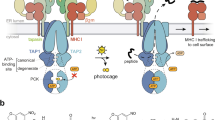
Key Points
-
Antigen processing and presentation is the mechanism by which whole antigens are degraded and loaded onto MHC molecules for display on the cell surface for recognition by T cells.
-
Both macrophages and dendritic cells (DCs) are considered professional antigen-presenting cells, although DCs possess the unique capacity to activate naive T cells.
-
DCs phagocytose antigens and whole microorganisms and place them into membrane-delimited compartments termed phagosomes. These structures are modified over time and ultimately fuse with lysosomes to form phagolysosomes.
-
Cross-presentation allows DCs to take up antigens from the extracellular environment and present them on MHC class I molecules for CD8+ T-cell activation. Although the exact trafficking patterns of antigens are not known, many hypotheses have been generated, including the recruitment of the endoplasmic reticulum dislocation machinery to the phagosome and protein-independent passing of antigens from the phagosome or phagolysosome into the cytosol.
-
The autophagy machinery is thought to have an important role in the generation of peptide antigens for MHC class II molecules.
Abstract
The principal components of both MHC class I and class II antigen processing and presentation pathways are well known. In dendritic cells, these pathways are tightly regulated by Toll-like-receptor signalling and include features, such as cross-presentation, that are not seen in other cell types. However, the exact mechanisms involved in the subcellular trafficking of antigens remain poorly understood and in some cases are controversial. Recent data suggest that diverse cellular machineries, including autophagy, participate in antigen processing and presentation, although their relative contributions remain to be fully elucidated. Here, we highlight some emerging themes of antigen processing and presentation that we think merit further attention.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Gell, P. G. H. & Benacerraf, B. Studies on hypersensitivity II. Delayed hypersensitivity to denatured proteins in guinea pigs. Immunology 2, 64–70 (1959).
Call, M. E. & Wucherpfennig, K. W. The T cell receptor: critical role of the membrane environment in receptor assembly and function. Ann. Rev. Immunol. 23, 101–125 (2005).
Martin, F. & Chan, A. C. B cell immunobiology in disease: evolving concepts from the clinic. Ann. Rev. Immunol. 24, 467–496 (2006).
Jensen, P. E. Recent advances in antigen processing and presentation. Nature Immunol. 8, 1041–1048 (2007).
Bryant, P. & Ploegh, H. Class II MHC peptide loading by the professionals. Curr. Opin. Immunol. 16, 96–102 (2004).
Lemaitre, B., Nicolas, E., Michaut, L., Reichhart, J.-M. & Hoffmann, J. A. The dorsoventral regulatory gene cassette spatzle/Toll/cactus controls the potent antifungal response in Drosophila adults. Cell 86, 973–983 (1996).
Reis e Sousa, C. Toll-like receptors and dendritic cells: for whom the bug tolls. Semin. Immunol. 16, 27–34 (2004).
Garrett, W. S. et al. Developmental control of endocytosis in dendritic cells by Cdc42. Cell 102, 325–334 (2000).
West, M. A. et al. Enhanced dendritic cell antigen capture via Toll-like receptor-induced actin remodeling. Science 305, 1153–1157 (2004). This paper shows that, on TLR stimulation, DCs initially increase macropinocytosis, which induces dramatic actin remodelling.
Trombetta, E. S., Ebersold, M., Garrett, W., Pypaert, M. & Mellman, I. Activation of lysosomal function during dendritic cell maturation. Science 299, 1400–1403 (2003).
Kleijmeer, M. et al. Reorganization of multivesicular bodies regulates MHC class II antigen presentation by dendritic cells. J. Cell Biol. 155, 53–64 (2001).
Boes, M. et al. T-cell engagement of dendritic cells rapidly rearranges MHC class II transport. Nature 418, 983–988 (2002).
Chow, A., Toomre, D., Garrett, W. & Mellman, I. Dendritic cell maturation triggers retrograde MHC class II transport from lysosomes to the plasma membrane. Nature 418, 988–994 (2002).
Savina, A. & Amigorena, S. Phagocytosis and antigen presentation in dendritic cells. Immunol. Rev. 219, 143–156 (2007).
Henry, R. M., Hoppe, A. D., Joshi, N. & Swanson, J. A. The uniformity of phagosome maturation in macrophages. J. Cell Biol. 164, 185–194 (2004).
Blander, J. M. & Medzhitov, R. On regulation of phagosome maturation and antigen presentation. Nature Immunol. 7, 1029–1035 (2006).
Stuart, L. M. & Ezekowitz, R. A. Phagocytosis and comparative innate immunity: learning on the fly. Nature Rev. Immunol. 8, 131–141 (2008).
Blander, J. M. & Medzhitov, R. Toll-dependent selection of microbial antigens for presentation by dendritic cells. Nature 440, 808–812 (2006). This paper shows that antigens complexed with TLR ligands are more efficiently presented to T cells.
Artavanis-Tsakonas, K., Love, J. C., Ploegh, H. L. & Vyas, J. M. Recruitment of CD63 to Cryptococcus neoformans phagosomes requires acidification. Proc. Natl Acad. Sci. USA 103, 15945–15950 (2006).
Meresse, S. et al. Controlling the maturation of pathogen-containing vacuoles: a matter of life and death. Nature Cell Biol. 1, E183–E188 (1999).
van der Wel, N. et al. M. tuberculosis and M. leprae translocate from the phagolysosome to the cytosol in myeloid cells. Cell 129, 1287–1298 (2007)). This paper shows that the mycobacterial gene products CFP-10 and ESAT-6 are required for M. tuberculosis and M. leprae to escape from phagolysosomes in human DCs and macrophages.
Kagan, J. C. & Roy, C. R. Legionella phagosomes intercept vesicular traffic from endoplasmic reticulum exit sites. Nature Cell Biol. 4, 945–954 (2002).
Mordue, D. G. & Sibley, L. D. Intracellular fate of vacuoles containing Toxoplasma gondii is determined at the time of formation and depends on the mechanism of entry. J. Immunol. 159, 4452–4459 (1997).
Yap, G. S., Shaw, M. H., Ling, Y. & Sher, A. Genetic analysis of host resistance to intracellular pathogens: lessons from studies of Toxoplasma gondii infection. Microbes Infect. 8, 1174–1178 (2006).
Geijtenbeek, T. B. et al. DC-SIGN, a dendritic cell-specific HIV-1-binding protein that enhances trans-infection of T cells. Cell 100, 587–597 (2000).
Geijtenbeek, T. B. et al. Identification of DC-SIGN, a novel dendritic cell-specific ICAM-3 receptor that supports primary immune responses. Cell 100, 575–585 (2000).
Jancic, C. et al. Rab27a regulates phagosomal pH and NADPH oxidase recruitment to dendritic cell phagosomes. Nature Cell Biol. 9, 367–378 (2007). This paper shows that limiting acidification in phagolysosomes in DCs allows antigen cross-presentation, as shown by RAB27A-deficient mice, which have increased phagosome acidification and antigen degradation, and a defect in antigen cross-presentation.
Asagiri, M. et al. Cathepsin K-dependent Toll-like receptor 9 signaling revealed in experimental arthritis. Science 319, 624–627 (2008). This study shows that cathespin K is required for TLR9-dependent signalling in DCs.
Xie, Z. & Klionsky, D. J. Autophagosome formation: core machinery and adaptations. Nature Cell Biol. 9, 1102–1109 (2007).
Schmid, D. & Munz, C. Innate and adaptive immunity through autophagy. Immunity 27, 11–21 (2007).
Huang, J. & Klionsky, D. J. Autophagy and human disease. Cell Cycle 6, 1837–1849 (2007).
Levine, B. & Deretic, V. Unveiling the roles of autophagy in innate and adaptive immunity. Nature Rev. Immunol. 7, 767–777 (2007).
Schmid, D., Pypaert, M. & Munz, C. Antigen-loading compartments for major histocompatibility complex class II molecules continuously receive input from autophagosomes. Immunity 26, 79–92 (2007). In DCs, autophagosomes formed by macroautophagy fuse with MHC class II-containing compartments. In this model, influenza-derived peptides are presented by MHC class II molecules.
Xu, Y. et al. Toll-like receptor 4 is a sensor for autophagy associated with innate immunity. Immunity 27, 135–144 (2007). This study shows that TLR4 ligands induce autophagy in macrophages, but do not serve as a signal for the cells to undergo apoptosis.
Delgado, M. A., Elmaoued, R. A., Davis, A. S., Kyei, G. & Deretic, V. Toll-like receptors control autophagy. EMBO J. 27, 1110–1121 (2008).
Sanjuan, M. A. et al. Toll-like receptor signalling in macrophages links the autophagy pathway to phagocytosis. Nature 450, 1253–1257 (2007).
Lee, H. K., Lund, J. M., Ramanathan, B., Mizushima, N. & Iwasaki, A. Autophagy-dependent viral recognition by plasmacytoid dendritic cells. Science 315, 1398–1401 (2007).
Stuart, L. M. et al. A systems biology analysis of the Drosophila phagosome. Nature 445, 95–101 (2007). This paper presents the most comprehensive list of proteins associated with the phagosome so far. Interesting relationships of proteins are shown in the systems analysis.
Garin, J. et al. The phagosome proteome: insight into phagosome functions. J. Cell Biol. 152, 165–180 (2001).
Jutras, I. et al. Modulation of the phagosome proteome by interferon-γ. Mol. Cell. Proteomics 7, 697–715 (2008).
Burlak, C., Whitney, A. R., Mead, D. J., Hackstadt, T. & Deleo, F. R. Maturation of human neutrophil phagosomes includes incorporation of molecular chaperones and endoplasmic reticulum quality control machinery. Mol. Cell. Proteomics 5, 620–634 (2006).
Liou, W., Geuze, H. J., Geelen, M. J. & Slot, J. W. The autophagic and endocytic pathways converge at the nascent autophagic vacuoles. J. Cell Biol. 136, 61–70 (1997).
Berg, T. O., Fengsrud, M., Strømhaug, P. E., Berg, T. & Seglen, P. O. Isolation and characterization of rat liver amphisomes. Evidence for fusion of autophagosomes with both early and late endosomes. J. Biol. Chem. 273, 21883–21892 (1998).
Paludan, C. et al. Endogenous MHC class II processing of a viral nuclear antigen after autophagy. Science 307, 593–596 (2005). This paper shows that a gene product of Epstein–Barr virus, EBNA1, is delivered by autophagy to the lysosome, where it intersects the MHC class II pathway.
Brazil, M. I., Weiss, S. & Stockinger, B. Excessive degradation of intracellular protein in macrophages prevents presentation in the context of major histocompatibility complex class II molecules. Eur. J. Immunol. 27, 1506–1514 (1997).
Dorfel, D. et al. Processing and presentation of HLA class I and II epitopes by dendritic cells after transfection with in vitro-transcribed MUC1 RNA. Blood 105, 3199–3205 (2005).
Nimmerjahn, F. et al. Major histocompatibility complex class II-restricted presentation of a cytosolic antigen by autophagy. Eur. J. Immunol. 33, 1250–1259 (2003).
Chicz, R. M. et al. Specificity and promiscuity among naturally processed peptides bound to HLA-DR alleles. J. Exp. Med. 178, 27–47 (1993).
Dongre, A. R. et al. In vivo MHC class II presentation of cytosolic proteins revealed by rapid automated tandem mass spectrometry and functional analyses. Eur. J. Immunol. 31, 1485–1494 (2001).
Muntasell, A. et al. HLA-DR4 molecules in neuroendocrine epithelial cells associate to a heterogeneous repertoire of cytoplasmic and surface self peptides. J. Immunol. 169, 5052–5060 (2002).
Dengjel, J. et al. Autophagy promotes MHC class II presentation of peptides from intracellular source proteins. Proc. Natl Acad. Sci. USA 102, 7922–7927 (2005).
Zhou, D. et al. Lamp-2a facilitates MHC class II presentation of cytoplasmic antigens. Immunity 22, 571–581 (2005).
Zwickey, H. L., Unternaehrer, J. J. & Mellman, I. Presentation of self-antigens on MHC class II molecules during dendritic cell maturation. Int. Immunol. 18, 199–209 (2006).
Boes, M. et al. T cells induce extended class II MHC compartments in dendritic cells in a Toll-like receptor-dependent manner. J. Immunol. 171, 4081–4088 (2003).
Vyas, J. M. et al. Tubulation of class II MHC compartments is microtubule dependent and involves multiple endolysosomal membrane proteins in primary dendritic cells. J. Immunol. 178, 7199–7210 (2007).
Jordens, I. et al. The Rab7 effector protein RILP controls lysosomal transport by inducing the recruitment of dynein-dynactin motors. Curr. Biol. 11, 1680–1685 (2001).
Bloom, O. et al. Spinophilin participates in information transfer at immunological synapses. J. Cell Biol. 181, 203–211 (2008).
Hiltbold, E. M., Poloso, N. J. & Roche, P. A. MHC class II-peptide complexes and APC lipid rafts accumulate at the immunological synapse. J. Immunol. 170, 1329–1338 (2003).
Shin, J.-S. et al. Surface expression of MHC class II in dendritic cells is controlled by regulated ubiquitination. Nature 444, 115–118 (2006). This study shows that regulation of cell-surface expression of MHC class II molecules by DCs is achieved in part by ubiquitylation of the cytoplasmic tail of MHC class II β-chain. Following DC maturation, the rate of this post-translational modification decreased, resulting in the accumulation of MHC class II molecules at the cell surface.
Hsu, S. C., TerBush, D., Abraham, M. & Guo, W. The exocyst complex in polarized exocytosis. Int. Rev. Cytol. 233, 243–265 (2004).
Yeaman, C., Grindstaff, K. K. & Nelson, W. J. Mechanism of recruiting Sec6/8 (exocyst) complex to the apical junctional complex during polarization of epithelial cells. J. Cell Sci. 117, 559–570 (2004).
Albert, M. L., Sauter, B. & Bhardwaj, N. Dendritic cells acquire antigen from apoptotic cells and induce class I-restricted CTLs. Nature 392, 86–89 (1998).
Bevan, M. J. Cross-priming for a secondary cytotoxic response to minor H antigens with H-2 congenic cells which do not cross-react in the cytotoxic assay. J. Exp. Med. 143, 1283–1288 (1976).
Bevan, M. J. Minor H antigens introduced on H-2 different stimulating cells cross-react at the cytotoxic T cell level during in vivo priming. J. Immunol. 117, 2233–2238 (1976).
Guermonprez, P. & Amigorena, S. Pathways for antigen cross presentation. Springer Semin. Immunopathol. 26, 257–271 (2005).
Hill, A. et al. Herpes simplex virus turns off the TAP to evade host immunity. Nature 375, 411–415 (1995).
Galocha, B. et al. The active Site of ICP47, a herpes simplex virus-encoded inhibitor of the major histocompatibility complex (MHC)-encoded peptide transporter associated with antigen processing (TAP), maps to the NH2-terminal 35 residues. J. Exp. Med. 185, 1565–1572 (1997).
Ackerman, A. L., Giodini, A. & Cresswell, P. A role for the endoplasmic reticulum protein retrotranslocation machinery during crosspresentation by dendritic cells. Immunity 25, 607–617 (2006). This study shows that the ER dislocation machinery participates in cross-presentation in DCs. Using an cell-free model, p97, a protein involved in ER dislocation, was the only cytosolic cofactor required for protein export from isolated phagosomes.
Loureiro, J. & Ploegh, H. L. Antigen presentation and the ubiquitin-proteasome system in host–pathogen interactions. Adv. Immunol. 92, 225–305 (2006).
Desjardins, M. ER-mediated phagocytosis: a new membrane for new functions. Nature Rev. Immunol. 3, 280–291 (2003).
Gagnon, E. et al. Endoplasmic reticulum-mediated phagocytosis is a mechanism of entry into macrophages. Cell 110, 119–131 (2002). This study uses electron microscopy to show that ER components are found on the surface of phagosomes, which suggests that the ER is directly involved in phagosome formation and maturation.
Ackerman, A. L., Kyritsis, C., Tampe, R. & Cresswell, P. Early phagosomes in dendritic cells form a cellular compartment sufficient for cross presentation of exogenous antigens. Proc. Natl Acad. Sci. USA 100, 12889–12894 (2003).
Guermonprez, P. et al. ER-phagosome fusion defines an MHC class I cross-presentation compartment in dendritic cells. Nature 425, 397–402 (2003). Cross-presentation occurs in a specialized compartment composed of a mix of ER and phagosome components. This study shows that peptides are transported through the TAP complex and access MHC class I molecules.
Houde, M. et al. Phagosomes are competent organelles for antigen cross-presentation. Nature 425, 402–406 (2003). This paper shows that the phagosome is endowed with the ability to load MHC class I molecules with peptides to engage CD8+ T cells.
Touret, N. et al. Quantitative and dynamic assessment of the contribution of the ER to phagosome formation. Cell 123, 157–170 (2005). Using biochemical, fluorescence imaging and electron microscopy techniques, this study identified that the plasma membrane donates most of the membrane to newly formed phagosomes, which was in sharp contrast with the conclusions of reference 71.
Wiertz, E. J. et al. The human cytomegalovirus US11 gene product dislocates MHC class I heavy chains from the endoplasmic reticulum to the cytosol. Cell 84, 769–779 (1996).
Koopmann, J. O. et al. Export of antigenic peptides from the endoplasmic reticulum intersects with retrograde protein translocation through the Sec61p channel. Immunity 13, 117–127 (2000).
Bernardi, K. M., Forster, M. L., Lencer, W. I. & Tsai, B. Derlin-1 facilitates the retro-translocation of cholera toxin. Mol. Biol. Cell 19, 877–884 (2008).
Mueller, B., Lilley, B. N. & Ploegh, H. L. SEL1L, the homologue of yeast Hrd3p, is involved in protein dislocation from the mammalian ER. J. Cell Biol. 175, 261–270 (2006).
Loureiro, J. et al. Signal peptide peptidase is required for dislocation from the endoplasmic reticulum. Nature 441, 894–897 (2006).
Mueller, B., Klemm, E. J., Spooner, E., Claessen, J. H. & Ploegh, H. An extended mammalian membrane protein complex that mediates glycoprotein dislocation. Proc. Natl Acad. Sci. USA (in the press).
Latz, E. et al. TLR9 signals after translocating from the ER to CpG DNA in the lysosome. Nature Immunol. 5, 190–198 (2004).
Kim, Y. M., Brinkmann, M. M., Paquet, M. E. & Ploegh, H. L. UNC93B1 delivers nucleotide-sensing Toll-like receptors to endolysosomes. Nature 452, 234–238 (2008).
Ploegh, H. L. A lipid-based model for the creation of an escape hatch from the endoplasmic reticulum. Nature 448, 435–438 (2007).
Barba, G. et al. Hepatitis C virus core protein shows a cytoplasmic localization and associates to cellular lipid storage droplets. Proc. Natl Acad. Sci. USA 94, 1200–1205 (1997).
Ohsaki, Y., Cheng, J., Fujita, A., Tokumoto, T. & Fujimoto, T. Cytoplasmic lipid droplets are sites of convergence of proteasomal and autophagic degradation of apolipoprotein B. Mol. Biol. Cell 17, 2674–2683 (2006).
Herce, H. D. & Garcia, A. E. Molecular dynamics simulations suggest a mechanism for translocation of the HIV-1 TAT peptide across lipid membranes. Proc. Natl Acad. Sci. USA 104, 20805–20810 (2007).
Smith, A. E. & Helenius, A. How viruses enter animal cells. Science 304, 237–242 (2004).
Peisajovich, S. G., Epand, R. F., Epand, R. M. & Shai, Y. Sendai virus N-terminal fusion peptide consists of two similar repeats, both of which contribute to membrane fusion. Eur. J. Biochem. 269, 4342–4350 (2002).
Rosenbluh, J. et al. Translocation of histone proteins across lipid bilayers and mycoplasma membranes. J. Mol. Biol. 345, 387–400 (2005).
Darji, A., Chakraborty, T., Wehland, J. & Weiss, S. TAP-dependent major histocompatibility complex class I presentation of soluble proteins using listeriolysin. Eur. J. Immunol. 27, 1353–1359 (1997).
Lilley, B. N., Gilbert, J. M., Ploegh, H. L. & Benjamin, T. L. Murine polyomavirus requires the endoplasmic reticulum protein Derlin-2 to initiate infection. J. Virol. 80, 8739–8744 (2006).
Daniels, R., Rusan, N. M., Wadsworth, P. & Hebert, D. N. SV40 VP2 and VP3 insertion into ER membranes is controlled by the capsid Protein VP1: implications for DNA translocation out of the ER. Mol. Cell 24, 955–966 (2006).
Tucker, S. C. & Casadevall, A. Replication of Cryptococcus neoformans in macrophages is accompanied by phagosomal permeabilization and accumulation of vesicles containing polysaccharide in the cytoplasm. Proc. Natl Acad. Sci. USA 99, 3165–3170 (2002).
Alvarez, M. & Casadevall, A. Phagosome extrusion and host-cell survival after Cryptococcus neoformans phagocytosis by macrophages. Curr. Biol. 16, 2161–2165 (2006).
Ma, H., Croudace, J. E., Lammas, D. A. & May, R. C. Expulsion of live pathogenic yeast by macrophages. Curr. Biol. 16, 2156–2160 (2006).
De Duve, C. & Wattiaux, R. Functions of lysosomes. Annu. Rev. Physiol. 28, 435–492 (1966).
Van Dyke, R. W. Acidification of rat liver lysosomes: quantitation and comparison with endosomes. Am. J. Physiol. 265, C901–C917 (1993).
Carbone, F. R. & Bevan, M. J. Class I-restricted processing and presentation of exogenous cell-associated antigen in vivo. J. Exp. Med. 171, 377–387 (1990).
Werneburg, N. W., Guicciardi, M. E., Bronk, S. F. & Gores, G. J. Tumor necrosis factor-α-associated lysosomal permeabilization is cathepsin B dependent. Am. J. Physiol. Gastrointest. Liver Physiol. 283, G947–G956 (2002).
Luke, C. J. et al. An intracellular Serpin regulates necrosis by inhibiting the induction and sequelae of lysosomal injury. Cell 130, 1108–1119 (2007).
Pfeifer, J. D. et al. Phagocytic processing of bacterial antigens for class I MHC presentation to T cells. Nature 361, 359–362 (1993).
Bachmann, M. F. et al. TAP1-independent loading of class I molecules by exogenous viral proteins. Eur. J. Immunol. 25, 1739–1743 (1995).
Liu, T. et al. TAP peptide transporter-independent presentation of heat-killed Sendai virus antigen on MHC class I molecules by splenic antigen-presenting cells. J. Immunol. 159, 5364–5371 (1997).
Schirmbeck, R., Bohm, W. & Reimann, J. Stress protein (hsp73)-mediated, TAP-independent processing of endogenous, truncated SV40 large T antigen for Db-restricted peptide presentation. Eur. J. Immunol. 27, 2016–2023 (1997).
Lizee, G. et al. Control of dendritic cell cross-presentation by the major histocompatibility complex class I cytoplasmic domain. Nature Immunol. 4, 1065–1073 (2003).
Yeager, M. & Harris, A. L. Gap junction channel structure in the early 21st century: facts and fantasies. Curr. Opin. Cell Biol. 19, 521–528 (2007).
Neijssen, J., Pang, B. & Neefjes, J. Gap junction-mediated intercellular communication in the immune system. Prog. Biophys. Mol. Biol. 94, 207–218 (2007). This study shows that gap junctions supply peptides of a relative molecular mass of up to approximately 1,800 daltons to neighbouring cells.
Neijssen, J. et al. Cross-presentation by intercellular peptide transfer through gap junctions. Nature 434, 83–88 (2005).
Sykulev, Y., Joo, M., Vturina, I., Tsomides, T. J. & Eisen, H. N. Evidence that a single peptide-MHC complex on a target cell can elicit a cytolytic T cell response. Immunity 4, 565–571 (1996).
Mizushima, N., Levine, B., Cuervo, A. M. & Klionsky, D. J. Autophagy fights disease through cellular self-digestion. Nature 451, 1069–1075 (2008).
Komatsu, M. et al. Impairment of starvation-induced and constitutive autophagy in Atg7-deficient mice. J. Cell Biol. 169, 425–434 (2005).
Komatsu, M. et al. Loss of autophagy in the central nervous system causes neurodegeneration in mice. Nature 441, 880–884 (2006).
Hara, T. et al. Suppression of basal autophagy in neural cells causes neurodegenerative disease in mice. Nature 441, 885–889 (2006).
Pierre, P. Dendritic cells, DRiPs, and DALIS in the control of antigen processing. Immunol. Rev. 207, 184–190 (2005).
Szeto, J. et al. ALIS are stress-induced protein storage compartments for substrates of the proteasome and autophagy. Autophagy 2, 189–199 (2006).
Rodgers, J. R. & Cook, R. G. MHC class Ib molecules bridge innate and acquired immunity. Nature Rev. Immunol. 5, 459–471 (2005).
Gutierrez, M. G. et al. Autophagy is a defense mechanism inhibiting BCG and Mycobacterium tuberculosis survival in infected macrophages. Cell 119, 753–766 (2004). Phagosomes containing M. tuberculosis evade fusion with lysosomes. This paper shows that autophagy pathways overcome this block and might show a general pathway for pathogen control in APCs.
Yorimitsu, T. & Klionsky, D. J. Eating the endoplasmic reticulum: quality control by autophagy. Trends Cell Biol. 17, 279–285 (2007).
Mijaljica, D., Prescott, M. & Devenish, R. J. Endoplasmic reticulum and Golgi complex: contributions to, and turnover by, autophagy. Traffic 7, 1590–1595 (2006).
Yorimitsu, T., Nair, U., Yang, Z. & Klionsky, D. J. Endoplasmic reticulum stress triggers autophagy. J. Biol. Chem. 281, 30299–30304 (2006).
Bernales, S., McDonald, K. L. & Walter, P. Autophagy counterbalances endoplasmic reticulum expansion during the unfolded protein response. PLoS Biol. 4, e423 (2006).
Ogata, M. et al. Autophagy is activated for cell survival after endoplasmic reticulum stress. Mol. Cell. Biol. 26, 9220–9231 (2006).
Dorn, B. R., Dunn, W. A. Jr & Progulske-Fox, A. Bacterial interactions with the autophagic pathway. Cell. Microbiol. 4, 1–10 (2002).
Ling, Y. M. et al. Vacuolar and plasma membrane stripping and autophagic elimination of Toxoplasma gondii in primed effector macrophages. J. Exp. Med. 203, 2063–2071 (2006).
Zhao, Y., Wilson, D., Matthews, S. & Yap, G. S. Rapid elimination of Toxoplasma gondii by γ-interferon-primed mouse macrophages is independent of CD40 signaling. Infect. Immun. 75, 4799–4803 (2007).
Andrade, R. M., Wessendarp, M., Gubbels, M. J., Striepen, B. & Subauste, C. S. CD40 induces macrophage anti-Toxoplasma gondii activity by triggering autophagy-dependent fusion of pathogen-containing vacuoles and lysosomes. J. Clin. Invest. 116, 2366–2377 (2006).
Luder, C. G. & Seeber, F. Toxoplasma gondii and MHC-restricted antigen presentation: on degradation, transport and modulation. Int. J. Parasitol. 31, 1355–1369 (2001).
Nakagawa, I. et al. Autophagy defends cells against invading group A Streptococcus. Science 306, 1037–1040 (2004).
Birmingham, C. L., Smith, A. C., Bakowski, M. A., Yoshimori, T. & Brumell, J. H. Autophagy controls Salmonella infection in response to damage to the Salmonella-containing vacuole. J. Biol. Chem. 281, 11374–11383 (2006).
Orvedahl, A. & Levine, B. Viral evasion of autophagy. Autophagy 4, 280–285 (2008).
Ogawa, M. & Sasakawa, C. Bacterial evasion of the autophagic defense system. Curr. Opin. Microbiol 9, 62–68 (2006).
Orvedahl, A. et al. HSV-1 ICP34.5 confers neurovirulence by targeting the Beclin 1 autophagy protein. Cell Host Microbe 1, 23–35 (2007). This study shows that the herpes simplex virus type 1 protein ICP34.5 binds to the mammalian autophagy protein beclin-1 and inhibits its autophagy function, which indicates that microorganisms can target key molecules in autophagy to evade immunity.
Ogawa, M. et al. Escape of intracellular Shigella from autophagy. Science 307, 727–731 (2005).
Mizushima, N., Yamamoto, A., Matsui, M., Yoshimori, T. & Ohsumi, Y. In vivo analysis of autophagy in response to nutrient starvation using transgenic mice expressing a fluorescent autophagosome marker. Mol. Biol. Cell 15, 1101–1111 (2004).
Kyewski, B. & Klein, L. A central role for central tolerance. Annu. Rev. Immunol. 24, 571–606 (2006).
Lee, J. W. et al. Peripheral antigen display by lymph node stroma promotes T cell tolerance to intestinal self. Nature Immunol. 8, 181–190 (2007).
Hampe, J. et al. A genome-wide association scan of nonsynonymous SNPs identifies a susceptibility variant for Crohn disease in ATG16L1. Nature Genet. 39, 207–211 (2007).
Rioux, J. D. et al. Genome-wide association study identifies new susceptibility loci for Crohn disease and implicates autophagy in disease pathogenesis. Nature Genet. 39, 596–604 (2007).
Parkes, M. et al. Sequence variants in the autophagy gene IRGM and multiple other replicating loci contribute to Crohn's disease susceptibility. Nature Genet. 39, 830–832 (2007).
Harris, J. et al. T helper 2 cytokines inhibit autophagic control of intracellular Mycobacterium tuberculosis. Immunity 27, 505–517 (2007).
Acknowledgements
We thank all members of the Ploegh laboratory for their discussions and K. Artavanis-Tsakonas, E. Frickel and A. Pollington for their critical review of this manuscript. We thank B. Mueller and T. DiCesare for assistance with the original artwork. J.M.V. is funded by NIH K08 AI57999 and MGH DOM start-up funds; A.G.V. is funded by the Boehringer Ingelheim Fonds; H.L.P. is funded by RO1 GM062502, RO1 AI34893, RO1 AI057182 and R37 AI33456.
Author information
Authors and Affiliations
Supplementary information
Supplementary information S1 (table)
The core machinery of autophagy. (PDF 216 kb)
Related links
Glossary
- Endolysosomal compartments
-
Endosomes that have fused with lysosomes. This acidic environment allows degradation of antigens.
- Pathogen-associated molecular patterns
-
Molecular patterns that are found in pathogens but not in mammalian cells. Examples include terminally mannosylated and polymannosylated compounds, which bind the mannose receptor, and various microbial products, such as bacterial lipopolysaccharides, hypomethylated DNA, flagellin and double-stranded RNA, which bind Toll-like receptors.
- Endolysosomal tubules
-
Highly dynamic subcellular structures that eminate from late endocytic–lysosomal and/or phagolysosomal compartments. They are known to contain MHC class II molecules, CD63, CD82 and lysosome-associated membrane protein 1 (LAMP1), and require microtubules for movement.
- Endocytic pathway
-
A trafficking pathway used by all cells for the internalization of molecules from the plasma membrane to the endolysosomes.
- Cross-presentation
-
The ability of certain antigen-presenting cells to load peptides that are derived from exogenous antigens onto MHC class I molecules. Cross-presentation is important for the initiation of immune responses to viruses that do not infect antigen-presenting cells.
- Autophagy
-
An evolutionarily conserved process in which acidic double-membrane vacuoles, known as autophagosomes, sequester intracellular contents (such as damaged organelles and macromolecules) and target them for degradation, through fusion to lysosomes. This process does not involve direct transport through the endocytic or vacuolar protein sorting pathways.
- Phagosome
-
A membrane-delimited compartment that confines ingested material such as microorganisms following phagocytosis. Unless counteracted by a pathogen survival strategy, the phagosome matures into a hostile environment (the phagolysosome) that is designed to kill and digest microorganisms by fusing with lysosomes.
- Phagolysosome
-
An intracellular compartment that results from the fusion of phagosomes, which enclose ingested extracellular material, and lysosomes, which contain lytic enzymes.
- Tetraspanin
-
A family of transmembrane proteins that have four transmembrane domains and two extracellular domains of different sizes, which are defined by several conserved amino acids in the transmembrane domains. Their function is not clearly known, but they seem to interact with many other transmembrane proteins and to form large multimeric protein networks.
- Immunological synapse
-
A large junctional structure that is formed at the cell surface between a T cell and an antigen-presenting cell. Important molecules that are involved in T-cell activation — including the T-cell receptor, numerous signal-transduction molecules and molecular adaptors — accumulate in an orderly manner at this site. Immunological synapses are now known to also form between other types of immune cells: for example, between dendritic cells and natural killer cells.
- Cathepsins
-
Proteases that are mostly located in lysosomes and lysosome-like organelles and can be divided into cysteine, aspartate and serine cathepsin subgroups according to their active-site amino acid.
- RAB
-
A cytosolic protein that has GTPase activity, which, in the GTP-bound form, associates with membranes. Different RAB proteins associate with different intracellular compartments — for example, RAB5 associates with early endosomes, RAB7 with late endosomes and RAB11 with recycling endosomes.
- Chaperone-mediated autophagy
-
The import and degradation of soluble cytosolic proteins by chaperone-dependent, direct translocation across the lysosomal membrane.
- Microautophagy
-
The uptake and degradation of cytoplasm by invagination of the lysosomal membrane.
- Macroautophagy
-
(Also known as autophagy). The largely non-specific autophagic sequestration of cytoplasm into a double-membrane-delimited compartment (an autophagosome) of non-lysosomal origin. Note that certain proteins, organelles and pathogens might be selectively degraded via macroautophagy.
- Lysosomal degradation
-
The digestion of macromolecules in lysosomal organelles, which are the terminal organelles of degradative pathways, such as phagosomal, endosomal and autophagy pathways.
- Endosomes
-
Membrane-delimited compartments that contain material ingested by endocytosis. Some material will be recycled to the cell surface whereas some cargo will transit to late endosomes and eventually fuse with lysosomes to form endolysosomes. Endosomes may also fuse with phagosomes to allow maturation of the phagosomal compartment.
- Central tolerance
-
Deletion of self-reactive T cells in the thymus.
- Peripheral tolerance
-
Control of self-reactive T cells in the periphery.
- Lipid rafts
-
Microdomains of the cell membrane that are enriched in sphingolipids. Several membrane-associated signalling molecules are concentrated in these rafts.
- Exocytosis
-
The release of material contained within vesicles by fusion of the vesicles with the plasma membrane.
- SNARE
-
(Soluble-N-ethylmaleimide-sensitive-factor accessory-protein receptor). A class of proteins that is required for membrane fusion events that occur in the course of vesicle trafficking and secretion.
- TAP complex
-
A heterodimeric complex composed of transporter associated with antigen processing 1 (TAP1) and TAP2 in the membrane of the endoplasmic reticulum. The complex transports peptides from the cytoplasm to the endoplasmic reticulum, where peptides can be loaded onto MHC class I molecules. Without these peptides, MHC class I molecules are unstable and are much less likely to transit to the cell surface or to remain there.
- Sphingosine
-
An amino alcohol that can be linked to a fatty acid via the amino group to form the basic structure of sphingolipids.
- Necrosis
-
A form of cell death that results from a decline in cellular ATP or ADP levels below the concentration that is required to maintain cellular organization and integrity.
- Aggresome
-
Proteinaceous inclusion body that forms when the cellular degradation machinery is impaired or overwhelmed and that leads to an accumulation of protein for disposal.
- Unfolded-protein response
-
A response that increases the ability of the endoplasmic reticulum to fold and translocate proteins, decreases the synthesis of proteins, and causes the arrest of the cell cycle and apoptosis.
Rights and permissions
About this article
Cite this article
Vyas, J., Van der Veen, A. & Ploegh, H. The known unknowns of antigen processing and presentation. Nat Rev Immunol 8, 607–618 (2008). https://doi.org/10.1038/nri2368
Issue Date:
DOI: https://doi.org/10.1038/nri2368