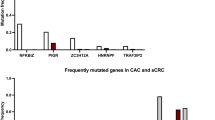
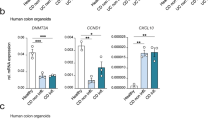
Abstract
Epigenetic fields for cancerization are involved in development of human cancers, especially those associated with inflammation and multiple occurrences. However, it is still unclear when such field defects are formed and what component of inflammation is involved in induction of aberrant DNA methylation. Here, in a mouse colitis model induced by dextran sulfate sodium (DSS), we identified three CpG islands specifically methylated in colonic epithelial cells exposed to colitis. Their methylation levels started to increase as early as 8 weeks after DSS treatment and continued to increase until colon cancers developed at 15 weeks. In contrast to the temporal profile of DNA methylation levels, infiltration of inflammatory cells spiked immediately after the DSS treatment and then gradually decreased. Exposure of cultured colonic epithelial cells to DSS did not induce DNA methylation and it was indicated that inflammation triggered by the DSS treatment was responsible for methylation induction. To clarify components of inflammation involved, severe combined immunodeficiency (SCID) mice that lack functional T- and B-cells were similarly treated. Even in SCID mice, DNA methylation, along with colon tumors, were induced at the same levels as in their background strain of mice (C.B17). Comparative analysis of inflammation-related genes showed that Ifng, Il1b and Nos2 had expression concordant with methylation induction whereas Il2, Il6, Il10, Tnf did not. These results showed that an epigenetic field defect is formed at early stages of colitis-associated carcinogenesis and that functional T and B cells are non-essential for the formation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Araki Y, Sugihara H, Hattori T . (2006). In vitro effects of dextran sulfate sodium on a Caco-2 cell line and plausible mechanisms for dextran sulfate sodium-induced colitis. Oncol Rep 16: 1357–1362.
Autschbach F, Giese T, Gassler N, Sido B, Heuschen G, Heuschen U et al. (2002). Cytokine/chemokine messenger-RNA expression profiles in ulcerative colitis and Crohn's disease. Virchows Arch 441: 500–513.
Bosma GC, Custer RP, Bosma MJ . (1983). A severe combined immunodeficiency mutation in the mouse. Nature 301: 527–530.
Braakhuis BJ, Tabor MP, Kummer JA, Leemans CR, Brakenhoff RH . (2003). A genetic explanation of Slaughter's concept of field cancerization: evidence and clinical implications. Cancer Res 63: 1727–1730.
Cappello M, Keshav S, Prince C, Jewell DP, Gordon S . (1992). Detection of mRNAs for macrophage products in inflammatory bowel disease by in situ hybridisation. Gut 33: 1214–1219.
Cheng H, Bjerknes M, Amar J . (1984). Methods for the determination of epithelial cell kinetic parameters of human colonic epithelium isolated from surgical and biopsy specimens. Gastroenterology 86: 78–85.
Dieleman LA, Ridwan BU, Tennyson GS, Beagley KW, Bucy RP, Elson CO . (1994). Dextran sulfate sodium-induced colitis occurs in severe combined immunodeficient mice. Gastroenterology 107: 1643–1652.
Esteller M . (2008). Epigenetics in cancer. N Engl J Med 358: 1148–1159.
Fraga MF, Herranz M, Espada J, Ballestar E, Paz MF, Ropero S et al. (2004). A mouse skin multistage carcinogenesis model reflects the aberrant DNA methylation patterns of human tumors. Cancer Res 64: 5527–5534.
Garrity-Park MM, Loftus Jr EV., Sandborn WJ, Bryant SC, Smyrk TC . (2010). Methylation Status of Genes in Non-Neoplastic Mucosa From Patients With Ulcerative Colitis-Associated Colorectal Cancer. Am J Gastroenterol.
Grivennikov S, Karin E, Terzic J, Mucida D, Yu GY, Vallabhapurapu S et al. (2009). IL-6 and Stat3 are required for survival of intestinal epithelial cells and development of colitis-associated cancer. Cancer Cell 15: 103–113.
Hahn MA, Hahn T, Lee DH, Esworthy RS, Kim BW, Riggs AD et al. (2008). Methylation of polycomb target genes in intestinal cancer is mediated by inflammation. Cancer Res 68: 10280–10289.
Hanada T, Kobayashi T, Chinen T, Saeki K, Takaki H, Koga K et al. (2006). IFNgamma-dependent, spontaneous development of colorectal carcinomas in SOCS1-deficient mice. J Exp Med 203: 1391–1397.
Hmadcha A, Bedoya FJ, Sobrino F, Pintado E . (1999). Methylation-dependent gene silencing induced by interleukin 1beta via nitric oxide production. J Exp Med 190: 1595–1604.
Hodge DR, Xiao W, Clausen PA, Heidecker G, Szyf M, Farrar WL . (2001). Interleukin-6 regulation of the human DNA methyltransferase (HDNMT) gene in human erythroleukemia cells. J Biol Chem 276: 39508–39511.
Hsieh CJ, Klump B, Holzmann K, Borchard F, Gregor M, Porschen R . (1998). Hypermethylation of the p16INK4a promoter in colectomy specimens of patients with long-standing and extensive ulcerative colitis. Cancer Res 58: 3942–3945.
Hur K, Niwa T, Toyoda T, Tsukamoto T, Tatematsu M, Yang HK et al. (2011). Insufficient role of cell proliferation in aberrant DNA methylation induction and involvement of specific types of inflammation. Carcinogenesis 32: 35–41.
Issa JP, Ahuja N, Toyota M, Bronner MP, Brentnall TA . (2001). Accelerated age-related CpG island methylation in ulcerative colitis. Cancer Res 61: 3573–3577.
Ito R, Shin-Ya M, Kishida T, Urano A, Takada R, Sakagami J et al. (2006). Interferon-gamma is causatively involved in experimental inflammatory bowel disease in mice. Clin Exp Immunol 146: 330–338.
Jones PA, Baylin SB . (2007). The epigenomics of cancer. Cell 128: 683–692.
Kass DH, Kim J, Rao A, Deininger PL . (1997). Evolution of B2 repeats: the muroid explosion. Genetica 99: 1–13.
Keshet I, Schlesinger Y, Farkash S, Rand E, Hecht M, Segal E et al. (2006). Evidence for an instructive mechanism of de novo methylation in cancer cells. Nat Genet 38: 149–153.
Kim YH, Lee HC, Kim SY, Yeom YI, Ryu KJ, Min BH et al. (2011). Epigenomic analysis of aberrantly methylated genes in colorectal cancer identifies genes commonly affected by epigenetic alterations. Ann Surg Oncol (doi:10.1245/s10434-011-1573-y).
Kondo Y, Kanai Y, Sakamoto M, Mizokami M, Ueda R, Hirohashi S . (2000). Genetic instability and aberrant DNA methylation in chronic hepatitis and cirrhosis—A comprehensive study of loss of heterozygosity and microsatellite instability at 39 loci and DNA hypermethylation on 8 CpG islands in microdissected specimens from patients with hepatocellular carcinoma. Hepatology 32: 970–979.
Kuhn R, Lohler J, Rennick D, Rajewsky K, Muller W . (1993). Interleukin-10-deficient mice develop chronic enterocolitis. Cell 75: 263–274.
Li Y, de Haar C, Chen M, Deuring J, Gerrits MM, Smits R et al. (2009). Disease-related expression of the IL-6/STAT3/SOCS3 signaling pathway in ulcerative colitis and ulcerative colitis-related carcinogenesis. Gut 59: 227–235.
Ligumsky M, Simon PL, Karmeli F, Rachmilewitz D . (1990). Role of interleukin 1 in inflammatory bowel disease—enhanced production during active disease. Gut 31: 686–689.
Maekita T, Nakazawa K, Mihara M, Nakajima T, Yanaoka K, Iguchi M et al. (2006). High levels of aberrant DNA methylation in Helicobacter pylori-infected gastric mucosae and its possible association with gastric cancer risk. Clin Cancer Res 12: 989–995.
McLaughlan JM, Seth R, Vautier G, Robins RA, Scott BB, Hawkey CJ et al. (1997). Interleukin-8 and inducible nitric oxide synthase mRNA levels in inflammatory bowel disease at first presentation. J Pathol 181: 87–92.
Nagao M, Ochiai M, Okochi E, Ushijima T, Sugimura T . (2001). LacI transgenic animal study: relationships among DNA-adduct levels, mutant frequencies and cancer incidences. Mutat Res 477: 119–124.
Nakajima T, Maekita T, Oda I, Gotoda T, Yamamoto S, Umemura S et al. (2006). Higher methylation levels in gastric mucosae significantly correlate with higher risk of gastric cancers. Cancer Epidemiol Biomarkers Prev 15: 2317–2321.
Ni J, Chen SF, Hollander D . (1996). Effects of dextran sulphate sodium on intestinal epithelial cells and intestinal lymphocytes. Gut 39: 234–241.
Niwa T, Tsukamoto T, Toyoda T, Mori A, Tanaka H, Maekita T et al. (2010). Inflammatory processes triggered by Helicobacter pylori infection cause aberrant DNA methylation in gastric epithelial cells. Cancer Res 70: 1430–1440.
Niwa T, Yamashita S, Tsukamoto T, Kuramoto T, Nomoto T, Wakazono K et al. (2005). Whole-genome analyses of loss of heterozygosity and methylation analysis of four tumor-suppressor genes in N-methyl-N’-nitro-N-nitrosoguanidine-induced rat stomach carcinomas. Cancer Sci 96: 409–413.
Popivanova BK, Kitamura K, Wu Y, Kondo T, Kagaya T, Kaneko S et al. (2008). Blocking TNF-alpha in mice reduces colorectal carcinogenesis associated with chronic colitis. J Clin Invest 118: 560–570.
Qian X, Huang C, Cho CH, Hui WM, Rashid A, Chan AO . (2008). E-cadherin promoter hypermethylation induced by interleukin-1beta treatment or H. pylori infection in human gastric cancer cell lines. Cancer Lett 263: 107–113.
Riggs AD, Xiong Z . (2004). Methylation and epigenetic fidelity. Proc Natl Acad Sci USA 101: 4–5.
Rosenberg DW, Giardina C, Tanaka T . (2009). Mouse models for the study of colon carcinogenesis. Carcinogenesis 30: 183–196.
Sadlack B, Merz H, Schorle H, Schimpl A, Feller AC, Horak I . (1993). Ulcerative colitis-like disease in mice with a disrupted interleukin-2 gene. Cell 75: 253–261.
Schulmann K, Sterian A, Berki A, Yin J, Sato F, Xu Y et al. (2005). Inactivation of p16, RUNX3, and HPP1 occurs early in Barrett's-associated neoplastic progression and predicts progression risk. Oncogene 24: 4138–4148.
Takeshima H, Yamashita S, Shimazu T, Niwa T, Ushijima T . (2009). The presence of RNA polymerase II, active or stalled, predicts epigenetic fate of promoter CpG islands. Genome Res 19: 1974–1982.
Tanaka T, Kohno H, Suzuki R, Yamada Y, Sugie S, Mori H . (2003). A novel inflammation-related mouse colon carcinogenesis model induced by azoxymethane and dextran sodium sulfate. Cancer Sci 94: 965–973.
Taniwaki K, Fukamachi H, Komori K, Ohtake Y, Nonaka T, Sakamoto T et al. (2007). Stroma-derived matrix metalloproteinase (MMP)-2 promotes membrane type 1-MMP-dependent tumor growth in mice. Cancer Res 67: 4311–4319.
Ushijima T . (2005). Detection and interpretation of altered methylation patterns in cancer cells. Nat Rev Cancer 5: 223–231.
Ushijima T . (2007). Epigenetic field for cancerization. J Biochem Mol Biol 40: 142–150.
Ushijima T, Okochi-Takada E . (2005). Aberrant methylations in cancer cells: where do they come from? Cancer Sci 96: 206–211.
Vuillemenot BR, Pulling LC, Palmisano WA, Hutt JA, Belinsky SA . (2004). Carcinogen exposure differentially modulates RAR-beta promoter hypermethylation, an early and frequent event in mouse lung carcinogenesis. Carcinogenesis 25: 623–629.
Wang D, Dubois RN . (2010). The role of COX-2 in intestinal inflammation and colorectal cancer. Oncogene 29: 781–788.
Yamashita S, Hosoya K, Gyobu K, Takeshima H, Ushijima T . (2009). Development of a novel output value for quantitative assessment in methylated DNA immunoprecipitation-CpG island microarray analysis. DNA Res 16: 275–286.
Yamashita S, Takahashi S, McDonell N, Watanabe N, Niwa T, Hosoya K et al. (2008). Methylation silencing of transforming growth factor-beta receptor type II in rat prostate cancers. Cancer Res 68: 2112–2121.
Yu L, Liu C, Vandeusen J, Becknell B, Dai Z, Wu YZ et al. (2005). Global assessment of promoter methylation in a mouse model of cancer identifies ID4 as a putative tumor-suppressor gene in human leukemia. Nat Genet 37: 265–274.
Acknowledgements
We thank Dr H Fukamachi for his kind provision of the LIF-16 cell line. This study was supported by the Third-term Comprehensive Cancer Control Strategy from the Ministry of Health, Labour and Welfare, Japan; and by the Global Research Laboratory Program from Korea Foundation for International Cooperation of Science & Technology. MK and YS are recipients of Research Resident Fellowships from the Foundation for Promotion of Cancer Research.
This study was supported by the Third-term Comprehensive Cancer Control Strategy from the Ministry of Health, Labour and Welfare, Japan (TU), and by the Global Research Laboratory Program from Korea Foundation for International Cooperation of Science & Technology (Y-JK and TU).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Rights and permissions
About this article
Cite this article
Katsurano, M., Niwa, T., Yasui, Y. et al. Early-stage formation of an epigenetic field defect in a mouse colitis model, and non-essential roles of T- and B-cells in DNA methylation induction. Oncogene 31, 342–351 (2012). https://doi.org/10.1038/onc.2011.241
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2011.241
Keywords
This article is cited by
-
Bridging tissue repair and epithelial carcinogenesis: epigenetic memory and field cancerization
Cell Death & Differentiation (2024)
-
Marked intestinal trans-differentiation by autoimmune gastritis along with ectopic pancreatic and pulmonary trans-differentiation
Journal of Gastroenterology (2024)
-
Genotoxicity assessment: opportunities, challenges and perspectives for quantitative evaluations of dose–response data
Archives of Toxicology (2023)
-
Intestinal microbiota regulates diabetes and cancer progression by IL-1β and NOX4 dependent signaling cascades
Cellular and Molecular Life Sciences (2022)
-
Autoimmune gastritis induces aberrant DNA methylation reflecting its carcinogenic potential
Journal of Gastroenterology (2022)