Abstract
Therapeutic targeting of tumours on the basis of molecular analysis is a new paradigm for cancer treatment but has yet to fulfil expectations. For many solid tumours, targeted therapeutics, such as inhibitors of oncogenic kinase pathways, elicit predominantly disease-stabilizing, cytostatic responses, rather than tumour regression. Combining oncogenic kinase inhibitors with direct activators of the apoptosis machinery, such as the BH3 mimetic ABT-737, may unlock potent anti-tumour potential to produce durable clinical responses with less collateral damage.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Weinstein, I. B. Cancer. Addiction to oncogenes — the Achilles heal of cancer. Science 297, 63–64 (2002).
Druker, B. J. Translation of the Philadelphia chromosome into therapy for CML. Blood 112, 4808–4817 (2008).
Nyati, M. K., Morgan, M. A., Feng, F. Y. & Lawrence, T. S. Integration of EGFR inhibitors with radiochemotherapy. Nature Rev. Cancer 6, 876–885 (2006).
Herbst, R. S. & Sandler, A. Bevacizumab and erlotinib: a promising new approach to the treatment of advanced NSCLC. Oncologist 13, 1166–1176 (2008).
Sequist, L. V. et al. First-line gefitinib in patients with advanced non-small-cell lung cancer harboring somatic EGFR mutations. J. Clin. Oncol. 26, 2442–2449 (2008).
Sordella, R., Bell, D. W., Haber, D. A. & Settleman, J. Gefitinib-sensitizing EGFR mutations in lung cancer activate anti-apoptotic pathways. Science 305, 1163–1167 (2004).
Paez, J. G. et al. EGFR mutations in lung cancer: correlation with clinical response to gefitinib therapy. Science 304, 1497–1500 (2004).
Zhang, J., Yang, P. L. & Gray, N. S. Targeting cancer with small molecule kinase inhibitors. Nature Rev. Cancer 9, 28–39 (2009).
Pao, W. et al. Acquired resistance of lung adenocarcinomas to gefitinib or erlotinib is associated with a second mutation in the EGFR kinase domain. PLoS Med. 2, e73 (2005).
Yun, C. H. et al. The T790M mutation in EGFR kinase causes drug resistance by increasing the affinity for ATP. Proc. Natl Acad. Sci. USA 105, 2070–2075 (2008).
Shah, N. P. et al. Multiple BCR–ABL kinase domain mutations confer polyclonal resistance to the tyrosine kinase inhibitor imatinib (STI571) in chronic phase and blast crisis chronic myeloid leukemia. Cancer Cell 2, 117–125 (2002).
Corbin, A. S., Buchdunger, E., Pascal, F. & Druker, B. J. Analysis of the structural basis of specificity of inhibition of the Abl kinase by STI571. J. Biol. Chem. 277, 32214–32219 (2002).
Engelman, J. A. et al. MET amplification leads to gefitinib resistance in lung cancer by activating ERBB3 signaling. Science 316, 1039–1043 (2007).
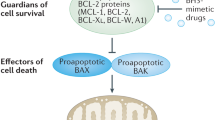
Strasser, A., O'Connor, L. & Dixit, V. M. Apoptosis signaling. Annu. Rev. Biochem. 69, 217–245 (2000).
Youle, R. J. & Strasser, A. The BCL-2 protein family: opposing activities that mediate cell death. Nature Rev. Mol. Cell Biol. 9, 47–59 (2008).
Chen, L. et al. Differential targeting of prosurvival Bcl-2 proteins by their BH3-only ligands allows complementary apoptotic function. Mol. Cell 17, 393–403 (2005).
Fesik, S. W. Promoting apoptosis as a strategy for cancer drug discovery. Nature Rev. Cancer 5, 876–885 (2005).
Kuroda, J. et al. Bim and Bad mediate imatinib-induced killing of Bcr/Abl+ leukemic cells, and resistance due to their loss is overcome by a BH3 mimetic. Proc. Natl Acad. Sci. USA 103, 14907–14912 (2006).
Kuroda, J. et al. Apoptosis-based dual molecular targeting by INNO-406, a second-generation Bcr–Abl inhibitor, and ABT-737, an inhibitor of antiapoptotic Bcl-2 proteins, against Bcr–Abl-positive leukemia. Cell Death Differ. 14, 1667–1677 (2007).
Deng, J. et al. Proapoptotic BH3-only BCL-2 family protein BIM connects death signaling from epidermal growth factor receptor inhibition to the mitochondrion. Cancer Res. 67, 11867–11875 (2007).
Oltersdorf, T. et al. An inhibitor of Bcl-2 family proteins induces regression of solid tumours. Nature 435, 677–681 (2005).
Chen, S., Dai, Y., Harada, H., Dent, P. & Grant, S. Mcl-1 down-regulation potentiates ABT-737 lethality by cooperatively inducing Bak activation and Bax translocation. Cancer Res. 67, 782–791 (2007).
Vogler, M. et al. Concurrent upregulation of BCL-XL and BCL2A1 induces ∼1000-fold resistance to ABT-737 in chronic lymphocytic leukemia. Blood 13 Nov 2008 (doi:10.1182/blood-2008-08-173310).
Chauhan, D. et al. A novel Bcl-2/Bcl-XL/Bcl-w inhibitor ABT-737 as therapy in multiple myeloma. Oncogene 26, 2374–2380 (2007).
Konopleva, M. et al. Mechanisms of apoptosis sensitivity and resistance to the BH3 mimetic ABT-737 in acute myeloid leukemia. Cancer Cell 10, 375–388 (2006).
Kline, M. P. et al. ABT-737, an inhibitor of Bcl-2 family proteins, is a potent inducer of apoptosis in multiple myeloma cells. Leukemia 21, 1549–1560 (2007).
Mason, K. D. et al. In vivo efficacy of the Bcl-2 antagonist ABT-737 against aggressive Myc-driven lymphomas. Proc. Natl Acad. Sci. USA 105, 17961–17966 (2008).
Kuroda, J. & Taniwaki, M. Involvement of BH3-only proteins in hematologic malignancies. Crit. Rev. Oncol. Hematol. 20 Nov 2008 (doi:10.1016/j.critrevonc.2008.10.004).
Huang, S. & Sinicrope, F. A. BH3 mimetic ABT-737 potentiates TRAIL-mediated apoptotic signaling by unsequestering Bim and Bak in human pancreatic cancer cells. Cancer Res. 68, 2944–2951 (2008).
Del Gaizo Moore, V. et al. Chronic lymphocytic leukemia requires BCL2 to sequester prodeath BIM, explaining sensitivity to BCL2 antagonist ABT-737. J. Clin. Invest. 117, 112–121 (2007).
Cragg, M. S. et al. Treatment of B-RAF mutant human tumor cells with a MEK inhibitor requires Bim and is enhanced by a BH3 mimetic. J. Clin. Invest. 118, 3651–3659 (2008).
van Delft, M. F. et al. The BH3 mimetic ABT-737 targets selective Bcl-2 proteins and efficiently induces apoptosis via Bak/Bax if Mcl-1 is neutralized. Cancer Cell 10, 389–399 (2006).
Miller, L. A. et al. BH3 mimetic ABT-737 and a proteasome inhibitor synergistically kill melanomas through noxa-dependent apoptosis. J. Invest. Dermatol. 6 Nov 2008 (doi: 10.1038/jid.2008.327).
Lin, X. et al. 'Seed' analysis of off-target siRNAs reveals an essential role of Mcl-1 in resistance to the small-molecule Bcl-2/Bcl-XL inhibitor ABT-737. Oncogene 26, 3972–3979 (2007).
Tahir, S. K. et al. Influence of Bcl-2 family members on the cellular response of small-cell lung cancer cell lines to ABT-737. Cancer Res. 67, 1176–1183 (2007).
Letai, A. G. Diagnosing and exploiting cancer's addiction to blocks in apoptosis. Nature Rev. Cancer 8, 121–132 (2008).
Lessene, G., Czabotar, P. E. & Colman, P. M. BCL-2 family antagonists for cancer therapy. Nature Rev. Drug Discov. 7, 989–1000 (2008).
Lock, R. et al. Initial testing (stage 1) of the BH3 mimetic ABT-263 by the pediatric preclinical testing program. Pediatr. Blood Cancer 50, 1181–1189 (2008).
Shoemaker, A. R. et al. Activity of the Bcl-2 family inhibitor ABT-263 in a panel of small cell lung cancer xenograft models. Clin. Cancer Res. 14, 3268–3277 (2008).
Park, C. M. et al. Discovery of an orally bioavailable small molecule inhibitor of prosurvival B-cell lymphoma 2 proteins. J. Med. Chem. 51, 6902–6915 (2008).
Tse, C. et al. ABT-263: a potent and orally bioavailable Bcl-2 family inhibitor. Cancer Res. 68, 3421–3428 (2008).
Ackler, S. et al. ABT-263 and rapamycin act cooperatively to kill lymphoma cells in vitro and in vivo. Mol. Cancer Ther. 7, 3265–3274 (2008).
Trudel, S. et al. The Bcl-2 family protein inhibitor, ABT-737, has substantial antimyeloma activity and shows synergistic effect with dexamethasone and melphalan. Clin. Cancer Res. 13, 621–629 (2007).
Kutuk, O. & Letai, A. Alteration of the mitochondrial apoptotic pathway is key to acquired paclitaxel resistance and can be reversed by ABT-737. Cancer Res. 68, 7985–7994 (2008).
Hann, C. L. et al. Therapeutic efficacy of ABT-737, a selective inhibitor of BCL-2, in small cell lung cancer. Cancer Res. 68, 2321–2328 (2008).
Kohl, T. M. et al. BH3 mimetic ABT-737 neutralizes resistance to FLT3 inhibitor treatment mediated by FLT3-independent expression of BCL2 in primary AML blasts. Leukemia 21, 1763–1772 (2007).
Stolz, C. et al. Targeting Bcl-2 family proteins modulates the sensitivity of B-cell lymphoma to rituximab-induced apoptosis. Blood 112, 3312–3321 (2008).
Whitecross, K. F. et al. Defining the target specificity of ABT-737 and synergistic anti-tumor activities in combination with histone deacetylase inhibitors. Blood 113, 1982–1991 (2009).
Cragg, M. S., Kuroda, J., Puthalakath, H., Huang, D. C. & Strasser, A. Gefitinib-induced killing of NSCLC cell lines expressing mutant EGFR requires BIM and can be enhanced by BH3 mimetics. PLoS Med. 4, 1681–1689; discussion 1690 (2007).
Gong, Y. et al. Induction of BIM is essential for apoptosis triggered by EGFR kinase inhibitors in mutant EGFR-dependent lung adenocarcinomas. PLoS Med. 4, e294 (2007).
Paoluzzi, L. et al. The BH3-only mimetic ABT-737 synergizes the antineoplastic activity of proteasome inhibitors in lymphoid malignancies. Blood 112, 2906–2916 (2008).
Costa, D. B. et al. BIM mediates EGFR tyrosine kinase inhibitor-induced apoptosis in lung cancers with oncogenic EGFR mutations. PLoS Med. 4, 1669–1679; discussion 1680 (2007).
Shepherd, F. A. et al. Erlotinib in previously treated non-small-cell lung cancer. N. Engl. J. Med. 353, 123–132 (2005).
Olejniczak, E. T. et al. Integrative genomic analysis of small-cell lung carcinoma reveals correlates of sensitivity to bcl-2 antagonists and uncovers novel chromosomal gains. Mol. Cancer Res. 5, 331–339 (2007).
Kang, M. H. et al. Activity of vincristine, L-ASP, and dexamethasone against acute lymphoblastic leukemia is enhanced by the BH3-mimetic ABT-737 in vitro and in vivo. Blood 110, 2057–2066 (2007).
Tagscherer, K. E. et al. Apoptosis-based treatment of glioblastomas with ABT-737, a novel small molecule inhibitor of Bcl-2 family proteins. Oncogene 27, 6646–6656 (2008).
Song, J. H., Kandasamy, K. & Kraft, A. S. ABT-737 induces expression of the death receptor 5 and sensitizes human cancer cells to TRAIL-induced apoptosis. J. Biol. Chem. 283, 25003–25013 (2008).
Acknowledgements
The authors would like to acknowledge their colleagues in the Cancer Sciences Division, Southampton, UK and The Walter and Eliza Hall Institute, Melbourne, Australia, and apologize to authors whose work has not been cited in this short article for space reasons.
Author information
Authors and Affiliations
Corresponding authors
Related links
Related links
DATABASES
National Cancer Institute Drug Dictionary
FURTHER INFORMATION
Rights and permissions
About this article
Cite this article
Cragg, M., Harris, C., Strasser, A. et al. Unleashing the power of inhibitors of oncogenic kinases through BH3 mimetics. Nat Rev Cancer 9, 321–326 (2009). https://doi.org/10.1038/nrc2615
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrc2615
This article is cited by
-
Inhibitors Targeting the F-BOX Proteins
Cell Biochemistry and Biophysics (2023)
-
BCL-2 protein family: attractive targets for cancer therapy
Apoptosis (2023)
-
Noxa and Mcl-1 expression influence the sensitivity to BH3-mimetics that target Bcl-xL in patient-derived glioma stem cells
Scientific Reports (2022)
-
Removal of BFL-1 sensitises some melanoma cells to killing by BH3 mimetic drugs
Cell Death & Disease (2022)
-
Design and optimisation of dendrimer-conjugated Bcl-2/xL inhibitor, AZD0466, with improved therapeutic index for cancer therapy
Communications Biology (2021)