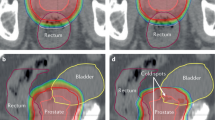
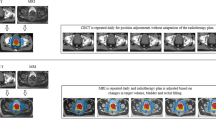
Key Points
-
Moderate and extreme hypofractionation are alternatives to conventional radiotherapy for organ-confined prostate cancer that were developed following technological advances in radiation delivery and tumour imaging
-
In strategies of moderate and extreme hypofractionation, increased radiation doses are administered per treatment session, reducing overall treatment duration and improving patient compliance
-
Prostate cancer seems particularly suitable for hypofractionated radiotherapy as these tumours have unique sensitivity to increased radiation dose fractions in comparison with surrounding healthy tissues
-
Trials investigating clinical and toxicity outcomes of moderate hypofractionation schedules have sufficient follow-up data to show that efficacy and toxicity of these schedules are similar to those of conventionally fractionated regimens
-
Several phase II trials of extremely hypofractionated radiotherapy in men with low-risk or intermediate-risk prostate cancer show excellent short-term outcomes, but extended follow-up monitoring is required to confirm long-term safety
-
Extreme hypofractionation schedules are highly cost-effective, despite the need for cutting-edge technologies, but the efficacy and safety of dose escalation and single-dose treatments still need to be confirmed in carefully conducted clinical trials
Abstract
Moderate hypofractionation of radiotherapy is widely considered a viable alternative to conventional fractionation for the treatment of patients with organ-confined prostate cancer, but stereotactic body radiotherapy (SBRT) is rapidly emerging as a novel treatment modality for this disease. Advances in treatment planning, image guidance, target position reproducibility and on-line tracking, coupled with a compelling radiobiological rationale, have promoted SBRT as a safe and effective treatment. Dose escalation to the tumour tissue through a decreased number of radiation fractions improves patient comfort and convenience, as well as treatment cost-effectiveness, compared with conventional radiotherapy regimens. Several clinical trials have investigated moderate and extreme hypofractionation of radiotherapy in patients with prostate cancer. Evidence is accumulating which suggests that the use of moderately hypofractionated radiotherapy can be recommended regardless of cancer risk group. Regimens of extremely hypofractionated radiotherapy have shown very good short-term efficacy and safety outcomes, but appropriately designed trials with extended follow-up monitoring are required to confirm long-term outcomes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Staffurth, J. A review of the clinical evidence for intensity-modulated radiotherapy. Clin. Oncol. (R. Coll. Radiol.) 22, 643–657 (2010).
Dawson, L. A. & Menard, C. Imaging in radiation oncology: a perspective. Oncologist 15, 338–349 (2010).
Verellen, D. et al. Innovations in image-guided radiotherapy. Nat. Rev. Cancer 7, 949–960 (2007).
Timmerman, R. D., Kavanagh, B. D., Cho, L. C., Papiez, L. & Xing, L. Stereotactic body radiation therapy in multiple organ sites. J. Clin. Oncol. 25, 947–952 (2007).
Steel, G. G. (ed.) Basic Clinical Radiobiology (Arnold, 2002).
Proust-Lima, C. et al. Confirmation of a low α/β ratio for prostate cancer treated by external beam radiation therapy alone using a post-treatment repeated-measures model for PSA dynamics. Int. J. Radiat. Oncol. Biol. Phys. 79, 195–201 (2011).
Miralbell, R., Roberts, S. A., Zubizarreta, E. & Hendry, J. H. Dose-fractionation sensitivity of prostate cancer deduced from radiotherapy outcomes of 5,969 patients in seven international institutional datasets: α/β=1.4 (0.9–2.2) Gy. Int. J. Radiat. Oncol. Biol. Phys. 82, e17–e24 (2012).
Dasu, A. & Toma-Dasu, I. Prostate α/β revisited — an analysis of clinical results from 14168 patients. Acta Oncol. 51, 963–974 (2012).
Fowler, J. F. The linear-quadratic formula and progress in fractionated radiotherapy. Br. J. Radiol. 62, 679–694 (1989).
Demaria, S. et al. Ionizing radiation inhibition of distant untreated tumors (abscopal effect) is immune mediated. Int. J. Radiat. Oncol. Biol. Phys. 58, 862–870 (2004).
Rubner, Y. et al. How does ionizing irradiation contribute to the induction of anti-tumor immunity? Front. Oncol. 2, 75 (2012).
Bonner, J. A. et al. Radiotherapy plus cetuximab for locoregionally advanced head and neck cancer: 5-year survival data from a phase 3 randomised trial, and relation between cetuximab-induced rash and survival. Lancet Oncol. 11, 21–28 (2010).
Heron, D. E. et al. Stereotactic body radiotherapy for recurrent squamous cell carcinoma of the head and neck: results of a phase I dose-escalation trial. Int. J. Radiat. Oncol. Biol. Phys. 75, 1493–1500 (2009).
Zaorsky, N. G., Ohri, N., Showalter, T. N., Dicker, A. P. & Den, R. B. Systematic review of hypofractionated radiation therapy for prostate cancer. Cancer Treat. Rev. 39, 728–736 (2013).
Koontz, B. F. et al. A systematic review of hypofractionation for primary management of prostate cancer. Eur. Urol. 68, 683–691 (2015).
Lukka, H. et al. Randomized trial comparing two fractionation schedules for patients with localized prostate cancer. J. Clin. Oncol. 23, 6132–6138 (2005).
Yeoh, E. E. et al. Hypofractionated versus conventionally fractionated radiotherapy for prostate carcinoma: final results of phase III randomized trial. Int. J. Radiat. Oncol. Biol. Phys. 81, 1271–1278 (2011).
Kuban, D. A. et al. Preliminary report of a randomized dose escalation trial for prostate cancer using hypofractionation. Int. J. Radiat. Oncol. Biol. Phys. 78, S58–S59 (2010).
Pollack, A. et al. Randomized trial of hypofractionated external-beam radiotherapy for prostate cancer. J. Clin. Oncol. 31, 3860–3868 (2013).
Arcangeli, S. et al. Updated results and patterns of failure in a randomized hypofractionation trial for high-risk prostate cancer. Int. J. Radiat. Oncol. Biol. Phys. 84, 1172–1178 (2012).
International Standard Randomised Controlled Trial Number Register. www.isrctn.comhttp://www.isrctn.com/ISRCTN85138529 (2015).
International Standard Randomised Controlled Trial Number Register. www.isrctn.comhttp://www.isrctn.com/ISRCTN97182923 (2015).
US National Library of Medicine. www.clinicaltrials.govhttps://clinicaltrials.gov/ct2/show/NCT00331773 (2016).
US National Library of Medicine. www.clinicaltrials.govhttps://clinicaltrials.gov/ct2/show/NCT00304759 (2015).
START Trialists' Group et al. The UK Standardisation of Breast Radiotherapy (START) Trial B of radiotherapy hypofractionation for treatment of early breast cancer: a randomised trial. Lancet 371, 1098–1107 (2008).
START Trialists' Group et al. The UK Standardisation of Breast Radiotherapy (START) Trial A of radiotherapy hypofractionation for treatment of early breast cancer: a randomised trial. Lancet Oncol. 9, 331–341 (2008).
Whelan, T. J. et al. Long-term results of hypofractionated radiation therapy for breast cancer. N. Engl. J. Med. 362, 513–520 (2010).
Aluwini, S. et al. Hypofractionated versus conventionally fractionated radiotherapy for patients with prostate cancer (HYPRO): late toxicity results from a randomised, non-inferiority, phase 3 trial. Lancet Oncol. 17, 464–474 (2016).
Lee, W. R. et al. Randomized phase III noninferiority study comparing two radiotherapy fractionation schedules in patients with low-risk prostate cancer. J. Clin. Oncol. http://dx.doi.org/10.1200/JCO.2016.67.0448 (2016).
Incrocci, L. et al. Hypofractionated versus conventionally fractionated radiotherapy for prostate cancer: 5-year oncologic outcomes of the Dutch randomized phase 3 HYPRO trial. Int. J. Radiat. Oncol. Biol. Phys. 94, A1–A2 (2016).
Dearnaley, D. LBA8. 5 year outcomes of a phase III randomised trial of conventional or hypofractionated high dose intensity modulated radiotherapy for prostate cancer (CRUK/06/016): report from the CHHiP Trial Investigators Group. Eur. J. Cancer 51, S712 (2015).
Collins C. D., Lloyd-Davies R. W. & Swan A. V. Radical external beam radiotherapy for localised carcinoma of the prostate using a hypofractionation technique. Clin. Oncol. (R. Coll. Radiol.) 3, 127–132 (1991).
Fuller, D. B. et al. Prospective evaluation of stereotactic body radiation therapy for low-and intermediate-risk prostate cancer: emulating high-doserate brachytherapy dose distribution. Int. J. Radiat. Oncol. Biol. Phys. 84, S149 (2012).
International Standard Randomised Controlled Trial Number Register. www.isrctn.comhttp://www.isrctn.com/ISRCTN45905321 (2009).
Fuller, D. B., Naitoh, J. & Mardirossian, G. Virtual HDR CyberKnife SBRT for localized prostatic carcinoma: 5-year disease-free survival and toxicity observations. Front. Oncol. 4, 321 (2014).
Madsen, B. L. et al. Stereotactic hypofractionated accurate radiotherapy of the prostate (SHARP), 33.5 Gy in five fractions for localized disease: first clinical trial results. Int. J. Radiat. Oncol. Biol. Phys. 67, 1099–1105 (2007).
Aluwini, S. et al. Stereotactic body radiotherapy with a focal boost to the MRI-visible tumor as monotherapy for low- and intermediate-risk prostate cancer: early results. Radiat. Oncol. 8, 84 (2013).
Loblaw, A. et al. Prostate stereotactic ablative body radiotherapy using a standard linear accelerator: toxicity, biochemical, and pathological outcomes. Radiother. Oncol. 107, 153–158 (2013).
Kim, D. W. N. et al. Predictors of rectal tolerance observed in a dose-escalated phase 1–2 trial of stereotactic body radiation therapy for prostate cancer. Int. J. Radiat. Oncol. Biol. Phys. 89, 509–517 (2014).
King, C. R., Brooks, J. D., Gill, H. & Presti, J. C. Long-term outcomes from a prospective trial of stereotactic body radiotherapy for low-risk prostate cancer. Int. J. Radiat. Oncol. Biol. Phys. 82, 877–882 (2012).
Bolzicco, G. et al. A single-center study of 100 consecutive patients with localized prostate cancer treated with stereotactic body radiotherapy. BMC Urol. 13, 49 (2013).
Chen, L. N. et al. Stereotactic body radiation therapy (SBRT) for clinically localized prostate cancer: the Georgetown University experience. Radiat. Oncol. 8, 58 (2013).
Oliai, C. et al. Stereotactic body radiation therapy for the primary treatment of localized prostate cancer. J. Radiat. Oncol. 2, 63–70 (2013).
Meier, R., Beckman, A. C., Henning, G. & Mohideen, N. Stereotactic body radiation therapy for intermediate-risk prostate cancer: five-year outcomes from a multi-institutional study. Int. J. Radiat. Oncol. Biol. Phys. 93(3S), S199 (2015).
Katz, A. J. & Kang, J. Quality of life and toxicity after SBRT for organ-confined prostate cancer, a 7-year study. Front. Oncol. 4, 301 (2014).
Hoppe, B. S. et al. Erectile function, incontinence, and other quality of life outcomes following protontherapy for prostate cancer in men 60 years old and younger. Cancer 118, 4619–4626 (2012).
Coen, J. J. et al. Long-term quality of life outcome after proton beam monotherapy for localized prostate cancer. Int. J. Radiat. Oncol. Biol. Phys. 82, e201–e209 (2012).
Demanes, D. J., Rodriguez, R. R., Schour, L., Brandt, D. & Altieri, G. High-dose rate intensity-modulated brachytherapy with external beam radiotherapy for prostate cancer: California endocurietherapy's 10-year results. Int. J. Radiat. Oncol. Biol. Phys. 61, 1306–1316 (2005).
Frank, S. J. et al. An assessment of quality of life following radical prostatectomy, high dose external beam radiation therapy and brachytherapy iodine implantation as monotherapies for localized prostate cancer. J. Urol. 177, 2151–2156 (2007).
King, C. R. et al. Stereotactic body radiotherapy for localized prostate cancer: pooled analysis from a multi-institutional consortium of prospective phase II trials. Radiother. Oncol. 109, 217–221 (2013).
King, C. R. et al. Health- related quality of life after stereotactic body radiation therapy for localized prostate cancer: results from a multi-institutional consortium of prospective trials. Int. J. Radiat. Oncol. Biol. Phys. 87, 939–945 (2013).
Sanda, M. G. et al. Quality of life and satisfaction with outcome among prostate-cancer survivors. N. Engl. J. Med. 358, 1250–1261 (2008).
Yu, J. B. et al. Stereotactic body radiation therapy versus intensity-modulated radiation therapy for prostate cancer: comparison of toxicity. J. Clin. Oncol. 32, 1195–1201 (2014).
Arcangeli, S., De Bari, B. & Alongi, F. Toxicity of stereotactic body radiation therapy versus intensity-modulated radiation therapy for prostate cancer: a potential comparison bias. J. Clin. Oncol. 32, 3454 (2014).
Evans, J. R. et al. Patient-reported quality of life after stereotactic body radiotherapy (SBRT), intensity modulated radiotherapy (IMRT), and brachytherapy. Radiother. Oncol. 116, 179–184 (2015).
US National Library of Medicine. www.clinicaltrials.govhttps://clinicaltrials.gov/ct2/show/NCT01434290 (2015).
Lukka, H., Stephanie, P., Bruner, D., Bahary, J. P. Lawton, C. A. F. & Efstathiou, J. A. Patient-reported outcomes in NRG Oncology/RTOG 0938, a randomized phase 2 study evaluating 2 ultra hypofractionated regimens (UHRs) for prostate cancer. Int. J. Radiat. Oncol. Biol. Phys. 94, A2 (2016).
Widmark, A. et al. SP-0299. Extreme hypofractionation: indications and results. Radiother. Oncol. 119 (Suppl. 1), S139 (2016).
Wiegner, E. A. & King, C. R. Sexual function after stereotactic body radiotherapy for prostate cancer: results of a prospective clinical trial. Int. J. Radiat. Oncol. Biol. Phys. 78, 442–448 (2010).
Bauman, G., Haider, M., Van der Heide, U. A. & Menard, C. Boosting imaging defined dominant prostatic tumors: a systematic review. Radiother. Oncol. 107, 274–281 (2013).
Pucar, D. et al. Clinically significant prostate cancer local recurrence after radiation therapy occurs at the site of primary tumor: magnetic resonance imaging and step-section pathology evidence. Int. J. Radiat. Oncol. Biol. Phys. 69, 62–69 (2007).
Zumsteg, Z. S. et al. Anatomical patterns of recurrence following biochemical relapse in the dose escalation era of external beam radiotherapy for prostate cancer. J. Urol. 194, 1624–1630 (2015).
Kron, T. et al. Intra-fraction prostate displacement in radiotherapy estimated from pre- and post-treatment imaging of patients with implanted fiducial markers. Radiother. Oncol. 95, 191–197 (2010).
Kupelian, P. et al. Multi-institutional clinical experience with the Calypso System in localization and continuous, real-time monitoring of the prostate gland during external radiotherapy. Int. J. Radiat. Oncol. Biol. Phys. 67, 1088–1098 (2007).
Teh, B. S. et al. The use of rectal balloon during the delivery of intensity modulated radiotherapy (IMRT) for prostate cancer: more than just a prostate gland immobilization device? Cancer J. 8, 476–483 (2002).
Sanghani, M. V. et al. Impact on rectal dose from the use of a prostate immobilization and rectal localization device for patients receiving dose escalated 3D conformal radiation therapy. Urol. Oncol. 22, 165–168 (2004).
van Lin, E. N., Hoffmann, A. L., van Kollenburg, P., Leer, J. W. & Visser, A. G. Rectal wall sparing effect of three different endorectal balloons in 3D conformal and IMRT prostate radiotherapy. Int. J. Radiat. Oncol. Biol. Phys. 63, 565–576 (2005).
Greco, C. Extreme hypofractionated image-guided radiotherapy (IGRT) for prostate cancer. EMJ Oncol. 1, 48–55 (2013).
US National Library of Medicine. www.clinicaltrials.govhttps://clinicaltrials.gov/ct2/show/NCT02570919 (2015).
The Department of Veterans Affairs Laryngeal Cancer Study Group. Induction chemotherapy plus radiation compared with surgery plus radiation in patients with advanced laryngeal cancer. N. Engl. J. Med. 324, 1685–1690 (1991).
Landoni, F. et al. Randomised study of radical surgery versus radiotherapy for stage Ib-IIa cervical cancer. Lancet 350, 535–540 (1997).
Crook, J. M. et al. Comparison of health-related quality of life 5 years after SPIRIT: surgical prostatectomy versus interstitial radiation intervention trial. J. Clin. Oncol. 29, 362–368 (2011).
Yabroff, K. R. et al. Annual patient time costs associated with medical care among cancer survivors in the United States. Med. Care 52, 594–601 (2014).
Hodges, J. C. et al. Cost-effectiveness analysis of stereotactic body radiation therapy versus intensity-modulated radiation therapy: an emerging initial radiation treatment option for organ-confined prostate cancer. J. Oncol. Pract. 83 (3 Suppl.), e31s–e37s (2012).
Fowler, J. F. The radiobiology of prostate cancer including new aspects of fractionated radiotherapy. Acta Oncol. 44, 265–276 (2005).
International Standard Randomised Controlled Trial Number Register. www.isrctn.comhttp://www.isrctn.com/ISRCTN17627211 (2015).
Ramsay, C. R. et al. Statistical assessment of the learning curves of health technologies. Health Technol. Assess. 5, 1–79 (2001).
Author information
Authors and Affiliations
Contributions
S.A. researched data for the article and wrote the manuscript. Both authors made substantial contributions to the discussion of the article content and reviewed and/or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Glossary
- Intensity-modulated radiotherapy
-
A type of 3D radiotherapy that uses thin radiation beams of differing intensities aimed at the tumour from many angles.
- Image-guided radiation therapy
-
Radiotherapy that employs image guidance at the time of radiation delivery to ensure adherence to the planned treatment.
- Stereotactic body radiotherapy
-
Radiotherapy that concentrates therapeutic doses into few, high-dose, highly conformal and precisely targeted treatments.
- α/β ratio
-
A parameter that determines the sensitivity of particular types of tissues to radiation doses, derived from the underlying dose–effect curves (linear or quadratic).
- Isoeffective
-
Radiotherapy regimens that achieve the same tumour control probability.
- Tumour control probability
-
The probability that a tumour is being eradicated or controlled by a prescribed dose of radiation taking into account factors such as cell proliferation between fractions, which can be used to compare different radiotherapy regimens.
- 3D-conformal radiotherapy
-
A type of radiotherapy that uses computers and special imaging techniques to define the size and shape of the tumour in three dimensions (width, height and depth).
- 2D-volume imaging
-
Any planar imaging that uses 'flat' X-ray images generated by a fluoroscopic simulator to plan the radiation treatments.
- Fiducial markers
-
Small metal spheres, coils or cylinders that are placed in or near a tumour to help guide the placement of radiation beams during treatment.
- Expanded prostate cancer index composite
-
A standardized self-administered disease-specific questionnaire developed to measure health-related quality of life in patients treated for prostate cancer.
- Fast flattening-filter-free beam radiation
-
Procedure that enables a considerable reduction in treatment time through radiotherapy delivery via unflattened beams.
Rights and permissions
About this article
Cite this article
Arcangeli, S., Greco, C. Hypofractionated radiotherapy for organ-confined prostate cancer: is less more?. Nat Rev Urol 13, 400–408 (2016). https://doi.org/10.1038/nrurol.2016.106
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrurol.2016.106
This article is cited by
-
New approaches for effective and safe pelvic radiotherapy in high-risk prostate cancer
Nature Reviews Urology (2019)
-
Late toxicity of image-guided hypofractionated radiotherapy for prostate: non-randomized comparison with conventional fractionation
La radiologia medica (2019)
-
Impact of image guidance on toxicity and tumour outcome in moderately hypofractionated external-beam radiotherapy for prostate cancer
Medical Oncology (2019)
-
“Give me five” ultra-hypofractionated radiotherapy for localized prostate cancer: non-invasive ablative approach
Medical Oncology (2018)