Abstract
Viruses of the genera Ebolavirus and Marburgvirus are filoviruses that cause haemorrhagic fever in primates, with extremely high fatality rates. Studies have focused on elucidating how these viruses enter host cells, with the aim of developing therapeutics. The ebolavirus glycoprotein has been found to play key parts in all steps of entry. Furthermore, recent studies have identified Niemann–Pick C1 (NPC1), a protein that resides deep in the endocytic pathway, as an important host factor in this process.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Feldmann, H. & Geisbert, T. W. Ebola haemorrhagic fever. Lancet 377, 849–862 (2011).
Sullivan, N. J., Martin, J. E., Graham, B. S. & Nabel, G. J. Correlates of protective immunity for Ebola vaccines: implications for regulatory approval by the animal rule. Nature Rev. Microbiol. 7, 393–400 (2009).
Kuhn, J. H. et al. Proposal for a revised taxonomy of the family Filoviridae: classification, names of taxa and viruses, and virus abbreviations. Arch. Virol. 155, 2083–2103 (2010).
Borio, L. et al. Hemorrhagic fever viruses as biological weapons: medical and public health management. JAMA 287, 2391–2405 (2002).
Miranda, M. E. & Miranda, N. L. Reston ebolavirus in humans and animals in the Philippines: a review. J. Infect. Dis. 204 (Suppl. 3), S757–S760 (2011).
Negredo, A. et al. Discovery of an ebolavirus-like filovirus in Europe. PLoS Pathog. 7, e1002304 (2011).
Dube, D. et al. Cell adhesion-dependent membrane trafficking of a binding partner for the ebolavirus glycoprotein is a determinant of viral entry. Proc. Natl Acad. Sci. USA 107, 16637–16642 (2010).
Backovic, M. & Rey, F. Virus entry: old viruses, new receptors. Curr. Opin. Virol. 2, 4–13 (2012).
Matsuno, K. et al. C-type lectins do not act as functional receptors for filovirus entry into cells. Biochem. Biophys. Res. Commun. 403, 144–148 (2010).
Alvarez, C. P. et al. C-type lectins DC-SIGN and L-SIGN mediate cellular entry by Ebola virus in cis and in trans. J. Virol. 76, 6841–6844 (2002).
Simmons, G. et al. DC-SIGN and DC-SIGNR bind ebola glycoproteins and enhance infection of macrophages and endothelial cells. Virology 305, 115–123 (2003).
Takada, A. et al. Human macrophage C-type lectin specific for galactose and N-acetylgalactosamine promotes filovirus entry. J. Virol. 78, 2943–2947 (2004).
Dube, D. et al. The primed ebolavirus glycoprotein (19-kilodalton GP1,2): sequence and residues critical for host cell binding. J. Virol. 83, 2883–2891 (2009).
Kondratowicz, A. S. et al. T-cell immunoglobulin and mucin domain 1 (TIM-1) is a receptor for Zaire Ebolavirus and Lake Victoria Marburgvirus. Proc. Natl Acad. Sci. USA 108, 8426–8431 (2011).
Lee, J. E. & Saphire, E. O. Ebolavirus glycoprotein structure and mechanism of entry. Future Virol. 4, 621–635 (2009).
Shimojima, M., Ikeda, Y. & Kawaoka, Y. The mechanism of Axl-mediated Ebola virus infection. J. Infect. Dis. 196 (Suppl. 2), S259–S263 (2007).
Takada, A. et al. Downregulation of β1 integrins by Ebola virus glycoprotein: implication for virus entry. Virology 278, 20–26 (2000).
Brindley, M. A. et al. Tyrosine kinase receptor Axl enhances entry of Zaire ebolavirus without direct interactions with the viral glycoprotein. Virology 415, 83–94 (2011).
Schornberg, K. L. et al. α5β1-integrin controls ebolavirus entry by regulating endosomal cathepsins. Proc. Natl Acad. Sci. USA 106, 8003–8008 (2009).
Mulherkar, N., Raaben, M., de la Torre, J. C., Whelan, S. P. & Chandran, K. The Ebola virus glycoprotein mediates entry via a non-classical dynamin-dependent macropinocytic pathway. Virology 419, 72–83 (2011).
Nanbo, A. et al. Ebolavirus is internalized into host cells via macropinocytosis in a viral glycoprotein-dependent manner. PLoS Pathog. 6, e1001121 (2010).
Saeed, M. F., Kolokoltsov, A. A., Albrecht, T. & Davey, R. A. Cellular entry of Ebola virus involves uptake by a macropinocytosis-like mechanism and subsequent trafficking through early and late endosomes. PLoS Pathog. 6, e1001110 (2010).
Bharat, T. A. et al. Cryo-electron tomography of Marburg virus particles and their morphogenesis within infected cells. PLoS Biol. 9, e1001196 (2011).
Sanchez, A., Geisbert, T. & Feldmann, H. in Fields Virology (eds Knipe, D. & Howley, P.) 1407–1448 (Lippincott Williams and Wilkins, 2007).
Teissier, E., Penin, F. & Pecheur, E.-I. Targeting cell entry of enveloped viruses as an antiviral strategy. Molecules 16, 221–250 (2011).
Tilton, J. C. & Doms, R. W. Entry inhibitors in the treatment of HIV-1 infection. Antiviral Res. 85, 91–100 (2010).
Carette, J. E. et al. Ebola virus entry requires the cholesterol transporter Niemann–Pick C1. Nature 477, 340–343 (2011).
Cote, M. et al. Small molecule inhibitors reveal Niemann–Pick C1 is essential for Ebola virus infection. Nature 477, 344–348 (2011).
Miller, E. H. et al. Ebola virus entry requires the host-programmed recognition of an intracellular receptor. EMBO J. 6 Mar 2012 (doi:10.1038/emboj.2012.53).
Lee, J. E. et al. Structure of the Ebola virus glycoprotein bound to an antibody from a human survivor. Nature 454, 177–182 (2008).
White, J. M., Delos, S. E., Brecher, M. & Schornberg, K. Structures and mechanisms of viral membrane fusion proteins: multiple variations on a common theme. Crit. Rev. Biochem. Mol. Biol. 43, 189–219 (2008).
Harrison, S. C. Viral membrane fusion. Nature Struct. Mol. Biol. 15, 690–698 (2008).
Wool-Lewis, R. J. & Bates, P. Endoproteolytic processing of the Ebola virus envelope glycoprotein: cleavage is not required for function. J. Virol. 73, 1419–1426 (1999).
Neumann, G. et al. Proteolytic processing of the Ebola virus glycoprotein is not critical for Ebola virus replication in nonhuman primates. J. Virol. 81, 2995–2998 (2007).
Chandran, K., Sullivan, N. J., Felbor, U., Whelan, S. P. & Cunningham, J. M. Endosomal proteolysis of the Ebola virus glycoprotein is necessary for infection. Science 308, 1643–1645 (2005).
Schornberg, K. et al. Role of endosomal cathepsins in entry mediated by the Ebola virus glycoprotein. J. Virol. 80, 4174–4178 (2006).
Hood, C. L. et al. Biochemical and structural characterization of cathepsin L-processed Ebola virus glycoprotein: implications for viral entry and immunogenicity. J. Virol. 84, 2972–2982 (2010).
Dias, J. M. et al. A shared structural solution for neutralizing ebolaviruses. Nature Struct. Mol. Biol. 18, 1424–1427 (2011).
Kuhn, J. H. et al. Conserved receptor-binding domains of Lake Victoria marburgvirus and Zaire ebolavirus bind a common receptor. J. Biol. Chem. 281, 15951–15958 (2006).
Manicassamy, B., Wang, J., Jiang, H. & Rong, L. Comprehensive analysis of Ebola virus GP1 in viral entry. J. Virol. 79, 4793–4805 (2005).
Brindley, M. A. et al. Ebola virus glycoprotein 1: identification of residues important for binding and postbinding events. J. Virol. 81, 7702–7709 (2007).
Mpanju, O. M., Towner, J. S., Dover, J. E., Nichol, S. T. & Wilson, C. A. Identification of two amino acid residues on Ebola virus glycoprotein 1 critical for cell entry. Virus Res. 121, 205–214 (2006).
Wong, A. C., Sandesara, R. G., Mulherkar, N., Whelan, S. P. & Chandran, K. A forward genetic strategy reveals destabilizing mutations in the Ebolavirus glycoprotein that alter its protease dependence during cell entry. J. Virol. 84, 163–175 (2010).
Brecher, M. et al. Cathepsin cleavage potentiates the Ebola virus glycoprotein to undergo a subsequent fusion relevant conformational change. J. Virol. 86, 364–372 (2012).
Kaletsky, R. L., Simmons, G. & Bates, P. Proteolysis of the Ebola glycoproteins enhances virus binding and infectivity. J. Virol. 81, 13378–13384 (2007).
Gregory, S. M. et al. Structure and function of the complete internal fusion loop from Ebolavirus glycoprotein 2. Proc. Natl Acad. Sci. USA 108, 11211–11216 (2011).
Carette, J. E. et al. Haploid genetic screens in human cells identify host factors used by pathogens. Science 326, 1231–1235 (2009).
Epp, N., Rethmeier, R., Kramer, L. & Ungermann, C. Membrane dynamics and fusion at late endosomes and vacuoles – Rab regulation, multisubunit tethering complexes and SNAREs. Eur. J. Cell Biol. 90, 779–785 (2011).
Huotari, J. & Helenius, A. Endosome maturation. EMBO J. 30, 3481–3500 (2011).
Pryor, P. R. & Luzio, J. P. Delivery of endocytosed membrane proteins to the lysosome. Biochim. Biophys. Acta 1793, 615–624 (2009).
Frolov, A. et al. Cholesterol overload promotes morphogenesis of a Niemann-Pick C (NPC)-like compartment independent of inhibition of NPC1 or HE1/NPC2 function. J. Biol. Chem. 276, 46414–46421 (2001).
Lloyd-Evans, E. & Platt, F. M. Lipids on trial: the search for the offending metabolite in Niemann-Pick type C disease. Traffic 11, 419–428 (2010).
Kobayashi, T. et al. Late endosomal membranes rich in lysobisphosphatidic acid regulate cholesterol transport. Nature Cell Biol. 1, 113–118 (1999).
Vitner, E. B., Platt, F. M. & Futerman, A. H. Common and uncommon pathogenic cascades in lysosomal storage diseases. J. Biol. Chem. 285, 20423–20427 (2010).
Deffieu, M. S. & Pfeffer, S. R. Niemann–Pick type C 1 function requires lumenal domain residues that mediate cholesterol-dependent NPC2 binding. Proc. Natl Acad. Sci. USA 108, 18932–18936 (2011).
Lozach, P.-Y., Huotari, J. & Helenius, A. Late-penetrating viruses. Curr. Opin. Virol. 1, 35–43 (2011).
Lloyd-Evans, E. et al. Niemann-Pick disease type C1 is a sphingosine storage disease that causes deregulation of lysosomal calcium. Nature Med. 14, 1247–1255 (2008).
Liu, R., Lu, P., Chu, J. W. & Sharom, F. J. Characterization of fluorescent sterol binding to purified human NPC1. J. Biol. Chem. 284, 1840–1852 (2009).
Ko, D. C., Gordon, M. D., Jin, J. Y. & Scott, M. P. Dynamic movements of organelles containing Niemann-Pick C1 protein: NPC1 involvement in late endocytic events. Mol. Biol. Cell 12, 601–614 (2001).
Sobo, K. et al. Late endosomal cholesterol accumulation leads to impaired intra-endosomal trafficking. PLoS ONE 2, e851 (2007).
Acknowledgements
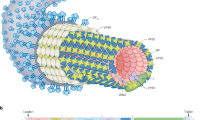
The authors acknowledge grant U54 AI057168 from the US National Institutes of Health for funding work on EBOV entry, M. Brecher and S. Gregory for help with figure 2b, and M. Brecher, D. Castle and J. Shoemaker for helpful comments on the text.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Rights and permissions
About this article
Cite this article
White, J., Schornberg, K. A new player in the puzzle of filovirus entry. Nat Rev Microbiol 10, 317–322 (2012). https://doi.org/10.1038/nrmicro2764
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrmicro2764