas and physiological data. K.M., S.K., K.O., M.U. and T. Miki. contributed to data analysis and performed the statistical analysis. All members drafted the article and critically revised it.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval and informed consent
The study protocol was approved by the Kagawa University Animal Care and Use Committee (15070–1) and was conducted in accordance with Animal Research: Reporting In Vivo Experiments (ARRIVE) guidelines and all other applicable guidelines and regulations.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tsuchiya, T., Nakamura, S., Sugiyama, Y. et al. Hydrogen gas can ameliorate seizure burden during therapeutic hypothermia in asphyxiated newborn piglets. Pediatr Res (2024). https://doi.org/10.1038/s41390-024-03041-6
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41390-024-03041-6